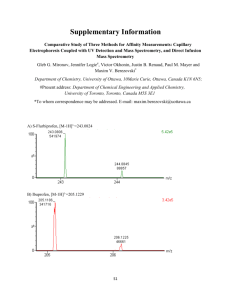
Journal of Photochemistry and Photobiology A: Chemistry 270 (2013) 14–18 Contents lists available at ScienceDirect Journal of Photochemistry and Photobiology A: Chemistry journal homepage: www.elsevier.com/locate/jphotochem Short note Photo-dehydro-Diels–Alder reaction of 1-phenyl-2-(pyridyl)acetylenes in acidic aqueous solution Junpeng Zhuang ∗ , Shuguang Zhang, Haijun Hao, Long Jiang Department of Organic Chemistry, Faculty of Science, Beijing University of Chemical Technology, Beijing 100029, PR China a r t i c l e i n f o Article history: Received 3 May 2013 Received in revised form 29 June 2013 Accepted 8 July 2013 Available online 23 July 2013 Keywords: Photo-dehydro-Diels–Alder reaction Photodimerization Diarylacetylene Naphthalene Head-to-tail a b s t r a c t The photocycloaddition of 1-(4-R-phenyl)-2-(4-pyridyl)acetylenes (R = H, Br, CH3 , Cl) and 1-(4-Rphenyl)-2-(2-pyridyl)acetylenes (R = H, Br, Cl) was carried out in acidic aqueous solution. The unexpected photo-dehydro-Diels–Alder reaction of these monomers was observed, and the results show that two monomers react in a head-to-tail manner and lead to the formation of 2-phenyl-1,3di(pyridyl)naphthalene derivatives. This reaction presents a direct metal-free method to construct the 1,2,3-triaryl substituted naphthalenes from diarylacetylenes. © 2013 Elsevier B.V. All rights reserved. 1. Introduction In recent years, photo-dehydro-Diels–Alder (PDDA) reaction has been reported, which provides a new method to construct a considerable variety of substituted naphthalene derivatives [1,2]. The diene moiety in PDDA reaction contains a C C triple bond and a C C double bond, which differs from the diene moiety in the classical Diels–Alder reaction. Wessig has reported the extensive studies on the intermolecular and intramolecular [4 + 2] PDDA reaction. The synthesis of phenylnaphthalenes [3–5], 1,1 -binaphthyls [6,7], N-heterocyclic analogs of phenylnaphthalenes [8], and highly strained naphthalenophanes [9] are accomplished by the PDDA reaction using 3-arylynones and arylacetylenes in organic solvents upon the irradiation of UV light. Though the PDDA reaction is an efficient route to synthesize naphthalenes, the starting materials always contain arylynones or arylacetylenes and the PDDA reaction of other functional molecules is seldom reported. Herein, the unexpected PDDA reaction of 1phenyl-2-(pyridyl)acetylene is presented. It is well-known that the photodimerization of diarylethenes will form a cyclobutane ring, and among these diarylethenes, stilbenes and styrylpyridine derivatives are the most studied ones [10–20]. In the past few years, we have been studying the photodimerization of some N-heteroarylethenes, such as ∗ Corresponding author. Tel.: +86 10 64412948. E-mail address: zhuangjp@mail.buct.edu.cn (J. Zhuang). 1010-6030/$ – see front matter © 2013 Elsevier B.V. All rights reserved. http://dx.doi.org/10.1016/j.jphotochem.2013.07.004 styrylpyridines, styrylbenzoxazoles and styrylphenyloxazoles in acidic aqueous solution [21–24]. In this paper, we synthesized 1-phenyl-2-(4-pyridyl)acetylene to explore its photochemical properties in 1 M HCl aqueous solution, and to verify whether the heteroarylacetylene has a similar photo property as heteroarylethene. To our surprise, a new photodimer with the structure of 1,2,3-triaryl substituted naphthalene is formed as the main product. A literature survey revealed that the unexpected photoreaction of 1-phenyl-2-(4-pyridyl)acetylene is a PDDA reaction. To the best of our knowledge, this is the first PDDA reaction of N-heteroarylacetylenes in acidic aqueous solution and this reaction is a direct method to construct 1,2,3-triaryl substituted naphthalene structure without metal catalyst or organic solvents (Scheme 1). 2. Results and discussion The synthesis of 1,2,3-triphenylnaphthalene derivatives from diphenylacetylenes by mesoporous graphitic C3 N4 catalyst [25], by RhCl(PPh3 )3 -based catalyst [26] or by other conditions have been reported [27–31]. In contrast to these method, the PDDA reaction of 1-phenyl-2-(pyridyl)acetylene can be carried out in mild conditions without the presence of any metal-catalyst and without inert atmosphere. Compared with the arylynones and arylacetylenes reported by Wessig et al. [1], the diarylacetylene monomers used in this paper is simple and easy to prepare. The PDDA reaction of such monomers extends the scope of PDDA reactions from monoarylacetylenes to diarylacetylenes and from organic solvent to aqueous solution. J. Zhuang et al. / Journal of Photochemistry and Photobiology A: Chemistry 270 (2013) 14–18 15 Scheme 1. The PDDA reaction of 1-phenyl-2-(pyridyl)acetylenes 1a–1g in acidic aqueous solution. The photoreaction of 1-phenyl-2-(4-pyridyl)acetylenes have rarely been studied. Alabugin and coworkers have studied the triplet acetylenes photoreactions as synthetic equivalents of 1,2-bicarbenes. Several diarylacetylenes derivatives react with 1,4-cyclohexadiene to form 1,5-diaryl substituted tetracyclo[3.3.0.02,8 .04,6 ]octanes in acetonitrile upon UV irradiation [32,33]. The 1a–1g monomers were synthesized from styrylpyridines by bromination with Br2 in acetic acid and then debromination with KOH in refluxing ethanol in moderate yield (from 40% to 70%) according to the literature method [34]. And the styrylpyridines were synthesized from 4-methylpyridine or 2-methylpyridine and benzaldehyde derivatives in refluxing acetic anhydride or in DMF in the presence of KO-tBu at 80 ◦ C [35]. When 1-phenyl-2-(4-pyridyl)acetylene 1a was dissolved in 1 M HCl solution and irradiated for several hours, TLC monitoring showed that a new spot was observed on the TLC plate without obvious side reaction. We used 1-(4-bromophenyl)-2-(4pyridyl)acetylene 1b to carry out the same reaction, two hours irradiation led to the formation of a sole product 2b with an isolated yield of 47%. The single crystal structure of 2b shows that a triarylnaphthalene is formed, which means the photo-dehydroDiels–Alder reaction takes place between the two diarylacetylene monomers. We carried out the experiments of 1a again with inner irradiation for about 20 h. 230 mg 2a and 680 mg unreacted 1a were isolated by column chromatography using 1 g 1a dissolved in 400 mL 1 M HCl solution. The yield of 2a based on the converted 1a is 72%, which means this photocycloaddition reaction in acidic aqueous solution is a new type of reaction with good yield and selectivity. The 1 H NMR spectrum and single crystal structure show that 2a has the same structure as 2b. The 4-methyl, 4-chloro substituted derivatives (1c and 1d) of 1a and 1-(4-R-phenyl)-2-(2pyridyl)acetylenes (1e, 1f, 1g; R = H, Br, Cl) were synthesized, the photocycloaddition of these monomers also led to the formation of triarylnaphthalene. In order to carry out the reaction more efficiently, we designed a photo reactor. As shown in Fig. 1, the photo reactor consists of two glass tubes and there is only narrow space between the two tubes. The solution of monomer is filled between the two tubes and is compressed to form a thin film. Each time we use only 2–3 mg monomer dissolved in several milliliter solution. Because the surface area of the solution exposed to UV light is largely increased and the thickness of the solution film is decreased, the conversion of monomer to photodimer is very quick and even finishes in several minutes. The photoreaction was monitored by HPLC using the photo reactor. Fig. 1 shows that 2a is the main product of 1a in acidic aqueous solution with UV irradiation, only trace amount of side product is observed. The chromatograms of 1b, 1c and 1d were also determined (supporting information). The results show that the PDDA reaction of such 1-phenyl-2-(4-pyridyl)acetylene derivatives is very efficient and the conversion of monomers to photodimers is very high in dilute solution in a thin film. Compared with the high yields of photodimerizations of 4-styrylpyridines [19], 2-styrylbenzoxazoles [21] and 5-phenyl2-styryloxazoles [22] in acidic aqueous solution, the isolated yields of 2a–2e using several hundred milligrams to 1 g of 1a–1e dissolved in several hundred milliliters of acidic aqueous solution are only 15–47% (Scheme 1 and supporting information) which indicated that the reactivity of this PDDA reaction in the preparative level is low. With prolonged irradiation several undetermined byproducts were formed gradually and the reaction was then terminated. It was found that 1a–1d containing 4-pyridyl were more active than 1e–1g containing Fig. 1. Photoreactor and the time-resolved HPL chromatogram of 1a irradiated for 0, 5, 10, 15, 25, 45 and 60 min. 16 J. Zhuang et al. / Journal of Photochemistry and Photobiology A: Chemistry 270 (2013) 14–18 Fig. 2. Crystal structure of 2a. 2-pyridyl. The substituents on the phenyl ring also affected the reactivity of the diarylacetylenes. We have synthesized 1-(4trifluoromethylphenyl)-2-(4-pyridyl)acetylene and found it was inert to the UV irradiation. Therefore, the electron-withdrawing groups decreased the reactivity and the electron-donating groups facilitated the reaction. It was found that 1b and 1f containing Br have high photoreactivity than the other monomers which was ascribed to the heavy atom effect of Br. We determined five single crystal structures of 2a, 2b, 2e, 2f and 2g. The structure of 2a as a typical example is shown in Fig. 2. Two pyridyl groups are situated on the 1,3 positions of the naphthalene ring, which indicates that the photocycloaddition of the two monomers take place in a head-to-tail manner just like the photodimerization of styrylpyridines in acidic aqueous solution. The dipole interaction between the protonated pyridyl ring and the phenyl ring facilitate the monomers to approach to each other in a head-to-tail manner. No other triarylnaphthalene isomers with a head-to-head configuration were observed in this reaction because of the repulsion interaction between the protonated pyridyl rings. The phenyl ring and the adjacent C C triple bond in one monomer act as the diene moiety and the C C triple bond in the other monomer act as the dienophile moiety to form the naphthalene ring by the PDDA reaction. The dihedral angles in 2a between the naphthalene ring and 1-(4-pyridyl), 2-phenyl and 3-(4-pyridyl) rings are 67.2◦ , 66.4◦ and 44.8◦ , and the corresponding dihedral angles in 2b are 79.5◦ , 67.7◦ and 42.7◦ , and that in 2e are 64.3◦ , 56.0◦ and 53.8◦ , respectively. As shown in Fig. 3(a) and (b), the maximum absorptions of free 1a–1d in dichloromethane range from 282 to 304 nm due to the –* transition of the conjugated diarylacetylene molecules. The Fig. 3. UV spectra of 1a–1d and 2a–2d. (a) Free 1a–1d in CH2 Cl2 , 5 × 10−5 mol/L; (b) protonated 1a–1d in HCl aqueous solution, 5 × 10−5 mol/L; and (c) free 2a–2d in CH2 Cl2 , 5 × 10−5 mol/L. J. Zhuang et al. / Journal of Photochemistry and Photobiology A: Chemistry 270 (2013) 14–18 17 References Fig. 4. The proposed mechanism of photocycloaddition of 1a in acidic aqueous solution. maximum absorption wavelengths of 1a–1d in acidic aqueous solution were shifted to about 330 nm because of the protonation of the pyridyl ring, which increase the absorption of the monomers in near ultraviolet region. When monomers form the naphthalene photodimer upon irradiation, the conjugated system of the monomer was changed, the maximum absorption wavelength of 2a–2d in CH2 Cl2 shifted to about 255 nm. We have tried to carry out the reaction by heating without UV irradiation. The monomer was unchanged even in the condition of refluxing for several hours. Then, we tried to use a mixed solvent of water and methanol or a pure solvent of methanol instead of the acidic aqueous solution to carry out this reaction upon irradiation, there was also no product formed. These results show that the protonation of the monomer and the UV irradiation are the main factors that lead to the formation of triarylnaphthalene. According to the literatures, the photocycloaddition of 1phenyl-2-(pyridyl)acetylenes in acidic aqueous solution is a typical photo-dehydro-Diels–Alder reaction [1,2]. The proposed mechanism is illustrated in Fig. 4. Two protonated monomers in solution approach to each other in a head-to-tail manner by intermolecular dipole interaction. Upon UV irradiation, a C–C single bond between the two alkyne moieties is formed providing a 1,3-butadiene-1,4diyl biradicals. Then the radical center connected to the pyridinium ring attacks an ortho position of the phenyl ring in the other monomer to give the highly strained cyclic allene. Then the migration of hydrogen in the cyclic allene leads to the formation of naphthalene ring. In summary, the PDDA reaction of 1-phenyl-2-(pyridyl) acetylene derivatives was carried out. This work is a valuable extension of the PDDA reaction to the diarylacetylene derivatives in acidic aqueous solution, which provides a new method for the synthesis of 1,2,3-triarylnaphthalenes. The study of other diarylacetylene derivatives containing N-heteroaryl groups, such as benzoxazole and phenyl-oxazole, is being carried out. Acknowledgments This work was supported by the National Natural Science Foundation of China (20902008). Appendix A. Supplementary data Supplementary data associated with this article can be found, in the online version, at http://dx.doi.org/10.1016/j.jphotochem. 2013.07.004. [1] P. Wessig, A. Matthes, C. Pick, The photo-dehydro-Diels–Alder (PDDA) reaction, Organic & Biomolecular Chemistry 9 (2011) 7599–7605. [2] P. Wessig, G. Müller, The dehydro-Diels–Alder reaction, Chemical Reviews 108 (2008) 2051–2063. [3] P. Wessig, G. Müller, A. Kühn, R. Herre, H. Blumenthal, S. Troelenberg, The photo-dehydro-Diels–Alder reaction: an efficient route to naphthalenes, Synthesis (2005) 1445–1454. [4] P. Wessig, G. Müller, R. Herre, A. Kühn, Synthesis of benzo[g]isochromenes through photo-dehydro-Diels–Alder reaction, Helvetica Chimica Acta 89 (2006) 2694–2719. [5] P. Wessig, G. Müller, C. Pick, A. Matthes, The photo-dehydro-Diels–Alder (PDDA) reaction – a powerful method for the preparation of biaryls, Synthesis 3 (2007) 464–477. [6] P. Wessig, G. Müller, Synthesis of 1 1 -binaphthyls by photo-dehydroDiels–Alder reactions, Chemical Communications (2006) 4524–4526. [7] P. Wessig, G. Müller, Facile photochemical synthesis of 1,1 -binaphthyls, Australian Journal of Chemistry 61 (2008) 569–572. [8] P. Wessig, C. Pick, Photochemical synthesis and properties of axially chiral naphthylpyridines, Journal of Photochemistry and Photobiology A: Chemistry 222 (2011) 263–265. [9] P. Wessig, A. Matthes, Preparation of strained axially chiral (1,5) naphthalenophanes by photo-dehydro-Diels–Alder reaction, Journal of the American Chemical Society 133 (2011) 2642–2650. [10] H. Shechter, W.J. Link, G.V.D. Tiers, Halogenation, dehydrohalogenation and dehalogenation of stilbene photo-dimers (1 2,3,4-tetraphenylcyclobutanes), Journal of the American Chemical Society 85 (1963) 1601–1605. [11] F.H. Quina, D.G. Whitten, Medium effects on photochemical reactions. Photochemistry of surfactant alkyl-4-stilbazole salts in solution, in the solid state, and in monolayer assemblies, Journal of the American Chemical Society 97 (1975) 1602–1603. [12] K. Takagi, B.R. Suddaby, S.L. Vadas, C.A. Backer, Photochemical reactions in organized assemblies. 49. Topological control of reactivity by interfacial orientation: excimer fluorescence and photodimerization of 4-stilbazolium cations in aerosol OT reversed micelles, Journal of the American Chemical Society 108 (1986) 7865–7867. [13] G.W. Coates, A.R. Dunn, L.M. Henling, J.W. Ziller, E.B. Lobkovsky, R.H. Grubbs, Phenyl-perfluorophenyl stacking interactions: topochemical [2 + 2] photodimerization and photopolymerization of olefinic compounds, Journal of the American Chemical Society 120 (1998) 3641–3649. [14] W.Q. Zhang, Z.M. Zhang, Y. Zheng, S.L. Wang, S.N. Zhao, Crystal structure and photochemical properties of r-1, c-2, t-3, t-4-1,3-bis(4-biphenyl)2,4-di(4-pyridyl)cyclobutane, Acta Physico-Chimica Sinica 16 (2000) 207–213. [15] X.H. Li, L.Z. Wu, L.P. Zhang, C.H. Tung, Controlled photocyclization, photodimerization, and photoisomerization of stilbazole salts within nafion membranes, Organic Letters 4 (2002) 1175–1177. [16] G.S. Papaefstathiou, T. Friščić, L.R. MacGillivray, A regiocontrolled ‘head-totail’ [2 + 2] photodimerization of a stilbene involving a ternary solid based on catechol, Journal of Supramolecular Chemistry 2 (2002) 227–231. [17] D.G. Amirsakis, A.M. Elizarov, M.A. Garcia-Garibay, P.T. Glink, J.F. Stoddart, A.J.P. White, D.J. Williams, Diastereospecific photochemical dimerization of a stilbene-containing daisy chain monomer in solution as well as in the solid state, Angewandte Chemie International 42 (2003) 1126–1132. [18] H. Maeda, K. Nishimura, K. Mizuno, M. Yamaji, J. Oshima, S. Tobita, Synthesis and photochemical properties of stilbenophanes tethered by silyl chains. control of (2 + 2) photocycloaddition, cis-trans photoisomerization, and photocyclization, Journal of Organic Chemistry 70 (2005) 9693–9701. [19] S. Yamada, N. Uematsu, K. Yamashita, Role of cation– interactions in the photodimerization of trans-4-styrylpyridines, Journal of the American Chemical Society 129 (2007) 12100–12101. [20] S. Yamada, Y. Tokugawa, Cation− controlled solid-state photodimerization of 4-azachalcones, Journal of the American Chemical Society 131 (2009) 2098–2099. [21] W.Q. Zhang, J.P. Zhuang, C.B. Li, H. Sun, X.N. Yuan, Structure and photochemical properties of r-1,c-2,t-3, t-4-1,3-bis[2-(5-R-benzoxazolyl)]2,4-di(4-R’-phenyl) cyclobutane, Chinese Journal of Chemistry 19 (2001) 695–701. [22] W.Q. Zhang, G. Shen, J.P. Zhuang, P.W. Zheng, X.R. Ran, Synthesis and photolysis of r-1,c-2,t-3,t-4-1,3-di(4-R-phenyl)-2,4-bis[2-(5-phenyloxazolyl)] cyclobutane, Journal of Photochemistry and Photobiology A: Chemistry 147 (2002) 25–30. [23] J.P. Zhuang, W.Q. Zhang, Cross-photodimerization of 4-(2-phenylethenyl) pyridine 2-(2-phenylethenyl)benzoxazole and 5-phenyl-2-(2-phenylethenyl) oxazole, Acta Physico-Chimica Sinica 20 (2004) 173–177. [24] J.P. Zhuang, W.Q. Zhang, Synthesis, structure and photochemical properties of cyclobutanes containing four different aryl groups, Chemical Journal of Chinese Universities 25 (2004) 659–662. [25] F. Goettmann, A. Fischer, M. Antonietti, A. Thomas, Mesoporous graphitic carbon nitride as a versatile, metal-free catalyst for the cyclisation of functional nitriles and alkynes, New Journal of Chemistry 31 (2007) 1455–1460. [26] L.Y. Huang, U.R. Aulwurm, F.W. Heinemann, H. Kisch, Rhodium-catalyzed synthesis of naphthalene derivatives through cyclodimerization of arylalkynes, European Journal of Inorganic Chemistry 1998 (1998) 1951–1957. 18 J. Zhuang et al. / Journal of Photochemistry and Photobiology A: Chemistry 270 (2013) 14–18 [27] K. Ueura, T. Satoh, M. Miura, Rhodium- and iridium-catalyzed oxidative coupling of benzoic acids with alkynes via regioselective C H bond cleavage, Journal of Organic Chemistry 72 (2007) 5362–5367. [28] J.-C. Hsieh, C.-H. Cheng, O-dihaloarenes as aryne precursors for nickel-catalyzed [2 + 2 + 2] cycloaddition with alkynes and nitriles, Chemical Communications (2008) 2992–2994. [29] S.H. Li, J.F. Xiang, X.N. Mei, C.H. Xu, One-pot synthesis of partially fluorinated naphthalene, anthracene, and chrysene derivatives, Tetrahedron Letters 49 (2008) 1690–1693. [30] T. Uto, M. Shimizu, K. Ueura, H. Tsurugi, T. Satoh, M. Miura, Rhodium-catalyzed oxidative coupling of triarylmethanols with internal alkynes via successive C H and C C bond cleavages, Journal of Organic Chemistry 73 (2008) 298–300. [31] K. Sakabe, H. Tsurugi, K. Hirano, T. Satoh, M. Miura, Rhodium/phosphine/amine HBr catalyst system for highly selective cross-cyclodimerization of aryl- and alkylalkynes: efficient access to multisubstituted naphthalene derivatives, Chemistry – A European Journal 16 (2010) 445–449. [32] T.A. Zeidan, R.J. Clark, I. Ghiviriga, S.V. Kovalenko, I.V. Alabugin, Triplet acetylenes as synthetic equivalents of 1,2-bicarbenes. Part II: new supramolecular scaffolds from photochemical cycloaddition of diarylacetylenes to 1,4-cyclohexadienes, Chemistry – A European Journal 11 (2005) 4953–4960. [33] T.A. Zeidan, S.V. Kovalenko, M. Manoharan, R.J. Clark, I. Ghiviriga, I.V. Alabugin, Triplet acetylenes as synthetic equivalents of 1,2-bicarbenes: phantom n,* state controls reactivity in triplet photocycloaddition, Journal of the American Chemical Society 127 (2005) 4270–4285. [34] J.M. Smith, H.W. Stewart, B. Roth, E.H. Northey, ␣-Aminophenacylpyridines and quinolines, Journal of the American Chemical Society 70 (1948) 3997–4000. [35] S.L. Wang, G.Y. Gao, T.I. Ho, L.Y. Yang, Excited-state proton transfer and excitedstate de-hydrogen bonding of the push–pull styryl system, Chemical Physics Letters 415 (2005) 217–222.