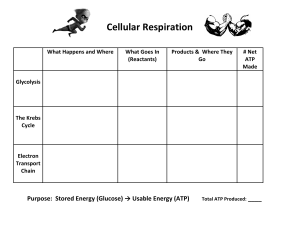
Cellular Respiration Labs Case Study Patient: Jared Sondheim: is a CIA agent that has been poisoned. Symptoms: Muscle weakness, Shortness of breath Dizziness In this case study you will be a Medical Toxicologist: a doctor who treats people that have been poisoned. It is possible that the poison Jared ingested is stopping Jared’s cells from producing ATP ATP is the energy used by the cell to carry out cellular work. Without ATP cell death occurs. Identify what toxin was used to poison Jared in order to cure Jared. Hypothesis Poisons that can stop Cellular Respiration at each step. 1.) Glycolysis can be stopped by 2-Deoxyglucose - Attaches to the first enzyme in glycolysis - Binds to the active site. - Acts as a competitive inhibitor. - Stops glucose being converted to pyruvate, therefore no pyruvate is made. 2.) The Krebs Cycle can be stopped by Arsenic - Attaches to the first enzyme in Krebs Cycle - Binds to the non-active site changing the shape of the enzyme. - Acts as a non-competitive inhibitor. - This means no pyruvate is being used in the Krebs Cycle - No NADH, FADH2, CO2 or ATP is made 3.) The Electron Transport Chain can be stopped by Cyanide - Attaches to the last enzyme in the ETC - No electrons leave ETC, no new electrons can enter ETC without electrons leaving the ETC. No new electrons result in ETC not working and no H+ are moved into the intermembrane space. Binds to the non-active site changing the shape of the enzyme. Acts as a non-competitive inhibitor. 4.) ATP synthase can be stopped by Oligomycin - Attaches to the hole/ pore of ATP synthase - Stops H+ from moving through ATP synthase - No ATP is made We will measure 3 products of cell respiration 1.) Pyruvate – Made by Glycolysis and used in Krebs Cycle 2.) NADH – Made by the Krebs cycle and used by the ETC 3.) Intermembrane H + - The H+ concentration gradient is made by the ETC as electrons move down the chain. The concentration H+ in the intermediate space will increase is the ETC is working. Analysis of Data The Effects of 2-Deoxyglucose on the Products of Cellular Respiration The Effects of Arsenic on the Products of Cellular Respiration The Effects of Cyanide on the Products of Cellular Respiration The Effects of Oligomycin on the Products of Cellular Respiration Data Summary from Experiment Background Information Chemical Energy - ATP Cells in the body need energy to survive. Energy is derived from food. Food has chemical energy which the body uses to produce molecules of ATP. ATP stores energy that will be used in various cellular processes Production of ATP is an example of metabolism Glucose is the molecule used to make ATP → This process is called Cellular Respiration. Mitochondrion Cellular respiration occurs in organelles namely mitochondria Consists of a Double Membrane (two membranes - outer and inner) The space between the membrane is the intermembrane space. We need two Molecules in the mitochondria so that cellular respiration occurs. - Sugar (C6H12O6) from food - Oxygen (O2) from the air The following are the outputs of cellular respiration: - Carbon Dioxide (CO2) - Water (H2O) - Chemical Energy (ATP) Chemical Reaction: C6H12O6 + 6O2 →6CO2 + 6H2O + ATP 4 Stages of Respiration Step 1: Glycolysis Is the breakdown of glucose - one molecule of glucose is split into two pyruvate molecules. This process takes place in the cytosol and requires 10 different enzymes. Glycolysis consists of two phases: - The Energy Investment Phase (Phase 1) - The Energy Payoff Phase (Phase 2) ● During the Energy Investment Phase two ATP molecules are used or invested. This investment of energy allows glucose to split into two Glyceraldehyde-3-Phosphate molecules (G3P) ● In the Energy Payoff Phase the two G3P molecules are converted to two pyruvates. This phase yields a payoff of 4 ATP and 2 NADH This process produces a NET payoff of 2 Pyruvate and 2 NADH Net Generation: 2 ATP AND 2 NADH Pyruvate is transported into the mitochondrion Step 2: Krebs Cycle Krebs cycle is a complex series of enzymes and co-enzymes that break down and, in the process Pyruvate to produce 2 important molecules: - NADH - FADH2 ● NADH AND FADH2 are important molecules because they are able to carry electrons around the cell. They are electron carrier molecules and can both accept and donate electrons ● The Kreb cycle also makes ATP and CO2 (a waste product). Step 3: The Electron Transport chain It is a series of proteins and molecules located in the folded inner membrane of the mitochondria. Protein molecules of the ETC are arranged into 4 groups called complexes. Electrons are supplied to the ETC by the electron carriers: NADH FADH2 NADH donates 2 electrons and at the same time it loses 1 Hydrogen ion (H+) and becomes NAD+. This occurs at Complex I. NADH → NAD+ FADH2 donates 2 electrons (at Complex II) and at the same time It loses 2 Hydrogen ions (H+) and will become FAD Note: Both Complex I and II will pass electrons to Complex III → from which they will be transported to complex IV At Complex IV the electrons leave the chain by combining with O 2 and H+ to H2O (water). Without Oxygen electrons will not be able to leave the ETC. This is why animals require Oxygen to live. As electrons move down the chain, Complexes Absorb energy from electrons. This energy is used to actively pump Protons (H+). As electrons move down the Electron Transport Chain hydrogen ions move into the intermembrane space from the Matrix - Creating a Proton-Gradient. This creates a high concentration of H+ in that space because H+ moves into that intermembrane space from the matrix, this makes the concentration of H + in the matrix low. Step 4: ATP Synthase (Enzyme) The Electron Transport Chain actively moves a lot of H+ ions into the intermembrane space. The matrix has a low concentration of H+ ions. This means that there is a concentration gradient across the membrane. This causes H+ ions to diffuse from the area of high concentration (intermembrane space) to an area of low concentration (the matrix). H + ions will want to move into the matrix. The concentration gradient can also be called the proton gradient. Since H+ ions have an electrical charge, they are not able to pass through the membrane on their own. H+ ions must therefore move into the matrix by passing through an enzyme called ATP Synthase. (Synth – means to make) and (Ase refers to an enzyme) ATP making enzyme As H+ pass through ATP synthase, ATP synthase rotates to produce ATP ATP is made by adding phosphate to ADP. If there is no H+ gradient, no H+ would move through ATP Synthase and therefore no ATP will be produced. Some ATP is made by Glycolysis and the Krebs Cycle but most of the ATP is made up by ATP Synthase. For one molecule of Glucose, Glycolysis and the Krebs Cycle both make 2 ATP molecules of ATP. The ETC and ATP Synthase makes 34 molecules of ATP. 1. Where do the H+ ions come from that make ATP Synthase Work? - ELECTRON TRANSPORT CHAIN 2. What about a summary of what Happens? - ATP synthase uses the proton gradient to make ATP - The Proton gradient is created by the passage of electrons down the Electron Transport Chain. - Electrons combine with O2 and H to leave the chain - H2O is formed in the processes Note: ATP Synthase uses protons gradient to make ATP. Cellular Respiration and Photosynthesis ● Photosynthesis uses light, CO2 and water to produce sugar and oxygen. ● 6CO2 + 6H2O → C6H12O6 + 6O ● Cellular Respiration uses sugar and oxygen to make CO2, H2O and ATP ● C6H12O6 + 6O2 → 6CO2 + 6H2O + ATP Note: The Outputs of photosynthesis are the inputs for Cellular respiration. The Outputs of Cellular Respiration are the inputs of photosynthesis Free Energy and Entropy ● In Photosynthesis the free energy is sunlight is used by plants to make sugar and glucose. ● In Cellular Respiration animals eat plants/ animals to get sugars. Animals have to eat plants/animals because we can’t make our own. Note: Light energy is the original source of the chemical energy in ATP. That is why the sun provides the energy for all life on earth. In photosynthesis, small molecules (Less ordered, high energy entropy state) are made into larger (more ordered, lower energy entropy state) molecules. In cellular respiration the free energy in sugar is released to help make ATP. Note: Moving from a less ordered state to a more ordered state requires work. The work is done using free energy in sunlight. The free energy in sunlight is converted to chemical energy as sugar. Conclusion and Questions Explain how cyanide caused Jared’s symptoms Cyanide, a poison inhibited Jared’s cells from carrying out cellular respiration. This caused a deficiency of ATP which is needed for effective muscle contractions. Hence, a lack of ATP would cause his muscles to feel weak as they cannot contract as effectively. Explain how the data from your experiments showed that cyanide was used to poison Jared and that 2-Deoxyglucose, arsenic and oligomycin were not used Cyanide inhibits the ETC which causes a deficiency of Hydronium ions in the intermembrane space. Since electrons cannot transfer at complex IV, new electrons will not be able to enter the cycle hence causing a build-up of electron carrier NADH. For each molecule of glucose, glycolysis only makes 2 ATP, the Krebs Cycle only makes 2 ATP and the ETC makes 34 ATP. If the suspects had given Jared the 2-deoxyglucose poison, would the decrease in ATP concentration be smaller, the same or smaller, the same or larger as when they used cyanide? Explain your answer The decrease in ATP would be significantly larger. 2-deoxyglucose would inhibit glycolysis which would, in turn, result in a pyruvate deficiency (also no ATP would be produced in the first step). With a pyruvate deficiency, the Krebs Cycle would not produce enough NADH and ATP thus causing even less ATP and so on. Describe how the antidote helped Jared’s cells make ATP (include the ETC and H+ gradient in your answer) The antidote would cause cyanide to release from the binding site as well as prevent it from binding to other complex IV proteins, thus restoring function to the ETC allowing electrons to transfer while providing energy to actively pump H+ ions into the intermembrane space, therefore, restoring the H+ ions gradient. Explain how the data supports your hypothesis. If needed, you can change your poison selection. High levels of NADH indicates that it is building up and not being consumed. This is due to electrons are building up in the ETC and not leaving the ETC for new electrons to transfer. If Electron Transfer is inhibited then H+ ions will not move into the Intermembrane space causing a deficiency in H+ ions. This is also apparent in the data present.