Atomic Theory Worksheet: History of Atomic Models
advertisement

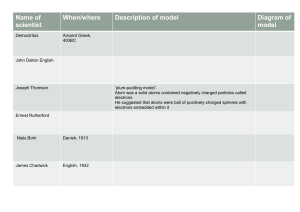
Atomic Theory Name: ___________________________________________ Date: __________ Fill in the blanks as the information is unveiled to you. Democritus All matter is composed of indestructible units, called ________________________. The ________________________ of the atom determined the characteristics. John Dalton All matter is made up of individual particles called atoms that cannot be __________________. Pictured atoms of __________________ __________________. J.J. Thomson __________________ __________________ model. Entire mass is __________________ charged. __________________ charges are evenly distributed throughout. Ernest Rutherford Experiment: Sent __________________ charged “alpha” particles through __________________ foil. Particles all __________________ from a central point. Why? Concluded that the atom’s __________________ charge is all concentrated in the __________________. Neils Bohr Focused on __________________ in __________________ orbits. Identified the idea of __________________ __________________. Evidence for energy levels: Energy __________________ or __________________ as electrons move energy levels __________________ produced. __________________ produced. Erwin Schrodinger Why did he think Bohr was incorrect? Proposed the __________________ __________________ model, which encompasses all of the __________________ locations of electron. Why does the cloud have different areas of density? Orbitals: Recap: 1. 2. 3. Region of space where electrons are most __________________ to be found. All are represented in the __________________ __________________. The __________________ the energy level, the __________________ number of orbitals. Each contains a maximum of __________________ electrons.