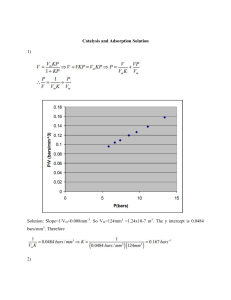
Adsorption, Partitioning and Interfaces Accumulation of Solutes at Interfaces Air KH = PoL/Csatw Kow = Csato/Csatw Koa = Csato/PoL A gas is a gas is a gas T, P Koa KH Po L Octanol Water Fresh, salt, ground, pore T, salinity Csatw Kow Pure Phase (l) or (s) Ideal behavior NOM, biological lipids, other solvents Csato Equilibrium partitioning between water and any organic liquid: Salinity will increase tendency to partition into the organic phase by decreasing the solubility (increasing the activity coefficient) of the solute in water. Assuming that salts are largely insoluble in the organic phase. Partitioning is driven by Van der Waal forces polarity/polarizability H bonding The better the match between chemical properties of solute and solvent, the higher the equilibrium constant for better extraction solvent phase change costs cancel LFERs for relating different organic solvent-water systems IF the two solvents are similar, then simple linear FER can be used for a series of similar compounds: log Ki1w a log Ki 2 w b For example, hexadecane and octanol: log Kihw 1.21 log Kiow 0.43 Works well for apolar and weakly polar solutes. Does not work for very polar compounds, such as phenols fig 7.1 Octanol-water partition coefficient: K ow C o (mol / Lo ) C w (mol / Lw ) Importance Huge database of Kow values is available Method of quantifying the hydrophobic character of a compound. Can be used to estimate aq. solubility Can be used to predict partitioning of a compound into other nonpolar organic phases: other solvents natural organic material (NOM) biota (like fish, cells, lipids, etc.) Why octanol? Has both hydrophobic and small hydrophilic character due to OH bond Therefore a broad range of compounds will have measurable Kow values Methods of measuring Kow Shake flask method (compounds with log Kow < 5) Solute is equilibrated between the two phases by shaking: octanol water Generator column method octanolsaturated water glass beads coated with compound of interest solid adsorbent Chromatographic data Relate Kow of known compounds to capacity factor, k', on a reverse-phase C18 HPLC column Measure k' for known compounds, develop relationship between Kow and k' Adsorption Equilibrium • Adsorption vs. Absorption – Adsorption is accumulation of molecules on a surface (a surface layer of molecules) in contact with an air or water phase – Absorption is dissolution of molecules within a phase, e.g., within an organic phase in contact with an air or water phase Adsorption PHASE I ‘PHASE’ 2 Absorption (“partitioning”) PHASE I PHASE 2 Pgas K H caq Henry’s Law Adsorption terminologies Cu2+ Cu2+ Cu2+ Cu2+ Cu2+ Cu2+ Cu2+ Cu 2+ Cu2+ Adsorbed species present at an overall concentration of cCu(ads) Dissolved adsorbate, at concentration cCu(aq) Cu2+ Cu2+ Cu2+ Adsorbent, in suspension at concentration csolid Adsorbed species, with adsorption density q mg Cu per g solid or per m2 Surface area per gram of solid is the specific surface area mg adsorbed mg adsorbed g solid per ci ,ads qi csolid per L of solution per g adsorbent L of solution Mechanisms of Adsorption • Dislike of Water Phase – ‘Hydrophobicity’ • Attraction to the Sorbent Surface – van der Waals forces: physical attraction – electrostatic forces (surface charge interaction) – chemical forces (e.g., - and hydrogen bonding) Adsorbents in Natural & Engineered Systems Natural Systems Sediments Soils Engineered Systems Activated carbon Metal oxides (iron and aluminum as coagulants) Ion exchange resins Biosolids Engineered Systems • Activated carbon (chemical functional groups) – Adsorption of organics (esp. hydrophobic) – Chemical reduction of oxidants • Metal oxides (surface charge depends on pH) – Adsorption of natural organic matter (NOM) – Adsorption of inorganics (both cations & anions) • Ion exchange resins – Cations and anions – Hardness removal (Ca2+, Mg2+) – Arsenic (various negatively charged species), NO3-, Ba2+ removal Adsorptive Equilibration in a Porous Adsorbent Pore Early Later Laminar Boundary Layer GAC Particle Adsorbed Molecule Diffusing Molecule Equilibrium Adsorption Isotherms Add Same Initial Target Chemical Concentration, Cinit, in each Control Different activated carbon dosage, Csolid, in each mg cinit c fin mg/L q fin csolid g/L g An adsorption ‘isotherm’ is a q vs. c relationship at equilibrium Theory of adsorption Gibb’s free energy at the interface dG (s ) S(s ) dT i(s ) dn i(s ) dA i Integrating at constant temperature, G (s ) i(s ) n i(s ) A i Taking the total derivative, dG (s ) i(s ) dn i(s ) n i(s ) d i(s ) dA Ad i i (s ) (s ) n d i i Ad 0 i d d i( s ) 2 d (2s ) Or For i (2s ) RT ln C(2s ) The Gibb’s adsorption isotherm can be written as C (2l) d d 2 (l) RT dC (2l ) RTd (ln C 2 ) Accumulation of solutes at the interface lowers the interface tension. Adsorption at Solid-Liquid and Solid-Gas Interfaces Langmuir isotherm, gas/solid: rate of evaporation = k1S1 rate of condensation = k2p(S – S1) At steady-state dS1 k 2 p(S S1 ) k 1S1 dt S1 = occupied sites k 2 pS k 2 pS1 k 1S1 0 k2 p S1 k1 bp k2 S 1 bp 1 p k1 Langmuir Isotherm, liquid/solid: backward rate of desorption = k1S1 forward rate of adsorption = k2C(S – S1) dS1 k 2 C(S S1 ) k 1S1 dt At steady-state bC e q 0 1 bC e Q Q 0 bC e q 1 bC e k 2 C e S k 2 C e S1 k 1S1 0 k2 Ce bC e S k1 1 k2 S 1 bC e 1 Ce k1 BET Isotherm – does not assume monolayer coverage V cX Vm (1 X )(1 (c 1) X ) H ads H vap c exp RT Freundlich Isotherm – empirical q e K f C en 1 Commonly Reported Adsorption Isotherms q klin c n Freundlich: q k f c Linear: Langmuir: K Lc q qmax 1 K Lc Steps in Preparation of Activated Carbon • Pyrolysis – heat in absence of oxygen to form graphitic char • Activation – expose to air or steam; partial oxidation forms oxygen-containing surface groups and lots of tiny pores • Starting materials (e.g., coal vs. wood based) and activation • Pores and pore size distributions • Internal surface area • Surface chemistry (esp. polarity) • Apparent density • Particle Size: Granular vs. Powdered (GAC vs. PAC) Characteristics of Some Granular Activated Carbons Characteristics of Activated Carbons (Zimmer, 1988) F 300 H 71 C25 Bituminous Coal Lignite Coconut Shell Bed Density, ρF (kg/m3) 500 380 500 Particle Density, ρP (kg/m3) 868 685 778 Particle Radius (mm) 0.81 0.90 0.79 Surface Area BET (m2/g) 875 670 930 0.33 0.21 0.35 ---- 0.38 0.14 ---- 0.58 0.16 ---- 1.17 0.65 Activated Carbon Raw Material Pore Volume (cm3/g) Micro- ( radius < 1nm) Meso- (1nm < r < 25nm) MacroTotal (radius > 25nm) Oxygen-Containing Surface Groups on Activated Carbon Mattson and Mark, Activated Carbon, Dekker, 1971 Kinetics of Atrazine Sorption onto GAC 167 mg GAC/L 333 mg GAC/L Metal Oxide Surfaces Coagulants form precipitates of Fe(OH)3 and Al(OH)3 which have –OH surface groups that can adsorb humics and many metals Humic substances where R is organic Sorption of NOM on Metal Oxide Example. Adsorption of benzene onto activated carbon has been reported to obey the following Freundlich isotherm equation, where c is in mg/L and q is in mg/g: 0.533 qbenz 50.1 cbenz A solution at 25oC containing 0.50 mg/L benzene is to be treated in a batch process to reduce the concentration to less than 0.01 mg/L. The adsorbent is activated carbon with a specific surface area of 650 m2/g. Compute the required activated carbon dose. Solution. The adsorption density of benzene in equilibrium with ceq of 0.010 mg/L can be determined from the isotherm expression: 0.533 qbenz 50.1 cbenz 4.30 mg/g A mass balance on the contaminant can then be written and solved for the activated carbon dose: ctot ,benz cbenz qbenz cAC 0.50 0.010 4.30 mg/g c AC cAC 0.114 g/L 114 mg/L Example If the same adsorbent dose is used to treat a solution containing 0.500 mg/L toluene, what will the equilibrium concentration and adsorption density be? The adsorption isotherm for toluene is: 0.365 qtol 76.6 ctol Solution. The mass balance on toluene is: ctot ,tol ctol qtol cAC 0.50 ctol 76.6 ctol 0.365 0.114 g/L ctol 3.93x104 mg/L Example The following data were gathered in a laboratory study of the adsorption of trichlorophenol (MW= 197.5 g/mol) on a commercial powdered activated carbon for the following conditions: Initial concentration of solute: 470 mg/L Volume of solvent: 500 mL Carbon Dose (mg) Ce (mg/L) 13.4 4.1 9.3 6.2 5.6 27.9 3.3 91.1 2.0 168.0 Fit these data to the Langmuir and Freundlich isotherms, and determine the constants of both the models. Which model fits better? Example • To a 1 L container holding 0.01mg/L carbontetrachloride, 25 mg/L of activated carbon was added. Determine the equilibrium concentration of tetrachloride in the water if the Fruendlich isotherm parameters for this carbon are given as: Kf = 11 (mg/g) (L/mg) 1/n and 1/n =0.83. Process Design Parameters • Contactors provide large surface area • Types of contactors – Continuous flow, slurry reactors – Batch slurry reactors (infrequently) – Continuous flow, packed bed reactors • Product water concentration may be – Steady state or – Unsteady state Powdered Activated Carbon (PAC) PAC + Coagulants Settled Water Sludge Withdrawal PAC particles may or may not be equilibrated PAC + Coagulants Flocculated Water Process Operates at Steady-State, cout = constant in time Packed Bed Adsorption v, cIN Natural Packed Bed – subsurface with groundwater flow Engineered Packed Bed- granular activated carbon EBCT = empty-bed contact time (Vbed/Q) Adsorptive capacity is finite (fixed amount of adsorbent in bed) v, cOUT Process operates at unsteady state, cOUT must increase over time Linear Equilibrium Partitioning Between Two Phases p pA pb n A RT pA V pA yA mole fraction of component A in the gas phase P p A x A p 0,A cA xA c Henry’s Law: pA=HxA Air–water partitioning coefficient: K AW c air A cA Octanol–water partitioning coefficient: ; K OW oct A c cA Example • A tube containing 50 mL of octanol, 50 mL of water, and 0.5 g of DDT is shaken until the DDT reaches its equilibrium concentration in the octanol and water phases. What is the concentration of DDT (mg/L) in the water phase when the Kow of DDT is 6.19? Partitioning and Separation in Flow Systems (1) 2) dc sys dc (out 2) V2 V1 Qc in( 2 ) Qc (out dt dt (1) 2) dc sys K 12dc (out 2) 2) dc (out dc (out 2) V2 V1K12 Qc in( 2) Qc (out dt dt 2) dc (out Q Q 2) (c in( 2) c (out ) dt V2 V1K12 V2 2) dc (out 1 ( 2) ( 2) (c in c out ) dt tR 2) c (out t 1 exp co tR 1 1 K12 V1 V2 2) (c in( 2) c (out ) V1 R 1 K12 V2