Document
advertisement

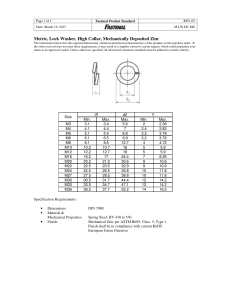
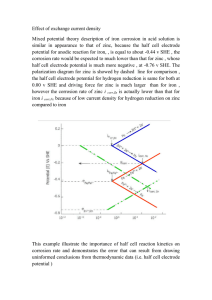
Zinc 鋅 Basic info. ◆Symbol: Zn ◇Atomic no.: 30 ◆Has 5 stable isotopes (64, 66, 67, 68, 70 Zn) ◇24th most abundant element in Earth’s crust ◆Large deposits in Australia, Canada, the US (with largest reserve in Iran) ◇The Earth has been estimated to have 46 years supply of Zn Discovery ◆Who: Andreas Marggraf ◇Nationality: German ◆When to discover: 1764 ◇How: heat a mixture of calamine and carbon in a closed vessel isolate zinc Physical property ◆Lustrous solid at room temp. ◇Hard/Brittle at most temp. ◆Malleable between 100-150oC ◇Density: 7.14g/cm3 ◆Boiling point: 907oC ◇Melting point: 419.53oC ◆Crystal structure: hexagonal ◇Fair conductor of electricity Production ◆~70% Zn comes from mining (most from sphalerite, ZnS deposits) ◇~30% Zn comes from recycling of secondary Zn ◆Production process: 1) Ore grinding 2) Froth flotation 3) Roasting 4) a) pyrometallurgy b) electrowinning 5) Electrolysis of molten ore Application: anti-corrosion Cathodic protection ◆Zn: an efficient sacrificial anode in cathodic protection ◇ Cathode of a car battery is always connected to the car body, supplying electrons to iron body Sacrificial ◆ Galvanization: Zn coating on Fe protection ◇ Zn is more reactive than Fe ◆ Zn releases electron to Fe ◇ Zn is corroded first to save Fe even when the Zn coating is scratched Application: alloy Brass Cadmium zinc telluride ◆Cu + Zn ◇low melting point: 900-940oC ◆more ductile / stronger than Cu alone ◇has superior corrosion resistance ◆Cadmium telluride + zinc telluride ◇Direct bandgap semiconductor Application: use of other compounds Zinc oxide ◆ used as a white pigment in paints ◇ protect its polymers from UV, thus often used in photocopying products ◆ deodorant Zinc chloride Zinc sulphide Zinc lactate ◇ often added to lumber as a fire retardant as a wood preservative ◆ used in luminescent pigments (eg in X-ray / television screens) ◇ used in toothpaste in order to prevent halitosis Biological importance ◆Zn: essential trace element for human ◇Red meat : rich in Zn in general ◆Some plants (eg nuts) contain Zn ◇Highest conc. of Zn in body: in part of the eye / prostate Biological importance ◆Zn: flexible coordinate geometry proteins use Zn to shift conformations perform biological reactions ◇Zn deficiency growth retardation / diarrhea, etc. ◆Zn overdose suppress Cu / Fe absorption ataxia / lethargy The end