Ionic, Covalent, Metallic Properties & Reactivity Experiments
advertisement

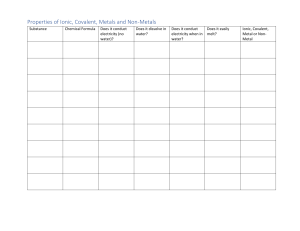
Challenge! Starter Task: Write your answers on the white boards 1. List the properties of ionic compounds 2. Compare the properties of ionic, metallic and covalent substances 3. Predict the electric conductivity of ionic, covalent and metallic substances Learning Objective: Understand how to perform an experiment to analyse the effect of atomic radius on the reactivity of metals Justify your answer to Q3 using your knowledge of Bonding and structure! Think! What is needed for a substance to conduct electricity? Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to determine the order of reactivity Evaluate the method to suggest improvements Properties of Ionic and covalent structures Learning Objective • Understand how to perform an experiment to analyse the Ionic and covalent properties Learning Outcomes • Identify the independent, dependent and controlled variables • Perform an experiment to determine the order of reactivity • Evaluate the method to suggest improvements Platinum:7 Bronze:4 Sssilver:5 Gold: 6 Understand how to perform an experiment to analyse the Ionic and covalent properties SC: Evaluate the SC SC: Write a 1. Identify the Design an method to conclusion independen experimental relating the suggest t, procedure to structures of ionic improvements dependent determine and covalent and control structure of a compounds variables in compound this investigatio n. Task 1: Aim of Experiment You have been given 3 different types of unknown solids. Your task is to design an investigation to determine which solid is a metal, covalent or ionic structure 1. Identify the independent variable in this investigation. 2. Identify the dependent variable in this investigation. Write your answers in your one note page! Learning Objective: Understand how to perform an experiment to analyse the Ionic and covalent properties Think! What properties should you investigate to differentiate between the solids? Challenge Q! Name another type of variable in a scientific investigation and explain its importance. Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to analyse the Ionic and covalent properties determine the nature of compounds Evaluate the method to suggest improvements Task 1: Aim of Experiment You have been given 3 different types of unknown solids. Your task is to design an investigation to determine which solid is a metal, covalent or ionic structure List the properties of ionic compounds ionic substance sheet List the properties of covalent compounds on the ionic substance sheet List the properties of metallic compounds on the ionic substance sheet Explain the difference in electric conductivity of metallic ionic and covalent substances Compare and contrast the solubility of metallic ionic compounds on the ionic substance sheet Learning Objective: Understand how to perform an experiment to analyse the Ionic and covalent properties Team Brainstorm! Evaluate your procedure to identify errors and suggest improvements. Need A Hint? Check the next silde Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to analyse the Ionic and covalent properties determine the nature of compounds Evaluate the method to suggest improvements Task 1: Aim of Experiment You have been given 3 different types of unknown solids. Your task is to design an investigation to determine which solid is a metal, covalent or ionic structure Use these guiding quesstions to write your procedure: Which solids conduct electricity? Solution of which solids conduct electricity? Which solids do not conduct electricity? Challenge: Create a table to help in your investigation Learning Objective: Understand how to perform an experiment to analyse the Ionic and covalent properties Team Brainstorm! Evaluate your procedure to identify errors and suggest improvements. Need A Hint? Check the next silde Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to analyse the Ionic and covalent properties determine the nature of compounds Evaluate the method to suggest improvements Task 2: Design Your Experiment • • • Collect one tray of apparatus per group Collect and put on your protective clothing Select the materials you will use to perform your experiment Learning Objective: Understand how to perform an experiment to analyse the Ionic and covalent properties Think! List any safety precautions that should be taken to perform this experiment. Challenge! Suggest how to make the results of the experiment more reliable. Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to analyse the Ionic and covalent properties determine the nature of compounds Evaluate the method to suggest improvements Team Brainstorm! You have been given 3 different types of unknown solids. Write the results of your experiment in your table below Electric conductivity in solid state Solid A Solid B Solid C Learning Objective: Understand how to perform an experiment to analyse the Ionic and covalent properties Solubility in water Electric conductivity of solution Evaluate this experiment to identify errors and suggest improvements. Need A Hint? How many variables have been changed in this experiment? Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to analyse the Ionic and covalent properties determine the nature of compounds Evaluate the method to suggest improvements Task 3: Perform Your Experiment Think! Describe how to accurately measure volume using a measuring cylinder. • Perform your experiment to determine which solid is covalent ionic or metallic • Put all apparatus back in your tray when you have finished Challenge! • • Fill in the table Write a conclusion explaining the relation between the physical properties and thee structure of the solid Suggest how you could obtain numerical data to analyse in this experiment. Learning Objective: Understand how to perform an experiment to analyse the Ionic and covalent properties Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to analyse the Ionic and covalent properties determine the nature of compounds Evaluate the method to suggest improvements Plenary Covalent? Ionic? or metallic? Solubility in water Solubility in Organic solvent Electric conductivity in solid Electric mpt conducti vity in solution A No No Yes n/a High B No Yes No n/a Low C Yes No No Yes High Learning Objective: Understand how to perform an experiment to analyse the Ionic and covalent properties LO Reflection Have you achieved your learning outcomes for this lesson? Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to analyse the Ionic and covalent properties determine the nature of compounds Evaluate the method to suggest improvements Reactivity of Metals Learning Objective • Understand how to perform an experiment to analyse the reactivity of metals Learning Outcomes • Identify the independent, dependent and controlled variables • Perform an experiment to determine the order of reactivity • Evaluate the method to suggest improvements Extension Task Discuss the reliability and validity of the procedure Learning Objective: Understand how to perform an experiment to analyse the reactivity of metals Learning Outcomes: Identify the independent, dependent and controlled variables Perform an experiment to determine the order of reactivity Evaluate the method to suggest improvements Summary of discovery Properties State at room temperature Electric conductivity Solubitlity in water Ionic compounds Covalent compounds Metalic solid Aim: To compare the reactivity of Group 1 and Group 2 metals 1. List the Group 1 elements tested 2. List the Group 2 elements tested Ionic compounds Covalent compounds Lithium 22 Strontium 20 Calcium 17 Sodium 28 Potassium 33 Magnesium 14 3. Formulate a conclusion that answers the aim of the experiment and is supported by data 4. Explain the conclusion using your knowledge of atomic structure 5. Suggest controlled variables in this experiment