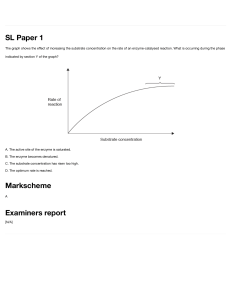
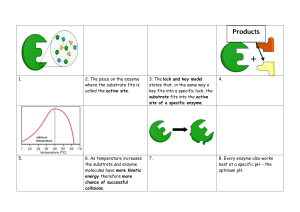
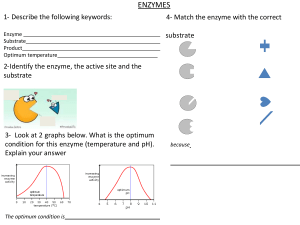
ENZYMES Justine Arla Gustilo, RMT ENZYME: MECHANISM of ACTION Biomedical importance: - presence and maintenance of a complete set of enzyme is essential for breakdown of nutrients to supply energy and chemical building blocks. - deficiency in quantity / catalytic activity of key enzyme can result from genetics, nutritional deficit / toxins Enzymes biological polymers that catalyze chemical reaction are proteins enhance the rate of corresponding non-catalyzedreaction neither consumed nor altered as a consequence of their participation in a reaction. • highly efficient • extremely selective catalyst • • • • • 1. specific for the type of reaction catalyzed • 2. for a single substrate or a small set of closely related substrate. • 3. stereospecific catalyst, catalyzing reaction only of one stereoisomer of a given compound. - binds substrate through at least 3 points of attachment | → chiral product - many contain small non-protein molecules and metal ions that participate directly in substrate binding / catalysis (prosthetic group, cofactors, and coenzyme) Names: -ase dehydrogenase – remove hydrogen atom protease – hydrolyze protein isomerase – catalyze rearrangement in configuration 6 classes of enzyme according to IUB 1. oxidoreductase 2. transferase 3. hydrolases 4. lyases 5. Isomerases 6. ligase Prosthetic groups – tightl and stable incorporation into a protein’s structure by covalent / noncovalent forces ex. Pyridoxal phosphate, FMN, FAD, thiamine pyrophosphate, biotin, metal ions ( Cu, Co, Mg, Mn, Zn, Fe) • Metalloenzyme Function of metal ions bound to enzyme: 1. facilitate binding and orientation of substrate 2. formation of covalent bonds with reaction intermediate (Co2+ in coenzyme B12) 3. interact with substrate to render them more electrophillic or nucleophillic Metal ions in redox reaction are complexed to prosthetic groups ex. Heme, iron-sulfur cluster Cofactors – binds in transient, dissociable manner, either to the enzyme or to the substrate such as ATP - must be present in the medium surrounding the enzyme for catalysis to occur • Metal-activated enzyme Coenzymes – serves as recyclable shuttles (group transfer agents) that transport many substrate from their point of generation to their point of utilization - stabilizes substrate ( H atom / hydride ion) in aqueous environment of the cell. - transport methyl group (folate) acyl group (coenzyme A), oligosaccharides (dolichol) Sources: Vitamin B ¤important component of coenzyme ¤ pantothenic acid ¤ folic acid and cobamide ¤ thiamine pyrophosphate ¤ nicotinamide ¤ riboflavin Active site › environment that is exquisitely tailored for single reaction › takes the form of a cleft or pocket. › 3-dimentional & shield substrate from solvent and facilitates catalysis › binds and orient cofactors or prosthetic groups 4 general mechanism used to enhance catalytic rate : 1. catalysis by proximity - enzyme binded to substrate creates a region of high local substrate concentration - also orient the substrate molecule spatially in a position for them to interact 2. acid-base catalysis act as acid/base ionizable functional group of amino acyl side chain prosthetic group kinds: a). Specific - rate of reaction is sensitive to changes of proton but independent of the concentration of other acid/base present in the solution or at the active site b). General – rate of reaction is responsive to all the acid / base present 3. Catalysis by strain - enzyme binds their substrate in a conformation slightly unfavorable for the bond that will undergo cleavage. 4. Covalent catalysis - involve the formation of a covalent bond between the enzyme and one or more substrate modified enzyme and becomes a reactant - induces a new reaction pathway whose activation energy is lower - chemical modification in enzyme is transient and it returns to its original form after completion of rxn - common among enzyme that catalyze grp transfer rxn - follows ping-pong mechanism Emil Fischer – compared the highly specific fit between the enzyme and its substrate to that of a lock and key. David Koshland induced fit model – when substrate approach and binds to an enzyme they induce a conformational change ( analogous to a hand and a glove) Isozyme › physically distinct version of a given enzyme, each of which catalyze the same reaction. › found in higher animals › arises through gene duplication › exhibit subtle difference in properties, like, sensitivity to particular regulatory factor or substrate affinity that adopt them to certain tissue / circumtances › provide back up copy of an essential enzyme Isozyme of lactate dehydrogenase: L-Lactate dehydrogenase is a tetrameric enzyme with 4 subunits in 2 isoform ( H / M ) isozyme I1 predominantly H – heart I5 predominantly M – liver - rate of catalytic reaction being monitored is proportionate to the amount of enzyme present – can infer enzyme concentration Enzyme detection: 1. Single molecule enzymology – measures rate of single catalytic event and for individual steps in catalysis. - uses fluorescence microscopy 2. High-throughput screening – uses optical detectors which are engineered to permit rapid analysis of multiple samples, thus a). Ideal for survey of numerous product of combinational chemistry b). Simultaneous synthesis of large libraries of chemical compound that contain all possible combination of sets of chemical precursors. - enzyme assay that produce a chromatogenic / fluorescent product 3. enzyme-linked immunoassay (ELISA) – detect protein that lack catalytic activity 4. Spectrophotometric assay -exploit the ability of a substrate / product to absorb light Functional plasma enzyme – perform a physiologic function I n the blood. - lipoprotein, lipase, pseudocholinesterase, proenzyme of blood coagulation and blood clot dissolution - majority are synthesized and secreted by the liver Nonfunctional plasma enzyme – enzyme in the plasma that perform no known physiologic function in the blood Detection of gene mutation using technique that relay on the catalytic efficiency and specificity of enzyme catalyst 1. polymerase chain reaction (PCR) – relies upon the ability of enzyme to serve as catalytic amplifiers to analyze the DNA present in biologic and forensic samples - thermostable DNA polymerase = appropriate oligonucleotide primer → thousands of copies of a sample of DNA to be detected 2. restriction fragment length polymorhism (RFLPs) – facilitates prenatal detection of hereditary disorder e.g., sickle cell trait, -thalassemia, PKU, huntington’s disease - involves cleavage of double stranded DNA by restriction endonucleases, which can detect subtle alteration in DNA that affect their recognized site