Uploaded by
Vicky Rajkoomar
Grade 8 Natural Science Test: Electromagnetism & Electrolysis
advertisement

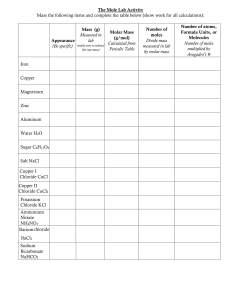
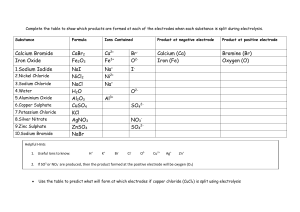
WERDA SCHOOL GRADE 8 NATURAL SCIENCE SEPTEMBER 2022 CONTROL TEST TOTAL: 20 MARKS EXAMINER: MRS. V. RAJKOOMAR TIME: 40 MINUTES MODERATOR: MRS B STOPFORTH This paper consists of 2 pages. INSTRUCTIONS: 1. 2. 3. 4. 5. 6. This paper consists of 2 questions Answer all the questions. Write all the answers in your answer booklet. Number the answers according to the numbering system used in this question paper. Present your answers according to the instructions of each question. Write neatly and legibly. QUESTION ONE. Study the experiment below and answer the questions that follow: 1.1. Name the effect that is produced when an electric current passes through the wire? (1) 1.2. State 2 ways by which the strength of an electromagnet can be increased? (2) 1.3. What is the difference between a magnet and an electromagnet? (2) 1.4. List TWO uses of an electromagnet? (2) [7] QUESTION TWO. Spa and Keagan want to find out if an electrical current can decompose a blue copper chloride solution and light up the bulb. Spa predicts that an electric current will break up the copper chloride into copper and chlorine and the bulb will indeed light up. 2.1. Provide a suitable hypothesis for Keagan. (2) 2.2. Define electrolysis. (1) 2.3. Provide one use of electrolysis. (1) 2.4. What is a Copper (II)Chlorine solution which conducts electricity called? (1) 2.5. What do we call the positive electrode? (1) 2.6. What do we call the negative electrode? (1) 2.7. What happens at the negative electrode during electrolysis and name the substance formed? (2) 2.8. What happens at the positive electrode during electrolysis and name the gas formed? (2) 2.9. How would you increase the reliability of this experiment? (1) 2.10. Provide a suitable conclusion for Keagan. (1) [13] TOTAL 20 MEMO 1.1Name the effect that is produced when an electric current passes through the wire? Electromagnetism (1) 1.2 State 2 ways by which the strength of an electromagnet can be increased? Increase the battery strength and increase the number of turns of the coil. (2) 1.3 What is the difference between a magnet and an electromagnet? (2) A magnet is permanent and a electromagnet is temporary (can be switched off and on) 1.4 List TWO uses of an electromagnet? In a scrap yard to lift scrap metal In medicine to remove fine metal that may be lodged in a patience eye. (Any 2 applicable applications) (2) 2.1. Provide a suitable hypothesis for Keagan. (2) If an electric current is passed through a copper chloride solution it will break it up into Copper and chloride gas and the bulb will light up OR If an electric current is passed through a copper chloride solution it will not break it up and the bulb will not light up. 2. Define electrolysis? (1) The use of electricity to separate chemicals in a solution. 3. Provide one use of electrolysis? (1) Electroplating or any other suitable application 4. What is a Copper (II)Chlorine solution which conducts electricity called? (1) Electrolyte ANODE (1) 6. What do we call the negative electrode? CATHODE (1) 5. What do we call the positive electrode? 7. What happens at the negative electrode during electrolysis and name the substance formed? (2) The positive charged copper ions are deposited on the negatively charged cathode. 8. What happens at the positive electrode during electrolysis and name the gas formed? The negatively charged chlorine ions are attracted and chlorine gas is seen as bubbles (2) 9. How would you increase the reliability of this experiment? (1) Repeat the Experiment or Use another batch of copper chloride solution 10. Provide a suitable conclusion for Keagan. Dependant on learners answer to question 2.1 My hypothesis was correct, when the electric current was passed through a copper chloride solution it did break it up into copper and chloride gas and the bulb did light up OR My hypothesis was incorrect since when the electric current was passed through the copper chloride solution it did break it up into copper and chloride gas and the bulb did light up (1)