•
•
•
Interpretation of Batch reactor Data
Batch Reactor-Homogeneous reactions
Flow Reactor-Heterogeneous reactions
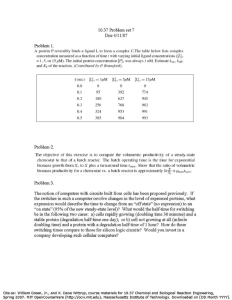
The experimental batch reactor is usually operated isothermally and
at constant volume.
Two Procedures for analyzing the kinetic data- Integral and differential
methods.
In the integral method of analysis
• We guess a particular form of rate equation and,
• After appropriate integration and mathematical manipulation,
• Predict that the plot of a certain concentration function versus time
should yield a straight line.
In the differential method of analysis
• We test the fit of the rate expression to the data directly and without
any integration.
• However, since the rate expression is a differential equation, we
must first find (l/V)(dNldt) from the data before attempting the fitting
procedure.
• CONSTANT-VOLUME BATCH REACTOR
It means the volume of reaction mixture is constant, and not the volume
of reactor.
For ideal gases Ci=p/RT
•
For gas reactions with changing numbers of moles, a simple way of
finding the reaction rate is to follow the change in total pressure of
the system
• The Conversion
And
Integral method of Analysis
Irreversible Unimolecular-Type First-Order Reactions
Suppose we wish to test the first-order rate equation of the following type
•
In terms of conversion
CA/CA0=1-XA
-ln(1-XA)=kt
Limitations
Zero-Order Reactions. A reaction is of zero order when the rate of
reaction is independent of the concentration of materials
APdts
-dCA/dt=k
-dCA=kdt
Separation and Integration
-dCA=kdt
∫dCA=k ∫dt
At
t=0
t=t
Concentration=CA0
Concentration=CA
CA0-CA=kt-----(1)
•
Interms of conversion
CA=CA0(1-XA)
CA=CA0-CA0XA
CA0-CA=CA0XA-----(2)
Compare (2) and (1)
CA0XA=kt
XA=(k/CA0)t
This equation is in
y =m
x
form
•
Test for a zero-order reaction, or rate equation
•
Empirical rate equation of nth order
A Pdts
The rate eqn becomes
-rA = -dCA/dt=kCAn
Separation and Integration
-dCA/dt=kCAn
-dCA/CAn=kdt
-∫dCA/CAn=k∫dt
At
t=0
t=t
Concentration=CA0
Concentration=CA
-∫CA-ndCA=k∫dt
-[CA-n+1/-n+1] CA0CA = kt
1/n-1 [1/Can-1] CA0CA = kt
•
Irreversible Bimolecular-Type Second Order Reaction
•
with corresponding rate equation
•
In terms of conversion
Noting that the amounts of A and B that have reacted at any time t are
equal
Separation and Integration,
•
Use the method of partial fractions
•
Suppose if M=1, complicated. Thus, for the second-order reaction
with equal initial concentrations of A and B, we have
2AProducts
The rate eqn becomes
Separation,
Limits,
At
t=0
t=t
Integration
-dCA/CA2=kdt
Concentration=CA0
Concentration=CA
-∫dCA/CA2=k∫dt
-{-1/CA} = kt
Substitute the limits
(1/CA)-(1/CA0) = kt
Or
(1/CA)=(1/CA0) + kt
This is
Y
=C
In terms of conversion
+mX
Separation
Integration
Use the substitution integration method
form
•
Take
1-xA=y
-dxA=dy
At xA=0 ; y=1
At xA=xA ;y =1-xA
•
Homogeneous catalyzed reactions
A R; reaction rate constant k1
A+C R+C; reaction rate constant k2