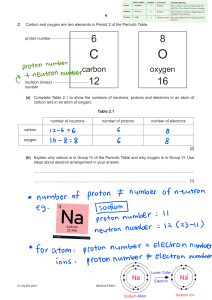
Name ________________________ ● Atoms, Elements, & the Periodic Table BUILD AN ATOM INTRO In this intro activity, you will explore the atom using a computer simulation. Part I - Explore the Atom (6 marks) Open the Build an Atom Simulation - click on the link (https://phet.colorado.edu/sims/html/buildan-atom/latest/build-an-atom_en.html). Click on the Atom option on the first screen Click on the green plus sign next to Net Charge Click on the green plus sign next to Mass Number Atoms are made of three subatomic particles - protons, neutrons, and electrons. The atom has two main parts - the nucleus and the orbits (which are also called the electron clouds) Drag a proton onto the atom. Where does it go? Drag a neutron onto the atom. Where does it go? Drag an electron onto the atom. Where does it go? What subatomic particles (protons, neutrons, and/or electrons) are found in the nucleus of an atom? What subatomic particles (protons, neutrons, and/or electrons) are found in the obits? What element did you make when you added one proton, one neutron, and one electron? Add another proton to the atom that you made. What changes? Add another neutron to the atom that you made. What changes? Add another electron to the atom that you made. What changes? What subatomic particle (proton, neutron, or electron) is responsible for determining the identity of an atom? What subatomic particles (protons, neutrons, and/or electrons) are responsible for determining the mass number of an atom? What subatomic particles (protons, neutrons, and/or electrons) are responsible for determining the net charge of an atom? Part II - Building Atoms of Elements (2.5 marks) Build the following atoms of elements. When each one is complete, record the protons, neutrons, and electrons in it, determine the identity of the element, and use colored pencils or markers to draw it on the model given. Directions Example Add 1 proton Subatomic Particles What Element p=1 Add 1 neutron n=1 Add 1 electron e=1 Hydrogen H Diagram Directions Subatomic Particles Add 5 protons p= Add 6 neutrons n= Add 5 electrons e= Add 3 protons p= Add 4 neutrons n= Add 3 electrons e= Add 10 protons p= Add 10 neutrons n= Add 10 electrons e= Add 9 protons p= Add 10 neutrons n= Add 9 electrons e= Add 6 protons p= Add 6 neutrons n= Add 6 electrons e= What Element Diagram Part III - Identifying Atoms of Elements (1.5 marks) Give the mass number and Identify the elements given their diagrams and subatomic particles. Diagram Subatomic Particles p=4 n=5 e=4 p=7 n=7 e=7 p=8 n=8 e=8 Mass Number What Element Part IV: Isotopes (5 marks) Test your understanding of isotopes by examining the relationships between the pairs of atoms listed below: Atom 1 Atom 2 12 6 13 6 C Carbon-12 Argon-40 11 5 B An atom with 13 protons and 13 neutrons C 12 6 C Argon-41 Boron-10 An atom with 14 protons and 13 neutrons Relationship between atom 1 and atom 2 Isotopes Same Atom, Not Isotopes of Each Other Different Element Isotopes Same Atom, Not Isotopes of Each Other Different Element Isotopes Same Atom, Not Isotopes of Each Other Different Element Isotopes Same Atom, Not Isotopes of Each Other Different Element Isotopes Same Atom, Not Isotopes of Each Other Different Element The periodic table has a great deal of information about every atom. Using your periodic table, answer the following questions: a) What is the atomic number of chlorine (Cl)? _____ b) What is the atomic number of tungsten (W)? _____ c) How many protons are there in any Cl atom? _____ d) How many protons are there in any Te atom? _____ Build any element that you want for this final section of this activity, and fill in the information below: a. b. c. d. e. f. Number of protons _____ Number of electrons _____ Number of neutrons _____ Atomic Mass ______ Atomic Number _______ Charge _______