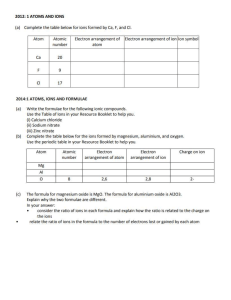
Name ________________________________________________ Date ________________ Period __________ TOTD Balancing Chemical Equations Introduction Part 1 Directions: 1) Put a circle around the product(s), 2) Put a square around the reactant(s), 3) Underline the subscripts, 4) Balance the chemical equations. Make sure there is the same number on both sides of the equation. A. S8 + O2 SO2 B. K10 + Na3 K + Na6 C. N2 + Ca2 N2Ca D. Al + C2 Al2C3 Part 2 Directions: Check each of the chemical equations and identify if the equation is balanced or not balanced. Then circle the color in the correct column and then add that color to all of the matching numbers on the picture on the back. Chemical Equation Balanced Not Balanced 1) 2Fe + 3Cl2 2FeCl3 Dark Blue Dark Purple 2) 4Fe + 3O2 2Fe2O3 Light Blue Light Purple 3) C2H4 + O2 CO2H2O Black Dark Gray 4) CH + CO C2 + HO Green Orange 5) 4H2SiCl2 + 4H2O H8Si4O4 + 8HCl Brown Dark Green Light Orange Yellow Light Green Purple 6) C7H16 + O2 7) 4HSiCl3 + H2O CO2 + H2O H10Si10 O15 + 3HCl