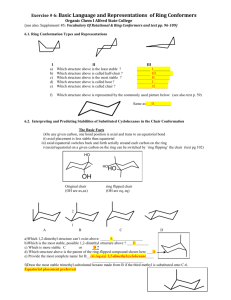
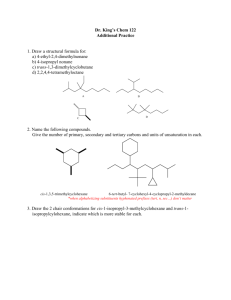
Writing Chairs and Chair Flips Methodology Process: 1. If starting from a name draw planar molecule a. Cis- and trans- show relative stereochemistry i. cis- means same side – both wedge OR both dash ii. trans- means opposite sides – one wedge and one dash b. R and S show absolute stereochemistry and are exactly either a wedge or dash. (8 Series Chap. 4, 118 Series Chap. 5) 2. Convert planar molecule to chair a. Wedged groups are on the top face b. Dashed groups are on the bottom face c. Label atoms clockwise or counterclockwise to mark where groups are. Keep all structures consistently clockwise or counterclockwise to maintain the same stereoisomer. Labeling every structure the same way will help prevent errors later due to habit formation. 3. Write the ring flip conformer 4. Compare axial/equatorial a. Structure with fewer axial groups is lower in energy b. If both structures have the same number of axial groups, those with the larger group equatorial will be lower in energy c. If both are identical = same energy d. We will not deal with cases where one flip has one large group axial and the other has two small groups axial – there is math for that but we’re not going to worry about it. e. H < X < OH < NH2 < CH3 < CH2R or 1o < CHR2 or 2o < CR3 or 3o Example: What is the low energy conformer of trans-1-ethyl-3-fluorocyclohexane? F F Add stereochemistry F 1 2 34 5 6 1 2 3 65 4 Add substituents wedge = up face dash = down face F1 2 3 65 4 Number Carbons Write chair and number F 1 2 3 6541 2 3 6 5 4 ring flip F one axial (F) one equatorial (ethyl) one axial (ethyl) one equatorial (F) Both structures have one axial and one equatorial Ethyl is larger so prefers equatorial Left structure is lower in energy © S. Lievens, November 2018 University of California, Davis For use in UCDavis Chemistry 8 and 118 Series Writing Chairs and Chair Flips Practice Write both chair flips of the given molecule and determine which is the lowest in energy or if both ring flips are the same energy. Br OH F NH2 Br OH F cis-1-bromo-2-methylcyclohexane trans-1-ethyl-3-methylcyclohexane cis-1-tert-butyl-4-isopropylcyclohexane trans-1-sec-butyl-2-ethylcyclohexane cis-1,3-dimethylcyclohexane trans-1-bromo-4-isobutylcyclohexane cis-1,2-diethylcyclohexane A) B) C) D) E) F) G) H) I) J) K) L) M) N) O) P) Q) R) S) T) U) V) © S. Lievens, November 2018 University of California, Davis For use in UCDavis Chemistry 8 and 118 Series Writing Chairs and Chair Flips Practice NH2 trans-1-butyl-3-methylcyclohexane cis-1-butyl-4-ethylcyclohexane trans-1,3-dimethylcyclohexane W) X) Y) Z) AA) AB) AC) AD) AE) AF) AG) Cl HO cis-1-