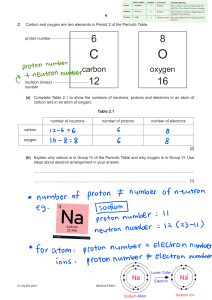
Name: ___________________________________________ Period #: ______ Candy Atoms Lab Do not touch/eat items until instructed to do so… Review: 1) How do you determine the number of protons in an atom? 2) How do you determine the number of electrons in an atom? 3) How do you determine the number of neutrons in an atom? 4) If an atom has 3 protons, what type of atom/element is it? Why? Modeling an Atom: Using the periodic table, draw what a Helium (He) atom would look like below: (Show A=P=E M-A=N work here) □protons □neutrons □electrons Lab Activity: Now choose a different element from the table (1-10): Draw the element box from P. Table My element is ____________________ Atomic number ___________________ Mass number (don’t forget to round!) _____ # of protons ______________ # of neutrons _____________ # of electrons ____________ Electrons configuration: ____ 1st level ____ 2nd level □protons □neutrons □electrons Where are the valence electrons? ________ __________________________________ STOP HERE!! Create your model on your paper plate. LET MS. SMITH CHECK BEFORE CONTINUING! Name: ___________________________________________ Period #: ______ Analysis Questions: 1) How many protons does Oxygen have? How do you know? 2) How many electrons does Boron have? How do you know? 3) How many neutrons does Phosphorous have? How do you know? 4) The atomic number of Argon is 18. How many protons does Argon have? 5) True/False: Aluminum has 16 neutrons. (If false, how many neutrons?) 6) What was one limitation of your candy atom model?