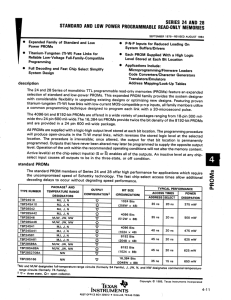
Lecture Notes (AMSE303)
Semiconductor Physics and Devices
Instructor: Prof. Soo Young Kim (Eng. Bldg., Room 608)
Teach Assistant: Mr. Jin Hyuk Cho(Eng. Bldg. 1, Room 638, Tel: 3290-3647)
Department of Materials Science and Engineering
Korea University
Grade: Midterm (40%), Final Exam (40%), Quiz (10%), Attendance (10%)
Semiconductor Physics and Devices
Chapter 1. The Crystal Structure of Solids
Semiconductors: A general introduction
Classification of Materials in terms of electrical resistivity:
• Insulators
1010 − 1018 Ω cm
• Semiconductors 10−4 − 108 Ω cm
• Conductors
10−6 − 10−4 Ω cm
Semiconductor Physics and Devices
Conductivity of semiconductor
The uniqueness of semiconductors is that their conductivity can be
controlled by us over a wide range, e.g. By
• adding minute quantities of impurities (10-7~ 10-1 %)
• applying electric field
• illumination
Current, I
Conducting (on)
Non conducting (off)
Voltage, V
Semiconductor Physics and Devices
Periodic table of the elements
Semiconductor Physics and Devices
Semiconductor Materials
Abbreviated periodic table of the elements
5
6
Be
12
B
13
Mg
30
48
Cd
80
Ge
50
In
81
Hg
15
32
49
Pb
O
16
P
33
S
34
As
51
Sn
82
Tl
N
Si
Al
Ga
8
C
14
31
Zn
7
Se
52
Sb
83
Te
84
Bi
Elements
ESi = 1.1 eV
EGe = 0.67eV
Po
Compounds
EGaAs = 1.43eV
EGaSb = 0.7eV
EGaN = 3.4eV
The most common semiconductor is Silicon
<Classification of Semiconductors>
- By the number of elements
1. Elemental Semiconductor: Group IV elements (C, Si, Ge)
2. Compound Semiconductor: metallurgically intermetallic compound
IV-IV (SiGe, SiC)
III-V (Al-P, As, Sb; Ga-N, P, As, Sb; In-N, P, As, Sb)
II-VI (Zn-S, Se, Te; Cd-S, Se, Te; Hg-Se, Te)
3. Ternary Semiconductor: III-V (GaxIn1-xAs, AlxGa1-xAs)
4. Quaternary Semiconductor: III-V (GaxIn1-xAsyP1-y)
Semiconductor Physics and Devices
Electron Mobility
GaAs: 7500 cm2/ Vs
Si: 1500 cm2/ Vs
Electron Mobility
(cm2/ V.s)
=
drift velocity (cm/ s)
electric field (V/ cm)
Semiconductor Physics and Devices
Direct and Indirect Bandgap of Semiconductors
Direct bandgap: most of compound semiconductor (optical + electrical)
Indirect bandgap: elemental semiconductor (electrical only)
Si
GaAs
Band
structure
Indirect
Direct
Band gap
1.12 eV
1.42 eV
Mobility
1500 cm2/Vs
7500 cm2/Vs
Devices
MOSFET
MESFET, HFET, LD
Applications
Logic Device, High speed, high
Memory, …
frequency devices,
optoelectronic devices
Semiconductor Physics and Devices
Electromagnetic Frequency Spectrum
X- ray: <1 nm, UV: 10~ 390 nm, Visible: 390 ~ 770 nm, IR: 770nm~ 1 mm
Semiconductor Physics and Devices
Bandgap Engineering of Compound Semiconductor
Indirect bandgap
Direct bandgap
ultraviolet
visible
Si
GaAs
InP
infrared
E (eV) = hc/λ = 1.24/ λ (µm)
Semiconductor Physics and Devices
Band gap engineering of AlGaInP/GaAs system
High-brightness system for red, orange, and yellow LEDs
From red to ultra-violet
Energy Gap (eV)
6.0
Direct bandgap
Indirect bandgap
AlN
MgS
5.0
4.0
• Laser diodes
• Light emitting diodes
MgSe
GaN(cubic) ZnS
GaN
3.0
GaAs
Sapphire
3.0
Si
InN 4.0
/high power electric devices
ZnSe
AlAs
GaP
2.0
• Photodetectors
• High temperature
AlP
SiC
1.0
Applications of GaN
5.0
InP
Ge
(MESFET, HFET, HBT)
CdSe
6.0
Lattice Constant (Å)
Visible: 390 ~ 770 nm
(3.18 eV ~1.61 eV)
E = hν = hc/λ = 1.24/ λ (µm)
E = h(c/λ)
Bandgap engineering of AlGaInN system
The GaInN material
system is suited for
UV, violet, blue, cyan
and green high-power
LEDs.
12
Application of Semiconductors
Si semiconductor
o MOSFET (Metal Oxide Semiconductor Field Effect Transistor)
(n-MOSFET, p-MOSFET, C-MOSFET)
- High density DRAM (Dynamic Random Access Memory)
- High density SRAM (Static Random Access Memory)
- High Speed Microprocessor (486, 586, 686, 786?)
- Custom IC (multi-functional switch and IC)
- Power IC
* Computers
o Biploar Transistor
- High Speed IC
- Power IC
o Micro-machine
Compound Semiconductor
o MESFET (Metal Semiconductor Field Effect Transistor)
- High frequency IC (1~10 GHz, Receiver & Transmitter)
- High speed IC with low power consumption (Cray Super Computer)
* Mobile communication
o HEMT (High Electron Mobility Transistor)
- Low-noise amplifier (12~14 GHz, Satellite Communication)
- Milimeter-wave IC (>30 GHz)
- Indoor wireless LAN (30~60 GHz)
o Laser Diodes, LEDs
- Optical Communications
- Displays (Red, Green, Blue)
Semiconductor Physics and Devices
Crystalline solids
The fact that one can alter the properties of semiconductors over a wide range
may have something to do with the atomic arrangement of atoms in these
materials. So, let us look at the crystal structure.
Lattice: Periodic arrangement of atoms. The atomic arrangement determines the macroproperties of the crystal.
Examples:
• Amorphous Si thin film transistors used as switching devices in LCDs
• Polycrystalline Si used as gate in MOSFETs
• Actual active region of MOSFET is fabricated in crystalline Si
Semiconductor Physics and Devices
Crystalline solids
• The Bravais lattices are the distinct lattice types,
which can fill the whole space when repeated
• Crystals could be divided into 14 unit cells,
which meet the following criteria
- The unit cell is (the simplest) repeating unit in
the crystal
- Opposite faces of a unit cell are parallel
- The edge of the unit cell connects equivalent
points
Name
Triclinic
Monoclinic
Orthorhombic
Tetragonal
Cubic
Rhombohedral
Condition
a 1≠a2≠a3, α≠β≠γ
a 1≠a2≠a3, α=β=90o≠γ
a 1≠a2≠a3, α=β=γ=90o
a 1=a2≠a3, α=β=γ=90o
a 1=a2=a3, α=β=γ=90o
a 1=a2=a3, α=β=γ<120o≠90o
#
1
2
4
2
3
1
Hexagonal
a 1=a2=a3, α=β=90o,γ=120o
1
Semiconductor Physics and Devices
Unit cell concept
The unit cell is a small portion of any given crystal that could be
used to reproduce a crystal.
Two different
ways of
representing
a unit cell
Simple 3-dimensional unit cells
simple cubic
body-centerd cubic
face-centerd cubic
Directions and Planes
(hkl): a crystal face or a family of planes throughout a crystal lattice.
[uvw]: a direction in the lattice from the origin to a point.
{hkl}: a set of faces that are equivalent by the symmetry of the
crystal. {100} in the isometric class includes (100), (010), (001), (100), (0-10) and (00-1), while for the triclinic {100} only the (100) is
included.
Miller indices of crystal planes:
Cubic system (Si, GaAs, etc.)
• Three steps that will lead you to its notating by Miller indices
- Identify the intercepts with the axis.
- Calculate the inverse of each of those intercepts.
- Find the smallest integers proportional to the inverse of the intercepts.
Multiplying each fraction with the product of each of the intercepts.
19
Bravais-Miller indices of crystal planes:
Hexagonal (GaN, etc.)
20
Crystal structure of Si and Ge and other common semiconductors
•
•
•
•
2 FCC lattices displaced by ((1/4) a, (1/4) a, (1/4) a) along body diagonal*
Diamond lattice : fcc + (1/4)[111]fcc (=fcc + (1/4) shifted diagonally)
8 atoms per unit cell, atomic packing factor = 34%
Diamond lattice (also called “zincblende” if interpenetrating FCC lattices are made of
different elements like in GaAs)
Each atom is bonded to 4 other atoms (tetrahedral bonding structure with covalent bonding)
The lattice constant or cubic edge is “a”. Generally a is expressed in Angstroms.
1 Å = 10–8 cm = 10–10 m
0
3/4
1/4
0
1/2
1/4
0
Diamond, Si (zincblende, GaAs)
0
1/2
1/2
3/4
1/2
0
Tetragonal Unit
Semiconductor Physics and Devices
Diamond and zincblende lattice unit cells
Diamond structure (Si)
Zincblend structure (GaAs)
Semiconductor Physics and Devices
* Surface Density of atoms in a crystal
1) BCC Structure
2) Diamond Strusture (Si)
a1
For (110) plane with a1 = 5 Å
2 atoms
Surface Density =
=
( a1 )( a1 2 )
= 5.66 x
2
2 (5 × 10 − 8 ) 2
lattice const = 5.43 Å
Surface Density =
4
2 (5.43 ×10 −8 ) 2
= 9.59 × 1014 atoms / cm 2
1014 atoms/cm2
* Density (# of atoms per cm3)
For GaAs,
a1 = 5.66Å
Density of GaAs =
8atoms
= 4.41× 1022 / cm3
−8 3
(5.66 × 10 )
Density of Ga Atoms = Density of As atoms = 2.205 × 10 22 / cm 3
* Doping Concentration
1016
1019/cm3
n-Si :
~
(ND < NC)
+
19
3
n -Si : > 5 x 10 /cm (ND > NC)
Source
n+-Si
Gate
n-Si
SiO2
Drain
n+-Si
MOSFET
Semiconductor Physics and Devices
Example
What is the number of Si atoms in 1 cm3 of Si?
Given is the lattice constant: a = 5.43 Å
8 atoms = 5×1022 atoms
a3
cm3
What is the density of Si?
Atomic weight of Si = 28.1 i.e. 1 mole (NA = 6.023 x 1023 atoms)
of Si has a mass of 28.1 g
gm
5×1022 atoms
×
28.1
3
mole = 2.33 g
cm
Density =
3
23 atoms
cm
6.02 ×10
mole
24
* Atomic Bonding
1) Ionic Bonding : Bonding between negatively charged ion and positively charged ion
Na +
Cl −
Na +
−
+
−
Cl
Na +
Na
Cl −
Cl
Na +
11
Na : 1S 2 2S 2 2 P6 3S1
One excess electron at outer orbit
17
Cl : 1S 2 2S 2 2 P6 3S 2 3P5
Depletion of one electron at outer orbit
2) Covalent Bonding : Bonding by sharing electrons between atoms
Si
H=H
Si
=
Si
Si = Si = Si
As = Ga = As
=
Si
Si
=
Si
As
=
H- - H
Si
As
For 14Si : 1S2 2S2 2P6 3S2 3P2 => SP3 bond => Tetrahedral Structure
Semiconductor Physics and Devices
2) Covalent Bonding : Bonding by sharing electrons between atoms
Si
H=H
Si
=
Si
Si
Si = Si = Si
=
Si
As = Ga = As
=
Si
As
=
H- - H
Si
As
For 14Si : 1S2 2S2 2P6 3S2 3P2 => SP3 bond => Tetrahedral Structure
Si
6
Be
B
13
Mg
30
Zn
48
Cd
Ge
50
In
81
Tl
Pb
16
33
n-type: P doping in Si
S
Si
34
As
51
Sn
82
O
P
Se
Si
52
Sb
83
Te
84
Bi
e
Si = P = Si
Po
p-type: B doping in Si
h
Si = B = Si
=
Hg
15
32
Ga
N
Si
Al
49
80
C
14
31
8
=
12
7
=
5
Si
Semiconductor Physics and Devices
• As temperature increases, thermal vibration may break the covalent bonding
- Generation of free electrons and hole
- Current generation
Moving directions for e- and h+ are different with each of
e- →
← h+
Si
e-
Si = Si = Si
=
Si
h+
3) Metallic bonding
For sodium atoms (1S2 2S2 2P6 3S1)
Eight nearest neighbors with each atom
Share valence electrons
Semiconductor Physics and Devices
* Imperfections and Impurities
1) Thermal vibration
• Atoms form solid (energy lowering)
E
repulsive
r0
a
=> affect to electrical properties, Lattice vibration (phonon scattering)
interactive
2) Vacancy, interstitial, Frenkel defect
<Vacancy>
<interstitial>
Semiconductor Physics and Devices
3) Impurities : foreign atoms and dopant
(B and P in Si, Be and Si in GaAs, Mg and Si in GaN)
Substitutional impurities
Interstitial impurities
Doping : diffusion, ion implantation, epitaxy
<substitutional>
<interstitial>
GaAs
Si
<dislocation>
Semiconductor Physics and Devices
Si-implanted GaAs
E = 120 keV
Dose = 1 x 1013/ cm3
10 nm
Source
+-Si
n+n-GaAs
Gate
n-GaAs
Drain
+-Si
n+n-GaAs
MESFET
Semiconductor Physics and Devices
From red to ultra-violet
Energy Gap (eV)
6.0
Direct bandgap
Indirect bandgap
AlN
MgS
5.0
4.0
• Laser diodes
• Light emitting diodes
MgSe
GaN(cubic) ZnS
GaN
3.0
GaAs
Sapphire
3.0
Si
InN 4.0
/high power electric devices
ZnSe
AlAs
GaP
2.0
• Photodetectors
• High temperature
AlP
SiC
1.0
Applications of GaN
5.0
InP
Ge
(MESFET, HFET, HBT)
CdSe
6.0
Lattice Constant (Å)
Visible: 390 ~ 770 nm
(3.18 eV ~1.61 eV)
E = h(c/λ)
E = hν = hc/λ = 1.24/ λ (µm)
Semiconductor Physics and Devices
Semiconductor Physics and Devices
Semiconductor Physics and Devices
Semiconductor Physics and Devices
Semiconductor Physics and Devices
Semiconductor Physics and Devices
Semiconductor Physics and Devices
Crystal Growth of Si: Czochralski Method
Substrates from ingots
Si (12”), GaAs (6”), InP (3”), GaN (2”4”6”)
Semiconductor Physics and Devices
Crystal Growth: MBE
Semiconductor Physics and Devices
Crystal Growth: Vapor phase epitaxy
Semiconductor Physics and Devices
Crystal Growth: Metal-Organic Chemical Vapor
Deposition (MOCVD)
Semiconductor Physics and Devices
How Si Chips are made?: From Sand to Intel Processors
• Silica
Reduced in presence of C
~ 2000oC
• Very impure Si (99% pure)
Chlorinated
• SiCl4 (liquid)
Distilled, etc.
• Ultrapure SiCl4
Reduced in hydrogen
atmosphere
• Ultrapure poly-Si (99.9999% pure)
From PCPlus.co.uk, http://www.pcplus.co.uk/node/3059
Semiconductor Physics and Devices
How Si Chips are made?: From Sand to Intel Processors
Growing a cylindrical Si crystal
Sorting the good chips from the bad
Slicing the crystal into wafers
Device processing and interconnection
From PCPlus.co.uk, http://www.pcplus.co.uk/node/3059
Semiconductor Physics and Devices
Wafer
Epitaxy + Process
Applications
Light
communication
Optoelectronics
process
Light Disk
Electronics
process
Semiconductor Physics and Devices