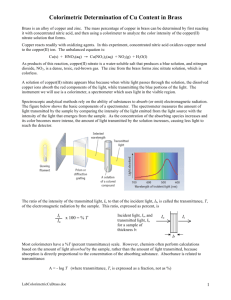
Determination of the Amount of Copper in Brass (Lab #2) Mosier 1 AP Chemistry 10/6/2016 Pre-Lab Questions: Part I: 1. What are the name, formula and molar mass of the colored species being measured? Copper nitrate, Cu(NO3)2, 187.56 g/mol. 2. How do you propose to determine the value of your test tubes? The test tubes will all be 5mL because the concentration of the copper nitrate are all half of each other. In order to get this effect, four of the five test tubes will be filled with 5mL of water. The last test tube which be filled with 10mL of the 4M copper nitrate. 5mL of the 4M copper nitrate will be poured into one of the test tubes with water, thoroughly mixed, then 5mL of this new 2M copper nitrate will be poured into another test tube with water. This process is repeated until there are 5 test tubes with declining concentration. This process will leave 10mL of the 0.025M copper nitrate, but half of it can be disposed, making all the test tubes contain 5mL of copper nitrate. 3. Why is it important to fill the cuvette ¾ full before inserting it into the colorimeter? It is important because having the cuvette filled at ¾ full means that the colorimeter will definitely pass through the sample and give accurate data. At the same time, it is better to not have it completely filled because that would make the cuvette more prone to spilling. 4. Explain how the graph (not shown) is used to determine the wavelength setting. The graph indicates that at the wavelength of 650nm the sample of Cu2+ has the highest absorbance. 5. (a) What is the equation for the calibration plot? A=2.465*c (where A = absorbance and c = concentration) Castillo 1 (b) Determine the concentration for a sample that has an absorbance of 0.669. 0.669 = 2.465 * c c = 0.271M This value was determined by plugging in known variables into the previously determined formula and solving for the unknown variable. Part II: 1. Write the balanced molecular equation for the reaction taking place with regard to the copper in brass in this laboratory exercise. Cu(s) + 4HNO3(aq) → Cu(NO3)2(aq) + 2NO2(g) + 2H2O(l) 2. Write the balanced net ionic for this reaction taking place in the laboratory exercise. 4H+ + 2NO3- → 2NO2 + 2H2O 3. Assuming the shell casing is 100% copper, calculate the minimum volume of 8M nitric acid required to completely dissolve a 3.579g brass shell. 3.579g Cu 1mol Cu 4mol HNO3 1L HNO3 63.546g Cu 1mol Cu 8mol HNO3 = 0.028L or 28mL 4. Calculate the percent copper in brass if the absorbance of their substance is 0.669. .1L Cu(NO3)2 0.271mol Cu(NO3)2 1mol Cu2+ 63.55g Cu2+ 1L Cu(NO3)2 1mol Cu(NO3)2 1mol Cu2+ ( 1.72 3.579 = 1.72g Cu2+ ) ×100 = 48.1% copper in brass Introduction: The purpose of the lab was to find the amount of copper present in a sample of brass. Ultimately, however, the lab is meant to explain how to find the concentration of a species through the application of colorimetry and Beer’s Law. The lab specifically dealt with finding the amount—or percent— copper in a brass shell. The process to find this unknown was multifaceted and left a lot of room for error. The lab can be broken down into two primary purposes: making a standard graph and equation, and applying data collect from the bullet to the graph and equation to find the desired values. In addition, the whole lab deals with a reaction that occurs in an aqueous solution. Many of the concepts that were discussed in Chapter 3, despite being quite irrelevant to the ultimate purpose, made appearance throughout the lab. Castillo 2 Materials: Data collection device Colorimeter Solid Cu(NO3)2 Balance ≈15.8M NHO3, nitric acid 100.0mL volumetric flask 10.0mL volumetric flask 5 test tubes Test tube rack 5 cuvettes Beral pipette Tissues Stir rod Brass sample Procedure: Part I: 1. Goggles and aprons were obtained and put on as per standard safety protocol. 2. Prepared .4M Cu(NO3)2 solution was obtained in a graduated cylinder. 3. Five test tubes are set up on a rack, four of them are filled with 5mL of water. 4. Different concentrations were made by taking 10mL of .4M Cu(NO3)2 and putting it into the test tube without water. Then 5mL was poured from the first test tube to one with water—this test tube now contains .2M Cu(NO3)2. 5mL of this .2M Cu(NO3)2 was put into a different water filled test tube to create a .1M solution. This process is repeated again twice to get a .05M and a .025M solution. 5. The colorimeter was set up, connected to the TI-83—which ran data collecting software—and calibrated. 6. The absorbance of each of the standard (known) solutions were taken by pouring the sample in a cuvette and put in the colorimeter. Between each trial the cuvette was cleaned for potential copper residue. 7. This data was put into a spreadsheet program and graphed a best fit line that represented Beer’s equation. 8. The solutions were safely discarded and the test tubes and graduated cylinder was cleaner. Part II: 1. A brass sample was collected and massed to the nearest 0.001g, then put into a volumetric flask. 2. The minimum amount of 15.8M nitric acid to completely react with the sample if it was entirely made of copper. 3. 20mL of concentrated 15.8M nitric acid was poured into the volumetric flask and left over the weekend to react completely. Then 50mL of deionized water was added with the remaining aqueous solution. The contents were then moved carefully to a 250mL volumetric flask using a pipette. 4. Diluted water was added to the mark on the volumetric flask. The flask was mixed thoroughly, letting the any solids settle. The solution was then drawn out with a clean pipette and placed in a small beaker. 5. The solution was put in the colorimeter and its absorbance was collected and compared to the standards to determine its concentration. Castillo 3 Diagram: [Creating the copper nitrate standards] Data: Concentration (M): 0.025 0.05 0.1 0.2 0.4 Absorbance 0.047 0.116 0.253 0.503 1.031 Absorbance of the Cu(NO3)2 (from the brass sample): 0.453 Castillo 4 Calculation: Amount 15.8M nitric acid required to completely react with the brass sample (if it was made entirely of copper): 4.035 Cu 1mol Cu 63.55g Cu 4mol HNO3 1L HNO3 1mol Cu 15.8mol HNO3 = 0.0161L HNO3 The slope of the standard function is: Absorbance = 2.613 * concentration - 0.015 Using this equation, the concentration of the Cu(NO3)2 can be determined. 0.453 = 2.613 * concentration - 0.015 Concentration = 0.179 Amount of copper in the solution: .250L Cu(NO3)2 0.179mol Cu(NO3)2 1L Cu(NO3)2 1mol Cu2+ 63.55g Cu2+ 1mol Cu(NO3)2 1mol Cu2+ = 2.84g Cu2+ Percent copper in brass: 2.84 ( ) ×100 = 70.5% copper in brass 4.035 Castillo 5 Conclusion: The calculated value of the percent copper in the brass casing sample was found to be 70.5%. This value seems to be well in the region of most cartridge brass—which typically range from 70-80% copper. This relatively accurate percent copper serves to indicate the general accuracy of the colorimetric method. In fact, the method of determining the amount of copper in the brass sample is similar to the procedure used by Crime Scene Investigators to determine the manufacturer of a murderer’s bullet. One of the probable causes of the accuracy in the data was standard graph and equation with had an r2 value of 1, meaning that all the points lie perfectly on a line without much (or any) deviation. As a side note, it was a shame that due to time constraints, the brass sample’s reaction with the nitric acid was done over the weekend. The reaction itself looked interesting on video and would have been nice to see in person. Experimental Sources of Error: As mentioned previously, the fact that the lab was split into two major parts left a lot of room for error. One of the errors that could have affected the results in not completely cleaning the cuvette in between data collection. If the cuvette is not completely cleaned, there would be remaining copper or water, which in turn would affect the absorbance readings. Because there is a higher concentration, the colorimeter would show a high absorbance reading. Since the use of colorimeter was present in both parts of the lab, there are a few possible outcomes of an uncleaned cuvette. One of these outcomes is that the standards get too high of a absorbance which would end up in a calibration graph that has a slope that is too steep. Another outcome would be that the copper nitrate sample gets an inaccurate absorbance reading (either too high or too low) and this would ultimately give an inaccurate calculated concentration. Additionally, one other source of error would be creating the different concentrations of Cu(NO3)2 for the standard. This error could end up making the trend line—which represents Beer’s equation— have an inaccurate equation. Similar to the other source of error, having an inaccurate trend line equation would result in an inaccurate calculated concentration value. More sources of errors will be discussed in the post lab questions. Post-Lab Questions: Part I: 1. Use computer graphing software to plot the data and run a linear regression analysis. Castillo 6 2. What statistical indicator convinces you that your data is trustworthy? The data that was collected the lab was accurate because the plotted points have a coefficient of determination value of 1. The coefficient of determination, or r2, is a statistical measure of the data point’s fit to the regression line. The r2 value for this lab was 1 which means 100% of the points lie on the regression line, indicating precision and likely accuracy. 3. Calculate the concentration of a solution having an absorbance equal to 0.600 and a molar absorptivity 1.5cm-1 M-1 with a path length of 1.0cm. A = abc (where a = molar absorptivity, b = path length, c = concentration) 0.600 = 1.5cm-1 M-1 * 1cm * c Concentration = 0.40M 4. Water is clinging to the bottom and sides of the cuvette. What, if anything should you do prior to measuring the absorbance of your sample? While the fact that water is on the cuvette indicates that the individuals who used it prior attempted to clean it, having anything extraneous in the cuvette while measuring absorbance will alter the measurement. In this case, excess water mixing with copper nitrate will slightly lower the concentration, thereby lowering the absorbance. To combat this, one must ensure that the cuvette is completely cleaned out and dried off before filling it with the desired solution. 5. .What effect would a drop of solution left on the outside of the cuvette have on the measured absorbance? The way that the colorimeter works is that a light is projected through the cuvette and the amount of light that reaches through the cuvette is used to calculate the absorbance. Light is absorbed by solutions which is what allows the colorimeter to determine its absorbance. The excess solution on the outside of the cuvette will absorb some of the light. The sensor on the other end of the light will detect this and will end up producing a absorbance reading that it too high. 6. What effect would a fingerprint left on the outside of the cuvette have on the measured absorbance? Similar to the effect that having a drop of solution on the outside of cuvette, fingerprints will also block out a bit of the light. This will cause the sensor to read that the solution has blocked more light than it really has. The lower light reading means that fingerprints on the cuvette will cause the colorimeter to display a absorbance that is too high. Castillo 7 Part II: 1. A student accidentally spills some of their solution prior to determining its absorbance. What effect does this error have on the calculated percent of copper present in the shell casing? The effect of the spill on the calculated percent of copper present depends on when the spill occurs. If the spill occurs after the adding the water to fill up the 250mL volumetric flask, then the calculations would remain the same (assuming that the student still uses 250mL in his calculations). However, if the spill occurs before filling the 250mL volumetric flask with water, then more water will be added. The additional water will cause the solution to become more dilute and have a lower concentration. The lower concentration will then cause the percent copper to go down. 2. A student wishes to repeat this experiment in order to determine the percent zinc in a brass shell casing. Is this experimental procedure suitable? With a few changes to the procedures, this experiment could be done to determine the mass of the zinc and therefore the percent zinc in the brass sample. First, one must create standards for a zinc solution Then, a brass sample must be dissolved and allow an aqueous solution to be formed from a reaction. Then, just like the lab, a regression analysis will done to the standard curve to create absorbance equation. The concentration of zinc solution from the brass sample can then be determined from the equation. Once a concentration is determine, dimensional analysis can be performed to calculate the mass of the zinc, then this mass can be compared to the total mass of the sample to find the percent zinc in brass shell casing. However, one also just subtract the percent copper from 100 and get a percent zinc (assuming no impurities). Castillo 8