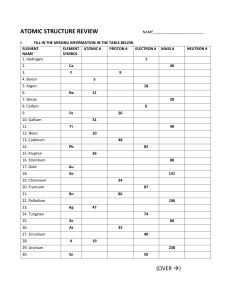
Name ______________________________ Date ______________ Atomic Structure 1. The basic unit of matter is the ___________. 2. Name the three basic particles of the atom, followed by the charge and atomic mass of each particle: a. ___________ ___ ___ b. ___________ ___ ___ c. ___________ ___ ___ 3. If a proton were the size of a basketball, the ______________ would be ___times larger and be some _____ __________ away. 4. The electron orbits at a speed of roughly 1/10th the speed of light, some _____________ miles per second, and is “everywhere at once in its (shape)_______________ orbit, called an electron __________. 5. The atom is roughly __________________________ empty space! 6. The electron takes up about __________ the space of a proton, but the proton weight ________ times more. 7. The smallest and the simplest atom is the element _______________, which is made up of ______________________________________________. 8. The number one rule of atomic structure is that every atom wants to have a _____________ _________ shell. The first shell can hold ___ electrons. 9. The second rule of atomic structure is that atoms want to have an equal number of positive ____________ and ____________ _____________. 10. If hydrogen loses its one electron it becomes ___________________ smaller and is just a(n) ____________ with a positive charge. This is called the hydrogen ______ and is abbreviated _____. 11. If two hydrogen atoms share ____________ this is written as _______ and we call this the ________________. 12. Hydrogen has the atomic number of ____ because it has one _____________. 13. The atom with atomic number of 2 is ____________; it has ____ protons, ___ neutrons and ____ electrons. The symbol for Helium is _____, and the outer shell of electrons is full/not full. 14. What is the atomic mass of helium? ___ 15. The size of a hydrogen atom is about ________________ or .2 __________________.