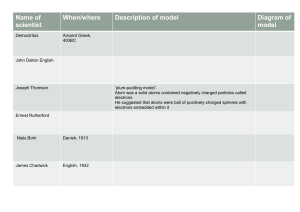
Introductory Chemistry Matter - anything that occupies space and has mass Solid, liquid, gas Solid - rigid, has a fixed volume and shape Liquid - has definite volume, assumes shape of container Gas - has no fixed volume or shape, takes the shape and volume of its container Physical Properties - Characteristics that are directly observable and unique to a substance - Odour, colour, volume, state(s,l,g) Chemical Properties - A substances ability to form new substances - Characteristics determine how the composition of matter changes as a result of contact with other matter - Eg. flammability, rusting, digestion Mixtures - A combination of two or more pure substances - Have variable composition - Eg wood, wine, coffee - Can be separated into pure substances Pure Substances - Fixed composition, cannot be further purified - Always have the same composition - Eg. water, carbon dioxide Physical Change - Change in one or more physical properties of a substance, not in its chemical composition - Eg boiling water (distance between molecules change) Chemical Change - A given substance becomes a new substance/s with different properties and different composition - Eg Bunsen burner - Water decomposes to hydrogen and oxygen H2O= H2+O Element - Substance that cannot be broken down into other substances by chemical methods Compound - Substance composed of a given combination of elements that can be broken down into those elements by chemical methods - A compound always has the same combination of elements - Eg water H2O Homogeneous matter - Uniform composition throughout - Consists of visibly indistinguishable parts - A solution - Does not vary in composition from one region to another - Eg pocari sweat (soft drink), air, brass, table salt stirred into water Heterogeneous matter - Nonuniform composition - Consists of visibly distinguishable parts - Contains regions that have different properties from those of other regions - Eg. oil and water (mixed), oil spill, sand and water Separation of mixtures - Mixtures can be separated based on different physical properties of the components - Eg. boiling(distillation), state of matter(filtration), adherence to a surface(chromatography), volatility(evaporation) Distillation - No chemical change occurs when salt water is distilled Filtration - Separates a liquid from a solid - Uses filter paper, sieve - Can separate sand from water, rocks, anything Chromatography - Uses a column and silica beads, certain parts in a mixture will adhere to the beads more than others causing it to move down the column slower - Separating on the charge of the molecule Sublimation - Sublimation is the process of going from a solid to a gas with no liquid state - Eg ice to steam - Iodine sublimes easily Elements - They consist of identical atoms’ there are 118 elements of which 88 occur in nature - One or two letters - Eg. C for carbon, H for hydrogen - Sometimes the symbol is taken from the elements original latin or Greek name How elements exist in their pure form - Monatomic elements: helium neon, argon (right most column on periodic table) - Diatomic: 2 atoms H2,N2, O2 - Triatomic: O3, Cl2, Daltons Atomic Theory (1808) 1. Elements are made of tiny particles called atoms 2. All atoms of a given element are identical 3. The atoms of a given element are different from those of any other element 4. Atoms of one element can combine with other element t form compounds. A given compound always has the same relative numbers and types of atoms 5. Atomos are indivisible in chemical processes. Atoms are not created or destroyed in chemical reactions. A chemical reaction simply changes the way the atoms are grouped together Antoine Lavoisier - Heated mercury calx (HgO) decomposing it to mercury and a gas. The total mass was the same - Then did the reverse experiment - “nothing is lost, nothing is created, everything is transformed - Chemical transformation - Discovered oxygen Following statements are no longer true All atoms of a given element are identical Atoms are indestructible Compound - Distinct substance that us composed of the atoms of two or more elements and always contains exactly the same relative masses of those elements Chemical formulas - Expresses the types of atoms and the given number of each type in each unit(molecule) of a given compound - DDT: C14H8Cl5 Rules for writing formulas - Each atoms present is represented by its element symbol - The number of each type of atom is indicated by a subscript written to the right of the element symbol - No need for subscript when there is only one of a given atom Electrons - 19th century: knew matter and electric charge were somehow related - 1890’s: determined atoms of elements contain tiny negative particles (SA-particles) - Postulated existence of electrons using cathode ray tubes - Trying to prove electrons had a mass Structure of an Atom - William Thomson (plum pudding model) - Atom is thought of as a uniform pudding of positive charge with enough negative electrons to counterbalance the positive charge - Ernest Rutherford (1911) disproving the plum pudding model Alpha particles: two protons and two neutrons - Shot alpha particles through gold foil - Explained atoms had a nucleus - Atom has a dense center of positive charge - Electrons travel around the nucleus at a large distance A proton has the same magnitude of charge as a electron but its charge is positive MOST nuclei also contain a neutral particle called the neutron Neutron has no charge Valence electrons: electrons on the outside of the shell Mass number: number of protons+neutrons Atomic number: number of protons Question 1. What is the element with 20 neutrons and a mass number of 37? 37-20=17 ,find 17 on period table, Chlorine Isotopes and atomic weight - Atoms of an element which have the same atomic number, but different atomic masses, ie they have a different number of neurons - Most elements found in nature have a mixture of isotopes Electronic Structure of Atoms - Electrons are confined to particular regions in the extranuclear space - Called shells of principle energy levels (1,2,3,4,5 ect) - Electrons have more energy the further away from the nucleus Divided into subshells: s,p,d,f Orbital: region of space that can old 2 electrons Each subshell can be numbered and then they are classified into a specific orbital S refers to spherical orbital P refers to a two lobed orbital D and f orbitals have 5 and 7 different shaped orbitals Orbitals is where you have an electron pair 90-95% of the time Orbitals are potential space for an electron but can be empty Pauli Exclusion Principle: an atomic orbital can hold a maximum of 2 electrons and those 2 electrons must have opposite spins (electrons spin like a top) - Creates a small magnetic field and they become attracted to each other Core electrons: inner most electrons Valencene electrons: electrons in the outermost principle energy level of an atom Ionic Bonds Lewis Dot Structures - Drawing the symbol of an element and representing the outermost electrons of an atom (electrons in its outer shell) is a lewis dot structure - Each dot represents one valence electron - Lewis dot structures can predetermine the way elements react or combine together to form compounds Ionic Bonds - Atoms gain or lose electrons - Ionic compound results when a metal reacts with a non metal - Electrons are transferred - “electrostatic attraction between a positive ion (cation) and a negative ion (anion) - Electron moves from metal to non metal - Each atom has the equal number of protons as there are electrons so the charge is neutral - Applying ionisation energy gives the atoms a positive or negative charge (kJ/mol) Octet rule: atoms gain or lose electrons to achieve an inert gas configuration (only for nobles gasses - Atoms like to have 8 electrons in outer shell to be stable - Group 1 elements: have 1 electron in outer shell and would like to loose this electron( Li, Na, K Rb) - Group 2 elements (Be, Mg, Sr, Ca) have 2 electrons they like to loose Covalent Bonding - Results from the force of attraction between two atoms in the mutual sharing of two electrons - Non polar covalent / polar covalent - Can for between two non-metals OR a non-metal and a metalloid - Eg. when two H atoms come close together the two electrons are simultaneously attracted by both nuclei, producing the bond H. + .H = H:H Electronegativity - The relative ability of a bonded atom to attract shared electrons Dipole Moment - Property of a molecule whose charge distribution can be represented by a center of positive charge and a centre of negative charge Binary compounds: composed of more than one element Elements to the left of the periodic table appear first in compounds Cation followed by anion Binary ionic compounds - Contains positive cations and negative anions - Type 1: metal present forms only one cation - Type 2: metal present can form 2 or more cations with different charges Type 1 - Cation is named first then anion - A cation takes its name from the name of the element - Anion is named by taking the name of the element and adding ‘ide’ Type 2- binary ionic compounds - Charge on the metal ion must be specified - Roman numeral indicates the charge of metal cation - Transition metals - Cation is named first then anion - Because cation can assume more than one charge, the charge is specified by a roman numeral in parentheses Polyatomic ions - Ions that are charge entities composed of several atoms bound together - They have special names - Eg. ammonium, nitrite, sulfate Type 3 - The first element in the formula is named first, and the full element name is used - The second element is named as though it were an anion - Prefixes are used to denote the numbers of atoms present - The prefix mono- is never used for naming the first element - Eg. carbon dioxide Alkanes - Molecules that only contain hydrogen and carbon that are joined by single bonds - Ending in ‘ane’ - Prefix dictates number of carbons in chain Alkanes with substituents - Parent chain: longest carbon chain - Substituents: any branch off the parent chain, named based on alkyl groups 1. Find the longest continuous chain of carbon atoms (parent chain) determines the base alkane name - Drop the ‘ane’ and add ‘yl’ 2. Number the carbons in the parent chain, starting at the right Valence Shell Electron Pair Repulsion Theory - Predicts bond angles - ‘a model that rationalises the shape of molecules and ions resulting from minimising electron pair repulsions around a central atom’ - Valence electrons of an atom may be involved in the formation of a single, double or triple bond, or they may be unshared - Bond angle made by 3 connected nuclei in a molecule - Angle between 0 and 180 Constitutional or structural isomers - Compounds with the same chemical formula but different order of attachment - Isomer: how many variations Organic compounds - Prefix: shows the number of carbons in the parent chain - Infix: shows the nature of the carbon bonding in the parent chain - Suffix: shows the functional group(s) present in the compound Carbonyl Group - C=O - Bonded to rest of molecule Cycloalkenes - Hydrogen atoms can be in 2 positions - Axial: perpendicular to ring - Equatorial: in the plane of ring Alkenes - Most simple alkene is ethene (distilled from crude Cis: on the same side Trans: on opposite sides Nomenclature - Double bond takes priority over all other functions Assigning configuration - Cis or trans? Hydrogenation- chemical reaction - Adds hydrogen on the double bond to turn it back to alkene - Need a catalyst for this reaction eg palladium, nickel Aromatic Compounds - Benzene if the first aromatic compound - - does not react with bromine? Single bond: 1.54A Double bond: 134A Carbon to carbon bond in benzene: 1.40A Benzene had 1.5 bonds Has Kekule structures Carbons can move around the ring system - This is called resonance hybrid Reason why it doesn’t act like a double bond and doesn’t react with bromine Extremely stable ( delocalisation of carbon bonds causes stability) - Donut type orbital Resonance energy: the difference in energy between a resonance hybrid and the most stable hypothetical contributing structure Other Aromatic compounds - Aspirin - Morphine and codeine Alkynes - Contains a carbon to carbon triple bond - Also called acetylenes Amines - Ammonia NH3 - Amines are derivatives of ammonia, one or more hydrogen atoms have been replaced by substituents such as an alkyl or aryl group - Smaller amines have the lower boiling points and larger have higher boiling points Stereoisomers Chirality - Molecule or object that is not superimposable on its mirror image is chiral - H2O, water is achiral (mirror image is the same) Enantiomers - Have the same boiling point, melting point and solubility - React differently towards other chiral molecules - a pair of molecules that exist in two forms that are mirror images of one another but cannot be superimposed one upon the other Stereochemistry - for a molecule to chiral it must have a stereogenic centre - stereogenic centre: a carbon atom bearing four different substituents -