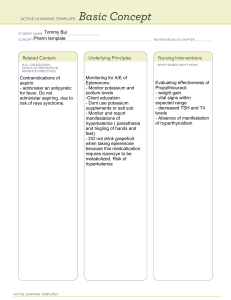
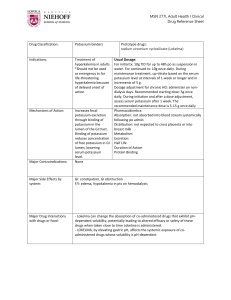
You Try! Potassium persulphate is a chemical used in the process of photo development. By mass, potassium persulphate contains 28.9% potassium, 23.7% sulphur, and the remainder is oxygen. What is the empirical formula? Answer: KSO4 You Try! Find the molecular formula for a compound that has a molar mass of 99g/mole. Its % composition is 24.2% C, 4.0% H, and 71.7% Cl. Answer: C2H4Cl2