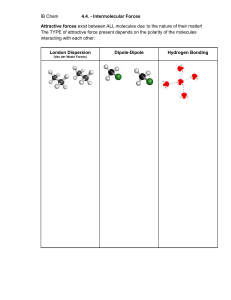
IR spectroscopy IR = Infra-red Functional groups • Functional groups are collection of atoms that attach the carbon skeleton of an organic molecule and confer specific properties • Each type of organic molecule has its own specific type of functional group • Functional groups in biological molecules play an important role in the formation of molecules like DNA, proteins, carbohydrates etc…. An example and basic principle • Carbonyl functional group in formaldehyde. • Carbonyl means C double bond O • In fact, this molecule (others as well are not static as it looks in this molecular representation. • In fact, the bonds are vibrating with some frequency at room temperature. • The vibrational frequency of the bonds is temperature dependent • When the IR radiations with the range of frequency is incident on to such sample, the radiations with the frequency equivalent to the vibrational frequency of the bonds will be absorbed (the concept of resonance) Actual IR spectra Use of Wavenumber instead Of frequency. Wavenumber tells you about how many waves fit into one Centimeter. Wavenumber is units of energy and therefore you can directly deduce the difference of energy between states Functional group region vs the fingerprint region • Functional group region: gives information about the presence of functional group present in the molecule • Fingerprint region: gives information about the exact central molecule. This is called the finger print because every individual molecule has different spectra in the finger print region. • Every individual human being have distinct finger prints. That’s why as an analogy, this region is called a finger print region where every individual molecule have distinct spectra (or metaphorically, the finger print). These finger prints can be checked through the database. Principle of IR spectroscopy and modes of molecular vibrations • IR active compounds will get IR spectra gets excited and shows vibrational, rotational spectra; which is characteristic spectra for a given functional group. • Two fundamental types of vibrations in IR spectroscopy: 1. Stretching I. Symmetrical stretching II. Asymmetrical stretching 2. I. II. III. IV. Bending Scissoring Rocking Wagging Twisting https://chem.libretexts.org/Bookshelves/Physical_and_Theoretical_Chemistry_Textbook_Maps/Supplemental_Mod ules_(Physical_and_Theoretical_Chemistry)/Spectroscopy/Vibrational_Spectroscopy/Vibrational_Modes/Number_o f_Vibrational_Modes_in_a_Molecule#:~:text=Molecular%20Vibrations,A%20molecule%20has&text=For%20a%20mode%20to%20be,(i.e.%20not%20diatomic%20molecules).&text=The%20 normal%20modes%20of%20vibration,and%20rocking%20for%20polyatomic%20molecules. Infrared spectroscopy • It is a type of absorption spectroscopy. • It is used to determine the functional groups in a given organic compound. (e.g., the detection of aldehydes, ketones, carboxylic groups etc….) • Infrared radiations are directed onto the sample. Some infrared photons are absorbed by the sample while the rest pass through (or transmit through) the sample • Vibrational rotational spectra occurs. • The essential requirements of the sample if it has to be analyzed using IR spectroscopy it should possess dipole moments • If the sample does not contain dipole moments, it means that it is IR inactive compound. Otherwise, it is IR active (means, IR spectroscopy can be used for the analysis of that compound). It will be able to absorb IR radiations of an appropriate wavenumber. • The molecules such as HF has a centre of positive charge and a centre of negative charge is said to be dipolar or to have a dipole moment • The dipolar character is represented by an arrow pointing towards the negative charge centre (the tail of the arrow points towards positive charge centre) Dipoles and Dipole moments • Dipole is the measure of separation of charge. The dipole in a bond is formed due to the difference in electronegativity of the individual atoms forming the bond. • Dipole moment is a measure of change in the dipole moment • Dipole moment of a molecule can be calculated using the following relationship • Why only polar bonds are IR active? Because the polar molecules have dipole moments. Under normal conditions, these molecules are in continuous vibrations e.g., stretching and bending. Any change in dipole moments of radiations means absorption of it by the molecule. It have been observed that this takes place by IR radiations. • Why not the vice versa? • How does the IR radiations interact with molecules based on the presence of dipoles and dipole moments? IR radiations are electromagnetic radiations • Every compound will have its distinct IR spectra. In other words, no two different compounds will have similar IR spectra. Enantiomers are exceptional in this case. • Categories of IR radiations: 1. Near IR 2. Mid IR (the one used in IR spectroscopy)\ 3. Far IR Wavelength Vs Wavenumber Wavelength = the distance between two successive crests of troughs in a wave Wavenumber = number of waves (or number of cycles) in a unit distance. IR spectroscopy vs FT IR spectroscopy • The principle in both is same • The prime difference is that in case of IR spectroscopy, the intensity of incident IR radiations is kept constant. • In case of FTIR, the intensity of incident radiations fluctuates to ensure complete detection of the functional groups and individual bonds.