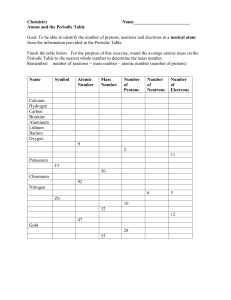
Atoms and the Periodic Table Mid-Topic Test Name:……………. 1. Protons and neutrons are found in the ……………………………… Electrons are found in…………………………………………………. (2) 2. Draw a picture of a chlorine atom: Atomics Number: 17 Mass Number: 35 Number of protons = Number of neutrons = Number of electrons = (6) 3. With reference to protons and electrons, explain why atoms are neutral. (2) 4. Fill out the Periodic Table (include the atomic number, symbol and electron configuration in each box): Grp:1 1 H 2 3 3 Li 4 Be 5 B 11 12 Mg 13 4 5 6 C 7 N 8 O 2, 4 2,8, 1 19 K 2, 8, 4 F 2, 6 15 Si 2, 8, 3 7 17 S 2, 8, 5 8 2 He 10 Ne 2,8 18 Ar 2, 8, 6 20 2, 8, 8, 2 (20) Look at Li, Na, K. What similarities do you notice about the outer electrons? (1) Look at Oxygen and Sulfur. What similarities do you notice about the number of electrons in their last shell? (1) Which 2 elements have 7 electrons in their outer shell?........................................... (1) Look at the periodic table, can you guess which other 2 elements also have 7 electrons in their outer shell? (2) 4. Fill in the table: Element Symbol Remember: Atomic Number Potassium Mass = proton + neutrons Number of neutrons = mass - protons Number of Protons 19 Number of Neutrons 20 Fluorine Mass Electron Configuration 19 Magnesium 12 Sulfur 32 (8) 5. Copper has the atomic number of 29 and a mass of 63. Number of Protons: Number of Neutrons: Number of Electrons: (3) 6. State the elements in these common compounds: NaCl………………………………......................................................................... KOH………………………………......................................................................... CuSO4……………………………......................................................................... CO2…….…………………………......................................................................... (8) 7. Look at the periodic table: What is the atomic number and mass of: Gold (Au): Atomic Number Mercury (Hg): Atomic Number Mass: Mass: (4) 8. Draw and describe in words the Atomic structure of sodium.