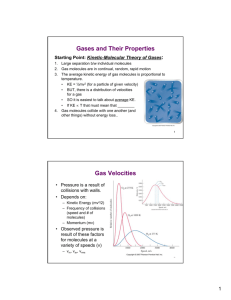
Gases Properties of Gases • Gases adopt the volume and shape of their containers • Gases are easy compressed, whereas liquids and solids are not • Gases placed within the same container mix uniformly and completely • Gases have much lower densities than liquids and solids Pressure • Gas molecules exert a pressure when they hit the surface of their container • Unit of pressure: force N 2 Pa (pascal) surface m • Atmospheric pressure (1 atm): 1 atm = 101 325 Pa = 1.01325 x 102 kPa Atmospheric Pressure • Atmospheric pressure is the pressure exerted by the column of air situated above a surface • We do not feel the atmospheric pressure because we are physiologically adapted for it Atmospheric Pressure Gas Pressure Boyle’s Law • Boyle observed that the volume of a gas decreases (increases) when the pressure exerted on the gas increases (decreases) • Boyle’s Law states that the volume of a given mass of gas at constant temperature is inversely proportional to its pressure Boyle’s Law • At constant temperature: Charles’ and Gay-Lussac’s Law • Charles and Gay-Lussac observed that at constant pressure for a given mass of gas, the volume increases with increasing temperature and decreases with decreasing temperature The Kelvin Scale • Plots of V vs. T have different slopes for different pressures, but the extrapolation of each line to V = 0 crosses the T axis at T = -273.15 oC • Kelvin proposed that -273.15 oC is the lowest temperature that can be achieved, i.e., the absolute zero • The Kelvin Scale: T(K) = t(oC) + 273.15 oC • N.B. These formulas are only valid if we express T in kelvins!!! Avogadro’s Law • Avogadro’s hypothesis (1811): At the same temperature and same pressure, equal volumes of different gases contain the same number of molecules • Avogadro's law also says that, at constant pressure and temperature, the volume of a gas is directly proportional to the number of moles of gas present Avogadro’s Law • Avogadro 's law insists that when two gases react together and the product(s) is a gas : -The ratio between the volumes of reactants is a simple number - The ratio between the total volume of reactants and the total volume of products is a simple number Ideal Gas Equation 1 • Boyle’ Law: V P (n, T constant) • Charles’ Law: V T (n, P constant) • Avogadro’s Law: V n (P, T constant) Ideal Gas Equation • We can put the three laws together: nT V P nT VR P • Ideal Gas Equation: PV = nRT • R is the ideal gas constant Ideal Gases • An ideal gas is a theoretical gas in which the pressure, volume, and temperature obey the ideal gas equation - There is no attraction or repulsion between the molecules of an ideal gas - The volume of the molecules in an ideal gas is negligible compared to the volume of the container (i.e., the space in the container is empty) • The approximation of an ideal gas is better at high T and low P Ideal Gas Constant • At 0 oC and 1 atm (STP: standard temperature and pressure), most real gases behave like ideal gases • It is observed that for one mole of any gas at STP, the volume is around 22.414 L PV (1 atm)(22.414 L) R nT (1 mol)(273.15 K) R 0.082 057 L atm • 8.3145 • R is the ideal gas constant K mol Ideal Gas Equation • Example: Calculate the volume (in litres) occupied by 2.12 moles of nitric oxide (NO) at 6.54 atm and 76oC. • Solution: nRT V P T = 349 K (2.12 mol)(0.0821 L atm 6.54 atm V = 9.29 L )(349 K) K mol Ideal Gas Equation • Example: What volume is occupied by 49.8 g of HCl at STP? • Solution: T = 273.15 K and P = 1 atm n (49.8 g) 1.366 mol (1.008 g/mol 35.45 g/mol) L atm )(273.15 K) nRT (1.366 mol)(0.0821 K mol V P 1.00 atm V = 30.6 L Ideal Gas Equation • A modified form of the ideal gas equation is sometimes more useful to study variations of P , V, T for a fixed amount of gas P1V1 PV PV P V R 2 2 1 1 2 2 n1T1 n 2T2 n1T1 n 2T2 • If the moles of a gas does not change: P1V1 P2 V2 T1 T2 Ideal Gas Equation • Example: A sample of radioactive radon gas that had an initial volume of 4.0 L, an initial pressure of 1.2 atm and a temperature of 66oC, undergoes a change that changes its volume and temperature to 1.7 L and 42oC . What is the final pressure? The number of moles remains constant. • Solution: T1 = 339 K and P2 P2 T2 = 315 K P1V1 T2 T1 V2 (1.2 atm)(4.0 L) 315 K 2.6 atm 339 K 1.7 L The Density and Molar Mass of a Gas • The ideal gas law allows us to determine the density (r) or molar mass (M) of a gas: n P PV nRT V RT m m P n M MV RT m PM ρ V RT mRT ρRT M PV P The Density and Molar Mass of a Gas • Example: The density of a gaseous organic compound is 3.38 g/L, at 40oC et at 1.97 atm. What is its molar mass? • Solution: M ρRT P (3.38 g )(0.0821 L atm )(313 K) L mol K M 1.97atm M 44.1 g/mol Dalton’s Law of Partial Pressures • The equations we have seen so far are for pure gases • Dalton’s Law of Partial Pressures states that the total pressure of a mixture of gases is the sum of the partial pressures of the gases that make up the mixture • The partial pressure of a gas in a mixture is the pressure that the gas would exert if it was alone • The Law of Partial Pressures is consistent with the lack of attractions/repulsion in an ideal gas Dalton’s Law of Partial Pressures • The partial pressure of gas A, PA, in a mixtures of gases is n A RT PA V • The Law of Partial Pressures states that the total pressure, PT, is given by PT PA PB PC n A RT n B RT n C RT PT V V V RT RT PT (n A n B n C ) nT V V Dalton’s Law of Partial Pressures nA nA XA nT nA nB nC n A RT PA nA V XA n RT T PT nT V PA X A PT Dalton’s Law of Partial Pressures • Example: A sample of natural gas contains 8.24 mol of CH4, 0.421 mol of C2H6 and 0.116 mol of C3H8. If the total pressure is 1.37 atm, what is the partial pressure of each gas? n T 8.24 0.421 0.116 8.78 mol • Solution: PCH 4 8.24 mol X CH 4 0.938 8.78 mol X CH 4 PT 0.938 1.37 atm 1.29 atm • In the same way, we can calculate the partial pressures of C2H6 and C3H8 to be 0.0657 atm and 0.0181 atm, respectively Dalton’s Law of Partial Pressures • We often collect a gas produced within a reaction by the displacement of water • e.g.; KClO3(s) decomposes to give KCl(s) and O2(g), and because O2(g) is not very soluble in water, the O2(g) displaces the water in the inverted bottle Dalton’s Law of Partial Pressures • In the inverted bottle, above water, one should not forget the pressure due to water vapour PT PO 2 PH 2O PO 2 PT PH 2O • The partial pressure due to water vapour is well known as a function of temperature • The same principle applies to each gas which is insoluble in water Dalton’s Law of Partial Pressures • Example: Hydrogen is prepared by reacting calcium with water. Hydrogen is collected using an assembly like the one just seen. The volume of gas collected at 30 oC and at 988 mm Hg is 641 mL. What is the mass of hydrogen that was produced? The pressure of water vapour at 30oC is 31.82 mm Hg. • Solution: PH 2 988 mm Hg 31.82 mm Hg 956 mm Hg 956 mm Hg PH 2 V n H 2 RT (1.258 atm)(0.641 L) 0.0324 mol L atm RT (0.0821 )(303 K) K mol 2.016 g 0.0324 mol 0.0653 g 1 mol n H2 mH2 PH 2 V 1atm 1.258 atm 760 mm Hg The Kinetic Theory of Gases • We arrived at the ideal gas equation empirically, i.e., we don’t know why PV = nRT • Maxwell and Boltzmann tried to explain the physical properties of a gas from the movements of its individual molecules • In an ideal gas, there are no attractions or repulsion between the gas molecules, so the energy of the gas comes entirely from the kinetic energy of individual molecules • The kinetic energy of a molecule is dependent only on the mass and velocity of that molecule Kinetic Theory of Gases: Assumptions • A gas is formed of molecules separated from one another distances much greater than their own dimensions, i.e., the volume of a molecule is negligible • Gas molecules are in constant motion in all directions, and they frequently collide and these collisions are perfectly elastic, i.e., the total (kinetic) energy of all the molecules of a system is constant • Gas molecules exert no attractive or repulsive force between each other • The average kinetic energy of gas molecules is proportional to the temperature of the gas in Kelvins, and two gases at the same temperature have the same average kinetic energy The Kinetic Theory of Gases • The average kinetic energy of gas molecules is given by ____ 2 1 ____2 Ec m v 2 where v is the average velocity squared ____ 2 2 2 v v v 2 N v2 1 N • The last assumption says that: 1 ____2 m v T 2 where k is the Boltzmann constant 1 ____2 m v kT 2 The Kinetic Theory of Gases • The Maxwell distribution describes the probability of finding a molecule with a given speed at a given temperature • The most probable speed increases as the temperature is increased • There is a greater dispersion in speeds at high temperature The Kinetic Theory of Gases • With the Kinetic Theory of Gases and the Maxwell distribution, we ____ 1 can derive the following equation: PV nM v 2 3 • But we know empirically that PV = nRT, thus ____ 1 nM v 2 nRT 3 ____ 2 v v quad ____ 2 v 3RT M 3RT M • The average quadratic velocity, vquadr, increases when T increases or the molar mass, M, decreases • So that vquadr is given in m/s (i.e., SI units), M must be given in kg/mol (SI) and R must be expressed as 8.314 J/K (SI units) The Kinetic Theory of Gases • Example: Calculate the average velocity of chlorine molecules (Cl2) in metres/seconds at 20oC. • Solution: T = 293 K and M = 70.90 g/mol = 0.07090 kg/mol v quadr (3)(8.314 J )(293 K) K mol 321 m s 0.07090 kg mol • N.B. The Kinetic Theory of Gases allows us to calculate vquadr for He and H2 and we observe that it approaches Earth’s escape velocity (1.1 x 104 m/s). Both gases can thus escape our atmosphere. • In a 5.00 L container, we have 8.22 g of O2(g) and a pressure of 1.00 atm. In another 5.00 L container, we have 8.22 g of N2(g) and a pressure of 1.00 atm. What is the average velocity (or quadratic velocity) of the molecules in each container? We put all of the O2(g) and all of the N2(g) in a third 5.00 L container that is maintained at 25.0 oC. What is the total pressure of this container? On average, do the molecules of O2(g) have more kinetic energy, the same kinetic energy, or less kinetic energy than the molecules of N2(g)? You do not need to explain your reasoning (that is to say, just provide the answer).