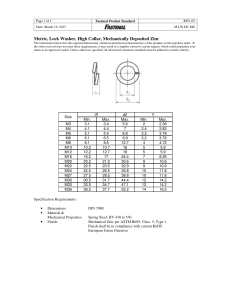
INTRODUCTION Zinc, a chemical element with symbol Zn and atomic number of 30 is a slightly brittle blueslivery metal. It ls approximated to be the 24th abundant element on earth crust. The most commonly available ore used in the extraction of Zinc is called Zinc Blende also known as Zinc Sulphide (ZnS). Historically extraction of zinc has been more difficult than extraction of other metals because at temperatures where metals such as iron typically begins to melt, zinc is a gas. Due to its very low boiling point, at such temperatures, zinc will escape with exhaust gases. Zinc Blende does not contain a very high percentage of zinc and hence it needs to be concentrated. The best concentration method for zinc ore is known as froth flotation. PRINCIPLE OF EXTRACTION The extraction of zinc from zinc blende involves all the communition processes, concentration, roasting, reduction, and purification. The extraction of zinc is done through either pyrometallurgical or hydrometallurgical process(route). PRIMARY PROCEDURES OF ZINC EXTRACTION The Concentration of Ore: Froth Flotation First of all, the ores are crushed into very fine powder-sized particles and are mixed with water. The mixture obtained is called Slurry. A Collector is used which will act as a surfactant and a chemical is added to the slurry. This process is done to enhance the hydrophobic nature of the mineral. The slurry is now being converted into pulp. This pulp is now added to the container which is filled with water and then air jets are forced into it in order to create bubbles. The required mineral is now repelled by the water surface and gets attached to the air bubbles. As these air bubbles rise above the surface with mineral particles sticking to them, these are the froth. This Froth is now collected, separated, and is taken for the next process of refining and extraction. The purpose of concentration is to beneficiate the ore to at least 55% of zinc. Roasting After the concentration of ore, the next step involves the extraction of zinc by Roasting. The concentrated ore is finely divided into small pieces and is then suspended in a rising stream of air. The sulfur content can be reduced hugely with this process. This also converts the Zinc blende to ZnO which is more reactive. 2ZnS + 3O2 → 2ZnO + 2SO2 Reduction: Zinc oxide is mixed with powdered coke and heated to 1673K in a fire clay in which ZnO is reduced to zinc metal. The carbon from coke will act as a reducing agent in this reaction. The reaction involved in the final step of extraction of zinc is: ZnO + C → Zn + CO. Purification: The last step involves the purification of Zinc, Zinc is purified by electrolytic refining. In this process, impure zinc is anode and cathode is of a pure thin sheet of zinc and the electrolyte used is ZnSO4 solution containing a small amount of H2SO4. On passing the electric current pure zinc gets deposited on the cathode side. 1) Pyrometallurgical treatment Production of zinc using blast furnace The roasting of the concentrate convert sulphide Zinc ores to its oxide, by O2 flow & removal of SO2 at T of 700-800oC. This reaction is exothermic, which increases the temp. up to 1000oC. The roasted concentrate is charge into a blast furnace for smelting. (Smelting) Metal production The zinc oxide is then reduced to the metal using either a thermal or an electrolytic process. ZnO is treated in the blast furnace, reduction done with carbon& limestone (CaCo3) at 500oC as a preheating, then O2 supplied from top furnace to produce carbon monoxide (CO) at 1300 oC to produce zinc vapor and lead(pd). Reduction smelting reaction as follows: C + 12O2 → CO C + O2 → CO2 CO2 + C → 2CO ZnO + CO → Zn+CO2 Vapor A flowsheet of pyro metallurgical route Zn vapor is drawn at 1000oC then cooled rapidly to 550 oC in molten lead bath to produce condensate zinc (contain small amount of Cd) which is denser than other component (Zn sink to the bottom), then tapped of, while slug (lighter) tapped from above. Two layers will form as follows: 1. Zinc contains 1wt%Pb drawn to more refining by distillation to produce 99.9% Zn. 2. Pb contains 2.25wt% Zn return to condenser. Then Redistillation at T of 765oC to vaporize Cd off. Zinc produced by this process is about 98.5% pure and can be further refined if required. Around 15% of the world's zinc is produced this way. The blast furnace used in this process is illustrated in figure below. 2)Electrolytic refining (Hydrometallurgy) They consist of 4 steps: leaching, purification, electrolysis, and melting & casting. The crude zinc oxide is dissolved in dilute sulphuric acid (H2SO4) to produce a solution of Zinc sulphate after filtration and purification of the solution. ZnO+H2SO4= ZnSO4 +H2O ZnSO4 electrolysed using a (Pb-1%Ag) anode and an aluminium cathode. O2 is released at anode, while Zinc is deposited on the cathode. H2SO4 is regenerated at the anode and can be used again. The zinc is stripped periodically from the cathode melted and cast into blocks (ingots) for many different purposes. Electrolytically refined zinc is generally 99.995% pure. About 80% of the world's zinc is produced using this process. Flowsheet of Hydrometallurgy of zinc extraction References Lead zinc extraction processes k. k. sahu and a. agrawal,archana india. Solvent extraction in the primaryand secondary processing of zinc by P.M. Cole* and K.C. Sole Van Staden, P.J. Colloquium: Bacterial Oxidation for the Recovery of Metals, South African Institute of Mining and Metallurgy, Johannesburg, 2000. Zinc solvent extraction in the process industries Article in Mineral Processing and Extractive Metallurgy Review · April 2003 RITCEY, G.M. and ASHBROOK, A.W. Solvent Extraction: Principles and Applications to Process Metallurgy, Part II, Elsevier, Amsterdam, 1984.p. 535.