Uploaded by
Abigail Amor De Guzman
Electron Configuration Worksheet: Chemistry Practice
advertisement

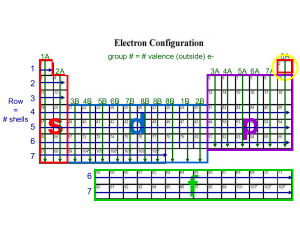
group # = # valence (outside) e- 1A 1 2A 8A 3A 4A 5A 6A 7A 2 Row 3 = 4 # shells 5 6 3B 4B 5B 6B 7B 8B 8B 8B 1B 2B s d p 7 6 7 f Electron Configuration 1 1s group # row # # valence eshell # possibilities are: possibilities are 1-7 s: 1 or 2 7 rows subshell p: 1-6 possibilities are d: 1-10 s, p, d, or f f: 1-14 4 subshells Total e- should equal Atomic # What element has an electron configuration of 1s1? Practice: Ask these questions every time you have to write an electron configuration • Lithium: 1. 2. 3. 4. 5. 6. 7. find the element on the periodic table atomic # = 3 what is the period number? 2 how many shells? 2 what is the group number? 1 how many valence electrons? 1 what subshell(s) does Li have? s what is the electron configuration? 1s2 2s1 Practice: Ask these questions every time you have to write an electron configuration • Boron: 1. 2. 3. 4. 5. 6. 7. find the element on the periodic table atomic # = 5 what is the row #? 2 how many shells? 2 what is the group #? 3 how many valence electrons? 3 what subshell(s) does B have? p what is the electron configuration? 1s2 2s2 2p1 Order of Electron Subshell Filling: It does not go “in order” 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f14 5s2 5p6 5d10 5f14 6s2 6p6 6d10 7s2 7p6 1s2 2s2 2p6 3s2 3p64s2 3d10 4p65s2 4d10 5p6 6s2 4f14 5d10 6p6 7s2 5f14 6d10 7p6 Subshells d and f are “special” group # = # valence e- 1A period # = # e- shells 1 2A 3A 4A 5A 6A 7A 2 3 3B 4B 5B 6B 7B 8B 8B 8B 1B 2B 4 6 3d 4d 5d 7 6d 5 8A d 6 4f 7 5f f Electron Configuration 1 1s group # row # # valence eshell # possibilities are: possibilities are 1-7 s: 1 or 2 7 rows subshell p: 1-6 possibilities are d: 1-10 s, p, d, or f f: 1-14 4 subshells Total e- should equal Atomic # What element has an electron configuration of 1s1? Practice: Ask these questions every time you have to write an electron configuration • Lithium: 1. 2. 3. 4. 5. 6. 7. find the element on the periodic table what is the row #? how many shells? what is the group #? how many valence electrons? what subshell(s) does Li have? what is the electron configuration? Practice: Ask these questions every time you have to write an electron configuration • Boron: 1. 2. 3. 4. 5. 6. 7. find the element on the periodic table what is the row #? how many shells? what is the group #? how many valence electrons? what subshell(s) does B have? what is the electron configuration? Order of Electron Subshell Filling: It does not go “in order” 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 4d10 4f14 5s2 5p6 5d10 5f14 6s2 6p6 6d10 7s2 7p6