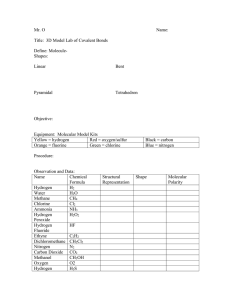
CHEMISTRY FOR ENGINEERS 20-21 QUIZ 2 27/11/2020 Code: 4 1/ Which of the following do you expect to be molecular solids? a) solid silicon tetrachloride, SiCl4 b) lithium bromide, LiBr c) cadmium, Cd d) solid chlorine, Cl2 2/ Tungsten has a body-centered cubic lattice with all atoms at the lattice points. The edge length of the unit cell is 316.5 pm. The atomic mass of tungsten is 183.8 amu. Calculate its density. Giving that NA=6.022x1023 atoms.mol-1 a) 19.00 g/cm3 b) 19.25 g/cm3 c) 19.50 g/cm3 d) 20.00 g/cm3 3/ Which compound contains both ionic and covalent bonds? a) NH4Cl b) H2CO c) MgCl2 d) HCl 4/ Which of the following can form hydrogen bonds with water? a) NiCo b) Ca c) CH3COOH d) CH4 5/ Which compound do you expect to be insoluble in Ethanol (C2H6O)? a) CH3OH b) CBr4 c) H2O d) NH3 6/ What is the interaction between NaCl and I2 ? a) dipole-induced dipole interaction. b) dipole-dipole interaction. c) ion-induced dipole interaction. d) dispersion interaction. 7/ A balloon is filled with 25 liters helium at 25oC. What is the same balloon volume outside at 15oC? a) 24 L b) 15 L c) 20 L d) 26 L 8/ In ethanol, there are covalent bonds, hydrogen bonds and Van der Waals’ forces. Which bonds or forces are broken when ethanol is vaporized? a) covalent bonds, hydrogen bonds and Van der Waals’ forces b) covalent bonds and hydrogen bonds c) covalent bonds and Van der Waals’ forces d) hydrogen bonds and Van der Waals’ forces 9/ Which of the following will conduct electricity? a) HCN b) Sugar solution c) KBr d) Water 10/ Which of the following does not represent a physical change? a) Copper compounds often form beautiful blue crystals when a solution of a given copper compound is evaporated slowly b) When you use the perfume, the liquid of the perfume evaporates quickly from your skin c) Mothballs pass directly into the gaseous state in your closet without first melting d) Oven cleaners contain sodium hydroxide, which converts the oil spatters inside the oven to water-soluble materials, which can be washed away 11/ The Van der Waals’ forces are weaker in PH3 than in NH3 because a) PH3 is polar, while NH3 is non-polar b) The molar mass of PH3 is greater than that of NH3 c) NH3 is hydrogen bonded, while PH3 is not hydrogen bonded d) PH3 is non-polar, while NH3 is polar 12/ The density of an unknown metal is 12.3 g/cm3, and its atomic radius is 0.134 nm. It has a face-centered cubic lattice. This metal is a) Au 101 g/mol b) Pt c) Pd d) Ag 13/ Which kind of reaction is this? NaOH(aq) + HC2H3O2(aq) -> H2O(l ) + NaC2H3O2 (aq) a) Redox reaction b) Acid-base reaction c) Precipitation reaction d) Combustion reaction 14/ What is vapor pressure of the solution at 25oC containing 82.8g Ethanol(s) and 561.6g pure Benzene? Note that the vapor pressure of pure Benzene at 25oC is 639.7 mmHg and the density of Benzene is 0.876 g/ml. a) 511.7 b) 511.6 c) 511.8 d) I don’t know 15/ Which type of solid is dry ice (solid carbon dioxide)? a) ionic b) molecular c) atomic d) none of the above 16/ What is the coordination number of an atom in a face-centered unit cell? a) 4 b) 6 c) 8 d) 12 17/ What is the volume of sulfur trioxide recorded by burning sulfur dioxide with 32g of oxygen gas. Note that the efficiency of the reaction is 70% and 1 mol in ideal condition contains 22.4L of gas. (choose the approximate value) a) 31.1 b) 31.3 c) 31.4 d) 31.2 18/ Methanol can be synthesized through the following reaction: CO(g) + 2H2(g) → CH3OH What is the weight (in grams) of methanol is synthesized by compressing the carbon monoxide gas at 1atm, at temperature 375K, and the volume is 0.068m3 with hydrogen gas? (3 digits) a) 0.0707 b) 70.4 c) 259 d) 80.6 19/ How many grams of fructose in a grape juice bottle. The concentration of fructose is 12% and the volume of the juice contained inside the bottle is 350ml. Note that the density of the solution is 1.04g/ml (choose the approximate value) a) 43.7 b) 43.5 c) 43.6 d) 43.8 20/ Shown below is the cubic unit cell of an ionic compound. Answer the questions by referring to this structure. Be careful to note that some atoms are hidden by those in front. How many red spheres are there in the unit cell? How many green ones? a) 14-8 b) 3-4 c) 2-6 d) 4-8