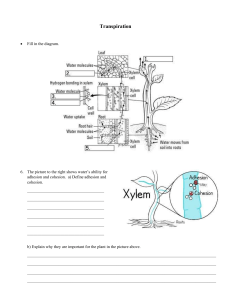
2.2 Water worksheet 1. Which of the following are connected by hydrogen bonds? A. Hydrogen to oxygen within a molecule of water B. Phosphate to sugar in a DNA molecule C. Base to sugar in a DNA molecule D. Hydrogen to oxygen between two different molecules of water (Total 1 mark) 2. What property of water makes it a good evaporative coolant? A. High latent heat of evaporation B. Relatively low boiling point C. Volatility D. Transparency (Total 1 mark) 3. Which statement about water is correct? A. The atoms within a molecule of water are held together by hydrogen bonds. B. Water has a low heat capacity allowing enzymatic reactions to happen at a wide range of temperatures. C. Water molecules are polar, therefore fatty acids do not dissolve. D. Ice has a higher density than liquid water, therefore some organisms can live under the ice. (Total 1 mark) 4. What is a difference between cohesion and adhesion? A. Only adhesion is involved in transpiration pull. B. Only cohesion is involved in the movement of water in soil. C. Only cohesion involves the interaction of water with soil mineral particles. D. Only adhesion involves the interaction of water with xylem. (Total 1 mark) IB Questionbank Biology 1 5. Which diagram best illustrates the interactions between water molecules? (Total 1 mark) B 6. Which diagram represents the polarity of a water molecule? (Total 1 mark) C 7. Blood is a water-based transport medium. Which property of water makes it a good transport medium? A. High specific heat B. Transparency C. Versatility as a solvent D. It has its greatest density at 4°C (Total 1 mark) IB Questionbank Biology 2 8. Blood is a water-based transport medium. Which property of water makes it a good transport medium? A. High specific heat B. Transparency C. Versatility as a solvent D. It has its greatest density at 4°C (Total 1 mark) 9. Explain how the properties of water are significant to living organisms. (9) The attraction of positive and negative molecules allows water to form strong bons with polar molecules allocated around it. This bonding causes water molecules to stick to one another and this process is called cohesion. Cohesion also relates to waters high boiling point and allows animal to regulate body temperature. Cohesion allows organism that are denser than water to float on the surface of water. Adhesion is the interaction that water molecules have with other molecules which explains the reason why water molecules stick to other polar molecules forming hydrogen bonds. Adhesion is vital to allow water to be transported up the stems of plants. Water has high specific heat capacity which is the energy or heat absorbed ability to break down hydrogen bonds in water. This high specific heat capacity makes ecosystems stable by not allowing ponds, lakes and seas to change their temperature rapidly. The cohesive properties allow water to be pulled from the roots to the leaves of plants and allows insects to float on the surface of water to allow them to catch preys. Adhesive properties help water move against gravity from the roots to the leaves supplying the leave with water for its survival. Thermal properties of water allow evaporation of sweat from body surfaces which maintain body temperature. This also makes aquatic ecosystems more stable than terrestrial. The solvent properties cause water to dissolve ions in the soil and bring them across xylem vessels form the roots to the entire plant. The water found in blood plasm dissolve a wide range of solutes and gases which permits blood to transport nutrients and gases around the body. Waters versatility makes water a universal solvent and a vital medium for many biochemical reactions. IB Questionbank Biology 3