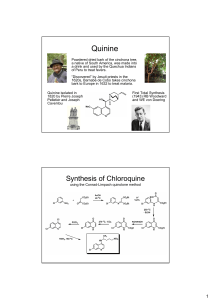
PAPAVERINE Synthesis By MOIN ULLAH PAPAVERINE General Info: A benzylisoquinoline alkaloid found in opium plant Vasodilator help the to dilate blood vessels by relaxing smooth muscle in blood vessels Overdose sympthoms: Weakness, dizziness, vomiting, double vision, sweating, faster heartbeat and uncontrolled eye moment. It has little or no analgesic( relieve pain) or hypnotic( sleep-inducing) properties Drug class: Peripheral Vasodilator Given via injection either through muscle or veins over prolonged duration(1 to 2 min) Sometimes used as an effective treatment for male impotence but this application has been diminished by orally in-takable sildenafil(ViagraR) Side effects: Nausea(1), constipation, diarrhea, headache, spinning sensation, uper stomach pain, loss of appetite and Jaundice(2) 1) sickness feeling or discomfort in the stomach that may come with an urge to vomit 2) skin or eyes yellowing PRODUCTION: Precursor : L-Tyrosine Percent present in opium : 1% Since its presence is very minute an alternate biotechnological process is employed to produce it in large quantities which has been demonstrated by producing substantial quantities of reticuline in a transgenic system, incorporating plant genes from Coptis japonica (Ranunculaceae) into E. coli BIOSYNTHETIC PROCESS (IN OPIUM PLANT) Firstly two L-Tyrosine molecule got converted into L-DOPA and 4-hydroxylphenyl-pyruvic acid in PLP presence L-DOPA yields into Dopamine and 4hydroxylphenyl-pyruvic acid into 4hydroxylphenyl-acetaldehyde via decarboxylation and with enzyme E1’s help Now Dopamine and 4-hydroxylphenylacetaldehyde combines to form (S)norcoclaurine in E2 enzyme presence (S)-norcoclaurine converts into (S)-coclaurine by replacing one of the hydroxyl- group with methaoxy-group with the help of E3 enzyme and SAM as regulator Now (S)-coclaurine converts into N-norreticuline And finally papaverine forms from N-norreticuline in final step via methylation and dehydrogenation DOPAMINE AND 4HYDROXYPHENYL-ACETALDEHYDE FORMATION: PART 1: PART 2: FORMATION OF (S)-COCLAURINE PART 1: PART 2: CONVERSION OF (S)COCLAURINE INTO N-NOR-RETICULINE: SUCCESSIVE O-METHYLATIONS OF N-NOR-RETICULINE : E1: Aromatic amino acid decarboxylase E2: Norcoclaurine synthase E3: Norcoclaurine 6-o-methyltransferase PLP (Pyridoxal phosphate) SAM (S-Adenosyl methionine) involved in methyl group transfers, transsulfuration, and aminopropylation