Aromatic Heterocyclic
Chemistry
D a v i d T . Babies
SmithKline Beecham Pharmaceuticals, Harlow, Essex
Series sponsor:
SEIMECA
Z E N E C A is a major international company active in four main areas of business:
Pharmaceuticals, Agrochemicals and Seeds, Specialty Chemicals, and Biological Products.
ZENECA's skill and innovative ideas in organic chemistry and bioscience create products
and services which improve the world's health, nutrition, environment, and quality of life.
ZENECA is committed to the support of education in chemistry.
OXFORD
NEW YORK
TOKYO
O X F O R D U N I V E R S I T Y PRESS
Oxford
University
Press,
Walton Street,
Oxford
Athens
New
Auckland
Calcutta
Istanbul
Kuala Lumpur
Madras
Madrid
Mexico
Nairobi
Paris
City
Taipei
Tokyo
and associated
is a trademark
Published
by Oxford
form
T. Davies,
fair
review,
Press.
No part
dealing for the purpose
licences
outside
may
system,
of research
study,
Oxford
Agency.
University
This book is sold subject
to the condition
the publisher's
prior
including
on the subsequent
A catalogue
of Congress
Aromatic
heterocyclic
QD400.D38
1991
David
Printed
I.
otherwise
of
binding
a
similar
imposed
the British
Library
Data
T.
8
91-34831
(Pbk)
in Great Britain
Avon
Davies.
Title.
547'.S9-dc20
The Bath Press,
not,
T.
chemistry.
ISBN 0 19 855660
above.
purchaser.
chemistry/David
I. Heterocyclic
be sent to
and without
from
by
or
of
concerning
should
in any form
Cataloging-in-Publication
Davies,
or
1988,
with the terms
that it shall
being
record for this book is available
Library
Act,
hired out, or
consent
this condition
of any
or criticism
at the address
or cover other than that in which it is published
condition
Oxford
Enquiries
Press,
be lent, re-sold,
of
in respect
and Patents
in accordance
Licensing
by way of trade or otherwise,
without
are allowed
Designs
in any
in writing
or private
reproduction
be
or transmitted,
those terms and in other countries
the Rights Department,
circulated
of this publication
the prior permission
issued by the Copyright
reproduction
1992
1995
under the Copyright,
in the case of reprographic
York
1992
Within the UK, exceptions
as permitted
New
1994,
stored in a retrieval
without
Press
States
Press Inc.,
1993 (with corrections),
or by any means,
University
University
in the United
University
All rights reserved.
reproduced,
in
Ibadan
First published
Reprinted
Melbourne
Singapore
companies
of Oxford
© David
Delhi
Karachi
Toronto
Berlin
Oxford
6DP
Bombay
Dar es Salaam
Hong Kong
0X2
York
Bangkok
Cape Town
Florence
Oxford
Series Editor's F o r e w o r d
A r o m a t i c h e t e r o c y c l i c c h e m i s t r y is an e n o r m o u s and c o m p l e x s u b j e c t of great
i n d u s t r i a l and a c a d e m i c s i g n i f i c a n c e . A n u m b e r of t h e m o l e c u l e s of l i f e a r e d e r i v e d
f r o m aromatic heterocycles and many important pharmaceutical and agrochemical
c o m p o u n d s are b a s e d o n a r o m a t i c h e t e r o c y c l e s . C o n s e q u e n t l y , the i m p o r t a n c e of
a r o m a t i c h e t e r o c y c l i c c h e m i s t r y h a s s t i m u l a t e d a vast a m o u n t of s y n t h e t i c a n d
theoretical w o r k in t h e area.
O x f o r d C h e m i s t r y P r i m e r s h a v e b e e n d e s i g n e d to p r o v i d e c o n c i s e i n t r o d u c t i o n s
r e l e v a n t to all s t u d e n t s of c h e m i s t r y , a n d c o n t a i n o n l y the essential m a t e r i a l that
w o u l d b e c o v e r e d in an 8 - 1 0 l e c t u r e c o u r s e . In this p r i m e r D a v i d D a v i e s h a s
p r o d u c e d an e x c e l l e n t i n t r o d u c t i o n to a r o m a t i c h e t e r o c y c l i c c h e m i s t r y that s h o u l d
s t i m u l a t e a n y r e a d e r to e x p l o r e f u r t h e r into this v a s t topic. T h i s p r i m e r will b e of
interest t o a p p r e n t i c e a n d m a s t e r c h e m i s t alike.
The Dyson
Perrins
Laboratory,
Stephen G. Davies
University of Oxford
Preface
H e t e r o c y c l i c c h e m i s t r y is a vast discipline a n d at first sight i m p o s s i b l e to d o j u s t i c e
to in a text of this size. T h e a i m of this b o o k is to p r e s e n t o n l y t h e essential f e a t u r e s
of t h e m o r e i m p o r t a n t r i n g s y s t e m s . M a n y reaction m e c h a n i s m s are d i s c u s s e d in
detail and several c o m p l e t e s y n t h e s e s of h e t e r o c y c l e s a r e p r e s e n t e d . I h o p e that t h e
r e a d e r will f i n d this t e x t b o t h interesting a n d instructive, a n d t h a t it will p r o v i d e t h e
p l a t f o r m f o r f u r t h e r s t u d y of this f a s c i n a t i n g s u b j e c t .
I w o u l d like t o t h a n k m y f r i e n d s a n d c o l l e a g u e s at S m i t h K l i n e B e e c h a m
P h a r m a c e u t i c a l s f o r t h e i r h e l p f u l c o m m e n t s , including A n g e l a G a d r e , C l a r e
Hayward, Chris Johnson, H e l e n Morgan and vacation students Peter Ainsworth
a n d F r a n c i s M o n t g o m e r y . I a m similarly g r a t e f u l t o Prit S h a h a n d c o l l e a g u e s of
G l a x o G r o u p R e s e a r c h . I a m i n d e b t e d t o R o g e r M a r t i n of S m i t h K l i n e B e e c h a m
P h a r m a c e u t i c a l s f o r h e l p i n g m e with t h e c h e m i c a l s t r u c t u r e d r a w i n g p a c k a g e .
P r o f e s s o r G u r n o s J o n e s m a d e s e v e r a l h e l p f u l c o m m e n t s . Finally, I w o u l d like t o
t h a n k t h e Series E d i t o r , S t e v e D a v i e s , f o r his advice a n d e n c o u r a g e m e n t , as
well as his a c c e p t a n c e of an i n d u s t r i a l scientist t o w r i t e an a c a d e m i c t e x t .
Harlow
J u n e 1991
D.T.D.
To Julie
Contents
1
Introduction
1
2
Pyrroles, thiophenes, and furans
10
3
Oxazoles, imidazoles, and thiazoles
20
4
Isoxazoles, pyrazoles, and isothiazoles
28
5
Pyridines
35
6
Quinolines and isoquinolines
46
7
Indoles
53
8
F i v e - m e m b e r e d ring heterocycles with three or four heteroatoms
61
9
Six-membered ring heterocycles containing one oxygen atom
67
10
Pyrimidines
73
1S
A n s w e r s to problems
78
Index
87
1. Introduction
1.1 Heterocyclic chemistry
H e t e r o c y c l i c c h e m i s t r y is a large and i m p o r t a n t b r a n c h of organic chemistry.
H e t e r o c y c l e s o c c u r in nature, f o r i n s t a n c e in n u c l e i c acids (see C h a p t e r 10)
and indole alkaloids (see C h a p t e r 7). Synthetic h e t e r o c y c l e s h a v e w i d e s p r e a d
uses as herbicides (e.g. 1.1), f u n g i c i d e s (e.g. 1.2), insecticides (e.g. 1.3), d y e s
(e.g. 1 . 4 ) , o r g a n i c c o n d u c t o r s (e.g. 1 . 5 ) , a n d , of c o u r s e , p h a r m a c e u t i c a l
products such as the anti-ulcer d r u g 1.6.
N-N
C1
A
r
HA-
O
*
EtN^N
„
NEt
/
7
X
/ ^ C l
fT t
O
1/ \
1
s
P h ^ o '
/ ^ Y ^ O
11
n - S ^ W * *
U
^ ^
1.2
1.3
1.2 Aromaticity and heteroaromaticity
A n y r i n g s y s t e m c o n t a i n i n g at least o n e h e t e r o a t o m (i.e. an a t o m other t h a n
c a r b o n - t y p i c a l l y n i t r o g e n , o x y g e n , o r s u l p h u r ) c a n b e d e s c r i b e d as
heterocyclic. This broad definition encompasses both aromatic heterocycles
(such as pyridine 5.1) and their n o n - a r o m a t i c counterparts (piperidine 1.7).
N
5.1
N
H
1.7
A r o m a t i c heterocycles a r e d e s c r i b e d as b e i n g heteroaromatic, a n d w e shall
c o n c e n t r a t e o n t h e s e s y s t e m s in this b o o k at the e x p e n s e of m o r e saturated
s y s t e m s . L e t us n o w c o n s i d e r t h e c o n c e p t of a r o m a t i c i t y w i t h r e g a r d to
benzene.
T h e c o m p o u n d numbering s y s t e m
in this chapter is not as odd as it
might seem. For more on
c o m p o u n d 5.1 s e e Chapter 5, etc.
H
„H
H,
H"
^H
H
1.8a
T h e c a r b o n a t o m s in b e n z e n e are sp2 hybridised, and the h y d r o g e n atoms
are in the s a m e p l a n e as the c a r b o n a t o m s . T h e r e m a i n i n g six p orbitals are at
right a n g l e s to t h e p l a n e of the ring a n d c o n t a i n six n e l e c t r o n s . B e n z e n e
f u l f i l s t h e H i i c k e l criteria f o r a r o m a t i c i t y as a p p l i e d to c y c l i c p o l y e n e s
c o n t a i n i n g 4 n + 2 e l e c t r o n s ( w h e r e n-1 in this c a s e ) in f i l l e d p o r b i t a l s
capable of overlap.
A l t h o u g h t w o m e s o m e r i c representations 1 . 8 a , b can be d r a w n f o r benzene,
this d o e s not i m p l y t w o r a p i d l y - i n t e r c o n v e r t i n g f o r m s . R a t h e r , t h e six 7t
electrons are delocalised in m o l e c u l a r orbitals resulting in an a n n u l a r electron
cloud a b o v e and b e l o w t h e p l a n e of the ring. B e n z e n e can also be represented
b y structure 1.9, w h i c h e m p h a s i s e s the cyclical a r r a n g e m e n t of electrons. In
a g r e e m e n t with this theory, the c a r b o n - c a r b o n b o n d lengths are all equivalent
(0.14 n m ) a n d i n t e r m e d i a t e b e t w e e n that of a single (0.154 n m ) and d o u b l e
(0.133 n m ) c a r b o n - c a r b o n bond. The extra thermodynamic stabilisation
i m p a r t e d to b e n z e n e b y this p h e n o m e n o n of electron delocalisation, called
' r e s o n a n c e ' , c a n b e d e t e r m i n e d i n d i r e c t l y . R e a l , d e l o c a l i s e d b e n z e n e is
t h e r m o d y n a m i c a l l y m o r e stable than a theoretical c y c l o h e x a t r i e n e m o l e c u l e
(i.e. n o n - d e l o c a l i s e d structure 1.8a) b y a r o u n d 150 kJ mol" 1 .
H o w d o e s this c o n c e p t of aromaticity apply to typical heterocycles such as
p y r i d i n e 5 . 1 and p y r r o l e 2.1? P y r i d i n e can f o r m a l l y be derived f r o m b e n z e n e
b y r e p l a c e m e n t of a C H u n i t b y an sp2
hybridised nitrogen atom.
C o n s e q u e n t l y , p y r i d i n e h a s a l o n e pair of e l e c t r o n s instead of a h y d r o g e n
atom. H o w e v e r t h e six 7t electrons are essentially u n c h a n g e d , and the pyridine
is a relatively aromatic heterocycle.
Q
N
H
S.l
2.1
A difficulty arises with f i v e - m e m b e r e d heterocycles such as pyrrole, w h i c h
at first sight w o u l d a p p e a r to h a v e only f o u r n electrons, t w o short of the 4 n
+ 2 Hiickel criteria f o r aromaticity. T h e n i t r o g e n a t o m is sp2 h y b r i d i s e d a n d
f o r m a l l y c o n t a i n s a l o n e p a i r of electrons in the r e m a i n i n g p orbital at right
angles to the ring. H o w e v e r , the s y s t e m is delocalised, as s h o w n b e l o w .
YW j
H
2.1
©N
H
©
t>
©N
H
o©N
H
— o
T h u s , d e l o c a l i s a t i o n of t h e n i t r o g e n l o n e p a i r c o m p l e t e s t h e sextet of
e l e c t r o n s r e q u i r e d f o r a r o m a t i c i t y . T h e s e t w o e x a m p l e s illustrate the p o i n t
that certain heterocycles (closely analogous to b e n z e n e and naphthalene) such
as p y r i d i n e 5 . 1 , p y r i m i d i n e 10.1, a n d q u i n o l i n e 6 . 1 a r e a r o m a t i c 'by right'
w h e r e a s o t h e r h e t e r o c y c l e s such as p y r r o l e 2.1, i m i d a z o l e 3 . 2 , a n d triazole
8 . 7 h a v e to 'earn' a r o m a t i c i t y b y d e l o c a l i s a t i o n of a l o n e p a i r of electrons
from the heteroatom.
5.1
10.1
6.1
2.1
3.2
8.7
W h a t are the c o n s e q u e n c e s of this c o n c e p t of lone pair delocalisation f o r a
related series of h e t e r o c y c l e s such as p y r r o l e 2 . 1 , t h i o p h e n e 2 . 2 , and f u r a n
2.3? A s delocalisation results in electron loss f r o m the h e t e r o a t o m concerned,
t h e e x t e n t of d e l o c a l i s a t i o n ( a n d h e n c e a r o m a t i c i t y ) will v a r y w i t h the
electronegativity of the h e t e r o a t o m . T h e h i g h l y electronegative o x y g e n a t o m
in f u r a n h o l d s on to e l e c t r o n d e n s i t y m o r e strongly than t h e h e t e r o a t o m in
t h i o p h e n e o r p y r r o l e . F u r a n is g e n e r a l l y c o n s i d e r e d to h a v e a n o n - a r o m a t i c
electron distribution fairly close to that depicted b y structure 2.3.
Q
o
o
H
2.2
2.3
2.1
In f a c t the thorny p r o b l e m as to h o w a r o m a t i c is a particular heterocycle or
series of h e t e r o c y c l e s h a s b e e n a p r e o c c u p a t i o n of p h y s i c a l organic c h e m i s t s
f o r s o m e time. B o n d l e n g t h s , heats of c o m b u s t i o n , s p e c t r o s c o p i c data, and
t h e o r e t i c a l l y - c a l c u l a t e d r e s o n a n c e e n e r g i e s h a v e all b e e n i n v o k e d , b u t an
a b s o l u t e m e a s u r e of a r o m a t i c i t y r e m a i n s e l u s i v e . N e v e r t h e l e s s , t r e n d s
r e g a r d i n g relative aromaticity will b e alluded to in this text as they arise.
1.3 Synthesis of heterocycles
T h e r e a r e m a n y s y n t h e s e s of t h e m a j o r h e t e r o c y c l e s a n d t h e y a r e o f t e n
c o m p l e m e n t a r y in that t h e y a f f o r d d i f f e r e n t substitution patterns on the ring.
M o s t of t h e synthetic m e t h o d s w e shall e x a m i n e are fairly classical (indeed
s o m e are decidedly ancient!) although m a n y of the specific e x a m p l e s are quite
m o d e r n . M a n y c l a s s i c a l s y n t h e s e s of h e t e r o c y c l e s r e v o l v e a r o u n d t h e
c o n d e n s a t i o n reaction in its v a r i o u s guises. L e t us consider the m e c h a n i s m of
a s i m p l e a c i d - c a t a l y s e d c o n d e n s a t i o n , that of g e n e r a l i s e d k e t o n e 1 . 1 0 and
a m i n e 1 . 1 1 to g i v e i m i n e 1.12.
P r o t o n a t i o n of the k e t o n e o x y g e n a t o m activates the k e t o n e to
n u c l e o p h i l i c a t t a c k b y t h e a m i n e . L o s s of a p r o t o n f r o m 1 . 1 3 p r o d u c e s
neutral intermediate 1.14. A second protonation, once again on the oxygen
a t o m a f f o r d s 1.15, w h i c h o n loss of a w a t e r m o l e c u l e a n d a p r o t o n gives the
For a review on the concept of
heterocyclic aromaticity see
Katritzky et a! (1991).
i m i n e 1 . 1 2 . A l l t h e s e s t e p s a r e r e v e r s i b l e , b u t in p r a c t i c e if w a t e r c a n b e
r e m o v e d f r o m the e q u i l i b r i u m (for instance by azeotropic distillation) then
s u c h r e a c t i o n s c a n b e f o r c e d t o c o m p l e t i o n . T h i s t y p e of r e a c t i o n o c c u r s
m a n y t i m e s i n this t e x t , b u t in f u t u r e w i l l n o t b e p r e s e n t e d in s u c h d e t a i l .
T h e s t u d e n t is s t r o n g l y a d v i s e d t o w o r k t h r o u g h , u s i n g p e n a n d p a p e r , t h e
m e c h a n i s m shown below and the m a n y subsequent mechanisms. Confidence
with reaction m e c h a n i s m s will facilitate u n d e r s t a n d i n g of
heterocyclic
c h e m i s t r y a n d o r g a n i c c h e m i s t r y in g e n e r a l .
Ri
+
1.10
R!
catalytic H
H2N-R2
1.11
/=N-R
-H,0
j+H®
Rl
-H
V
"
K-l
Rl
H,N - R,
r2^h
1.13
1.12
-H,0
-H
r>„H
2
+H®
Kj
i
r2
i
R2
1.14
1.15
The disconnection approach to synthesis essentially involves working
backwards
from
a target c o m p o u n d
in a logical
manner
(so-called
r e t r o s y n t h e s i s ) , s o t h a t a n u m b e r of p o s s i b l e r o u t e s a n d s t a r t i n g m a t e r i a l s a r e
s u g g e s t e d . T h i s a p p r o a c h h a s b e e n a p p l i e d m a i n l y to a l i c y c l i c , c a r b o c y c l i c ,
a n d s a t u r a t e d h e t e r o c y c l i c s y s t e m s . R e t r o s y n t h e t i c a n a l y s e s a r e p r e s e n t e d in
this t e x t n o t as a n a l l - e m b r a c i n g a n s w e r t o s y n t h e t i c p r o b l e m s , b u t r a t h e r as
a n aid to u n d e r s t a n d i n g t h e a c t u a l c o n s t r u c t i o n of u n s a t u r a t e d h e t e r o c y c l e s .
R e t u r n i n g t o t h e c o n d e n s a t i o n p r e s e n t e d a b o v e , this l e a d s to a n i m p o r t a n t
d i s c o n n e c t i o n . T h e i m i n e - l i k e l i n k a g e p r e s e n t in s e v e r a l
heterocycles
( g e n e r a l i s e d s t r u c t u r e 1 . 1 6 ) c a n a r i s e f r o m c y c l i s a t i o n of 1 . 1 7 , c o n t a i n i n g
amino and carbonyl functionalities.
T h e s y m b o l => d e n o t e s a
d i s c o n n e c t i o n , a n analytical
p r o c e s s in w h i c h a structure is
t r a n s f o r m e d into a suitable
precursor
-NH,
N'
1.16
1.17
N o w c o n s i d e r c o n d e n s a t i o n of a m m o n i a w i t h k e t o e s t e r 1.18. T h e i s o l a t e d
p r o d u c t is n o t i m i n e 1 . 1 9 b u t t h e t h e r m o d y n a m i c a l l y m o r e s t a b l e e n a m i n e
tautomer 1.20 which has a conjugated double bond system and a strong
intramolecular hydrogen-bond. Although not a heterocyclic example, 1.20
i l l u s t r a t e s t h a t a n e n a m i n e - l i k e l i n k a g e , as in g e n e r a l i s e d h e t e r o c y c l e 1 . 2 1 , is
also accessible b y a condensation reaction.
In a retrosynthetic sense, f o r m a l hydrolysis of the carbon-nitrogen b o n d of
1.21 reveals enol 1.22 w h i c h w o u l d exist as the m o r e stable ketone tautomer
1 . 2 3 . N o t e that in the h y d r o l y t i c d i s c o n n e c t i o n step the c a r b o n b e c o m e s
attached to a hydroxy g r o u p and the nitrogen to a hydrogen atom - there is n o
change in the oxidation levels of carbon or nitrogen.
H
1.21
1.22
1.23
U n l i k e o u r initial i m i n e d i s c o n n e c t i o n w h i c h is restricted to n i t r o g e n
h e t e r o c y c l e s (with o n e o r t w o specific e x c e p t i o n s such as p y r y l i u m salts, see
C h a p t e r 9), the h e t e r o a t o m in the e n a m i n e or e n a m i n e - l i k e d i s c o n n e c t i o n
could b e divalent. T h e r e f o r e this disconnection is also applicable to oxygenand sulphur-containing heterocycles, typified by 1.24 and 1.25.
1.25
Let us see h o w this disconnection approach can rationalize the synthesis of
pyrrole 2.16.
2.16
+
Retrosynthetic analysis suggests a double c o n d e n s a t i o n b e t w e e n diketone
1.26 and a m m o n i a . P y r r o l e 2 . 1 6 can actually b e p r e p a r e d if this w a y - see
Chapter 2.2.
A n o t h e r aid to u n d e r s t a n d i n g heterocyclic synthesis in general is the fact
that a large n u m b e r of five- and six-membered heterocycles can be constructed
f r o m v a r i o u s c o m b i n a t i o n s of small acyclic m o l e c u l e s by c o m p l e m e n t a r y
m a t c h i n g of nucleophilic and electrophilic functionality.
C-C
©C
1.26
o
o
NH,
t -
C©
e 3
NH
2
Returning to the synthesis of pyrrole 2.16, diketone 1.26 can b e regarded as a
f o u r - c a r b o n bis-electrophilic f r a g m e n t and a m m o n i a , in this instance, as a
bis-nucleophilic nitrogen f r a g m e n t . A m m o n i a can f o r m up to three b o n d s in
a nucleophilic manner.
NH3
1.26
In this particular i n s t a n c e the correct oxidation level automatically results
f r o m the c o n d e n s a t i o n reaction, g i v i n g p y r r o l e 2 . 1 6 directly. In other cases
c y c l i s a t i o n d o e s n o t a f f o r d the c o r r e c t o x i d a t i o n level and an u n s a t u r a t e d
s y s t e m has to be oxidised to a c h i e v e aromaticity. For instance, 1,5-diketones
1.27 react with a m m o n i a to give dihydropyridines 1.28 which can be oxidised
to p y r i d i n e s 1.29.
nh3^
•o
-H,0
O — R
[O] ^
II
11
R - S R
H
2
1.27
- 2H
„
1.28
1.29
E x a m p l e s of this c y c l i s a t i o n - o x i d a t i o n strategy i n c l u d e the s y n t h e s i s of
p y r i d o t r i a z i n e 5 . 3 2 ( p a g e 4 2 ) and s y n t h e s e s of quinolines and isoquinolines
( C h a p t e r 6). S o m e e x a m p l e s of nucleophilic and electrophilic f r a g m e n t s are
s h o w n in T a b l e 1.1. S e v e r a l points arise f r o m the table.
C o n s i d e r a c y l a t i n g species such as acid chlorides. A c y l a t i o n of d i a m i n e
1.30 initially gives a m i d e 1 . 3 1 w h i c h u n d e r g o e s a c o n d e n s a t i o n to p r o d u c e
b e n z i m i d a z o l e 1 . 3 2 . T h e c a r b o n y l m o i e t y is a c t i n g e x c l u s i v e l y as an
electrophilic centre.
1.32
H o w e v e r , delocalisation of the n i t r o g e n l o n e pair in the a m i d e l i n k a g e (see
mesomeric representations 1.33a,b) produces a nucleophilic oxygen atom
which can react with electrophiles as s h o w n .
o
R
I
H
1.33a
K®A
N'
I
H
1.33b
O J
vj
I
H
.
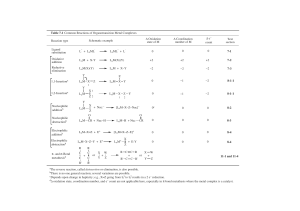
Nucleophilic fragments
Table 1.1
No. of ring atoms
1
NH3,H20,H2S
2
(see Chapters 2 and 5)
H2N— NH 2 , H2N— OH
(see pyrazole and isoxazole synthesis,
Chapter4)
(see quinoline synthesis, Chapter 6)
(see benzimidazole synthesis, Chapter 1)
(see isoquinoline synthesis, Chapter 6)
)
NH 2
V
J
Electrophilic fragments
No. of ring atoms
O
A
R
(X = leaving group, eg. CI- see benzimidazole synthesis, Chapter 1
x
and isoquinoline synthesis, Chapter 6)
2
(see thiazole synthesis, Chapter 3)
O
^
i f
Rj,k2
^See 4u'n°l'ne
synthesis, Chapter 6)
= aiKyi or u-aiKyi
(see pyrazole and isoxazole synthesis, Chapter 4, and pyrimidine synthesis, Chapter 10)
^
^
\\
O
U
^
(see pyrrole, thiophene, and furan syntheses, Chapter 2)
O
Nucleophilic / Electrophilic fragments
No. of ring at
2
(see Chapter 1 and oxazole synthesis, Chapter 3)
(see Chapter 1 and coumarin synthesis, Chapter 9)
(see oxazole synthesis, Chapter 3, and Knorr pyrrole
synthesis, Chapter 2)
The reaction of an acylating species
with a nucleophile is shown below.
A m i d e s c a n c y c l i z e in this m a n n e r as, f o r e x a m p l e , in the acylation of a m i n o
acids 1.34 to a f f o r d o x a z o l i d i n o n e s 1.35.
c°
O
R,
r
R
Nuc
Nuc
e
o
0"O
O
excess
R,
a
->
OH
NH2
Acylating
N^/
1.35
1.34
species
are
thus
included
in
both
electrophilic
and
n u c l e o p h i l i c / e l e c t r o p h i l i c c a t e g o r i e s in T a b l e 1.1. F o r a r e l a t e d e x a m p l e see
the s y n t h e s i s of o x a z o l e s in C h a p t e r 3.
o
1,3-Dicarbonyl c o m p o u n d s , such as malonate derivatives, can also b e
c l a s s i f i e d u n d e r t w o c a t e g o r i e s . A s w e l l as r e a c t i n g s i m p l y as a t h r e e - a t o m
R
Nuc
b i s - e l e c t r o p h i l i c f r a g m e n t (as in t h e s y n t h e s i s of b a r b i t u r a t e 1 0 . 2 5 ( p a g e 7 7 ) ,
a n alternative reactivity is aivailable.
C o n d e n s a t i o n (by n u c l e o p h i l i c attack)
of t h e a c t i v e m e t h y l e n e c a r b o n a n d e l e c t r o p h i l i c r e a c t i o n at j u s t o n e of t h e
c a r b o n y l g r o u p s is a t w o - a t o m n u c l e o p h i l i c / e l e c t r o p h i l i c p r o f i l e , as s e e n in
the p r e p a r a t i o n of c o u m a r i n 9.16.
For the spke of simplicity in this text
the two-stage process is abbreviated
thus:
H
Br.
r'
O
0 H
OEt
EtO ' ^ O
- H20
- EtOH
(T0
|
C0 2 Et
Br.
9.16
O
Nuc
OEt
©
Nuc"
EtO
o
R^Nuc
^O
T h e s e c o n c e p t s of retrosynthesis a n d h e t e r o c y c l e construction will h e l p p u t
the syntheses encountered
in t h e f o l l o w i n g c h a p t e r s i n t o a
broader
perspective.
1.4 References
Textbooks
Acheson, R.M. (1967).
compounds,
An introduction
to the chemistry
of
heterocyclic
( 2 n d edn). W i l e y , N e w Y o r k .
Paquette, L.A. (1966).
Principles
of modern
heterocyclic
chemistry.
Benjamin, N e w York.
Joule, J.A. and
Smith, G.F. (1979).
Heterocyclic
chemistry,
(2nd edn).
Van Nostrand Reinhold, New York.
Gilchrist, T.L. (1985).
Heterocyclic
chemistry.
Longman,
Harlow.
T h e first t w o ( A c h e s o n and P a q u e t t e ) are still very g o o d texts e v e n today. Of
the m o r e r e c e n t pair, b o t h are w a r m l y r e c o m m e n d e d . J o u l e a n d S m i t h is
p o s s i b l y a m o r e i n t r o d u c t o r y text t h a n G i l c h r i s t , w h i c h c o n t a i n s m a n y
j o u r n a l r e f e r e n c e s and is pitched at the a d v a n c e d u n d e r g r a d u a t e / p o s t g r a d u a t e
l e v e l . S e e G i l c h r i s t f o r a d i s c u s s i o n of t h e n u c l e o p h i l i c / e l e c t r o p h i l i c
f r a g m e n t a p p r o a c h to heterocyclic synthesis.
W a r r e n , S. ( 1 9 7 8 ) . Designing
organic
syntheses,
p.150-172. Wiley,
Chichester.
W a r r e n S. ( 1 9 8 2 ) . Organic
synthesis
- the disconnection
approach,
p.
3 2 6 0 - 3 4 5 . W i l e y , Chichester.
Reference books and series
C o f f e y , S. (ed.) ( 1 9 7 3 - 1986). Heterocyclic
compounds
(Vols. 4 A - 4 K of
Rodd's chemistry of carbon compounds).
Elsevier, A m s t e r d a m .
E l d e r f i e l d , R . C . (ed.) ( 1 9 5 0 - 1967). Heterocyclic
chemistry,
V o l s . 1 - 9.
Wiley, N e w York.
K a t r i t z k y , A . R . a n d B o u l t o n , A . J . (ed.) ( 1 9 6 3 - 1989). Advances
in
heterocyclic
chemistry,
V o l s . 1 - 4 5 . A c a d e m i c Press, O r l a n d o .
Katritzky, A . R . and R e e s , C . W . (ed.) (1984). Comprehensive
heterocyclic
chemistry,
Vols. 1 - 8. P e r g a m o n Press, O x f o r d .
K a t r i t z k y , A . R . et al, (1991). Heterocycles,
32, 1 2 7 - 1 6 1 .
S a m m e s , P.G. (ed.) (1979).
Heterocyclic
chemistry
( V o l . 4 of
Comprehensive
organic
chemistry,
ed. D . B a r t o n and W . D . Ollis).
P e r g a m o n Press, O x f o r d .
W e i s s b u r g e r , A . and T a y l o r , E . C . (ed.) ( 1 9 5 0 - 1990). The chemistry
of
heterocyclic
compounds.
W i l e y Interscience, N e w Y o r k .
All of these sources c o n t a i n e x c e l l e n t r e v i e w s o n virtually e v e r y aspect of
h e t e r o c y c l i c c h e m i s t r y . In p a r t i c u l a r , K a t r i t z k y and R e e s is a t h o r o u g h l y
c o m p r e h e n s i v e w o r k . F o r t h o s e p a r t i c u l a r l y interested in t h e s y n t h e s i s of
heterocycles as pharmaceutical agents see:
L e d n i c e r , D . a n d M i t s c h e r , L . A . ( 1 9 7 7 , 1980, 1984, a n d 1990).
chemistry of drug synthesis, V o l s . 1 - 4 . W i l e y , N e w Y o r k .
Organic
Experimental references
In this i n t r o d u c t o r y t e x t t h e r e is little d e t a i l r e g a r d i n g s o l v e n t s , y i e l d s ,
w o r k u p p r o c e d u r e s , etc. H o w e v e r , s e v e r a l c h a p t e r s r e f e r e n c e a r e l e v a n t
e x p e r i m e n t a l p r o c e d u r e (taken f r o m Organic syntheses or Vogel) which the
s t u d e n t is s t r o n g l y e n c o u r a g e d to r e a d . F o r an e x c e l l e n t s e l e c t i o n of
experimental procedures f o r the synthesis of heterocycles see:
F u r n i s s , B.S., H a n n a f o r d , A.J., S m i t h , P . W . G . , and Tatchell, A . R . (1989).
Vogel's
textbook
of practical
organic
chemistry
(5th edn),
pp. 1127 - 1194. L o n g m a n , H a r l o w .
2. Pyrroles, thiophenes, and
furans
2.1 Introduction
Pyrrole
2.1,
thiophene 2.2, and furan 2.3,
are
five-membered
ring
heteroaromatic c o m p o u n d s containing one heteroatom. T h e y derive their
aromaticity from delocalisation
of a lone pair of e l e c t r o n s f r o m
the
h e t e r o a t o m . C o n s e q u e n t l y t h e l o n e p a i r is n o t a v a i l a b l e f o r p r o t o n a t i o n a n d
T h e n u m b e r i n g of h e t e r o c y c l e s
generally starts at the h e t e r o a t o m
hence these heterocycles are not basic.
4/
H
N
N© H
H
2.1
H
U n d e r e x t r e m e c o n d i t i o n s of
acidity p y r r o l e is p r o t o n a t e d , but
at the C2 position.
' a
2.2
2.3
T h e b a s i s a n d e x t e n t of t h e i r a r o m a t i c i t y is d i s c u s s e d in C h a p t e r 1. I n
s u m m a r y , the capacity for the lone pair on a particular heteroatom to be
d e l o c a l i s e d is i n v e r s e l y r e l a t e d to t h e e l e c t r o n e g a t i v i t y of t h e h e t e r o a t o m . F o r
i n s t a n c e , f u r a n is t h e l e a s t a r o m a t i c of t h e t r i o b e c a u s e o x y g e n h a s t h e
greatest electronegativity and hence mesomeric representations 2.4b-e m a k e
r e l a t i v e l y l e s s of a c o n t r i b u t i o n to t h e e l e c t r o n i c s t r u c t u r e of f u r a n t h a n t h e y
d o in t h e c a s e s of p y r r o l e a n d t h i o p h e n e . T h e o r d e r of a r o m a t i c i t y is f u r a n <
p y r r o l e < t h i o p h e n e . W e s h a l l s e e l a t e r h o w this v a r i a t i o n in a r o m a t i c i t y
a f f e c t s t h e reactivities of t h e s e three related h e t e r o c y c l e s .
QNFFI
h/\H
N o t e that protonation of the
p y r r o l e nitrogen w o u l d l e a d to a
n o n - a r o m a t i c cation.
P
e
1
e Q
yv
2.4b
2.4a
2.4c
—
t >
2.4d
-
C I
2.4e
X = NH,S,O
A s m a l l n u m b e r of s i m p l e p y r r o l e s s u c h as 2 . 5 a n d 2 . 6 o c c u r n a t u r a l l y .
Far more important are the tetramic pyrrole derivatives (porphyrins) such as
chlorophyll-a 2.7 and h a e m 2.8.
\-o
\J
2.5
o.
C20H34O
A c e t y l e n i c t h i o p h e n e 2 . 9 , f o u n d in s o m e s p e c i e s of h i g h e r p l a n t s , is o n e
C h l o r o p h y l l - a is a plant p i g m e n t
i n v o l v e d in the c r u c i a l
p h o t o s y n t h e t i c p r o c e s s in w h i c h
the e n e r g y of sunlight is
h a r n e s s e d to i n c o r p o r a t e c a r b o n
d i o x i d e into plant m e t a b o l i s m .
H a e m , h o w e v e r , is f u n d a m e n t a l to
m a m m a l i a n biology, b e i n g the
o x y g e n - b i n d i n g c o m p o n e n t of
haemoglobin. Oxygen absorbed
f r o m the air is t r a n s p o r t e d a r o u n d
the b o d y w h i l e t e m p o r a r i l y coo r d i n a t e d to the iron a t o m of
h a e m o g l o b i n , w h i c h o c c u r s in t h e
red b l o o d cells.
of t h e f e w n a t u r a l l y - o c c u r r i n g t h i o p h e n e s . H o w e v e r , t h e t h i o p h e n e r i n g is
u s e d in s e v e r a l i m p o r t a n t p h a r m a c e u t i c a l p r o d u c t s , s u c h as t h e p e n i c i l l i n
antibiotic 2.10.
CO,H
CO,H
2.10
In c o n t r a s t to t h e p y r r o l e a n d t h i o p h e n e series, t h e f u r a n n u c l e u s o c c u r s in
m a n y p l a n t - d e r i v e d t e r p e n e s s u c h as 2 . 1 1 . T h e m o s t i m p o r t a n t f u r a n c o n t a i n i n g d r u g is 2 . 1 2 , w h i c h r e d u c e s g a s t r i c acid s e c r e t i o n a n d is i m p o r t a n t
in t h e t r e a t m e n t of ulcers.
/y
9
H„/
NHMe
O
u
,
NMe,
2.12
2.11
2.2. Synthesis of pyrroles, thiophenes, and furans
We
shall
first examine
a general
synthesis
applicable
to all
three
h e t e r o c y c l e s , then c o n s i d e r t w o s p e c i f i c s y n t h e s e s of p y r r o l e s .
R e t r o s y n t h e t i c c l e a v a g e of a c a r b o n - h e t e r o a t o m b o n d in 2 . 1 3 g i v e s e n o l
2 . 1 4 w h i c h is e q u i v a l e n t t o k e t o n e 2 . 1 5 . R e p e a t i n g t h e p r o c e s s g i v e s u s a
1 , 4 - d i c a r b o n y l c o m p o u n d a n d t h e h e t e r o a t o m - c o n t a i n i n g f r a g m e n t s u c h as a
primary amine or hydrogen sulphide.
Terpenes are plant-derived
natural p r o d u c t s c o n s t r u c t e d of
multiples of the f i v e - c a r b o n
hydrocarbon isoprene.
T h e f o r w a r d p r o c e s s is k n o w n as the P a a l - K n o r r synthesis. T h i s is a very
s t r a i g h t f o r w a r d s y n t h e s i s l i m i t e d o n l y b y t h e a c c e s s i b i l i t y • of t h e 1,4dicarbonyl precursors.
The mechanism is illustrated by
the preparation of 2,5-dimethyl
pyrrole 2.16 and is simply two
consecutive condensations.
T h e P a a l - K n o r r s y n t h e s i s c a n s i m i l a r l y be a p p l i e d to t h i o p h e n e s , e.g.
c o m p o u n d s 2 . 1 7 - 2.20.
W h e n h y d r o g e n s u l p h i d e is t h e h e t e r o a t o m s o u r c e t h e m e c h a n i s m is
similar to the p y r r o l e case.
H o w e v e r , t h e s i t u a t i o n is slightly d i f f e r e n t w h e n p h o s p h o r u s ( V )
s u l p h i d e is used. T h i s r e a g e n t c o n v e r t s k e t o n e s to thioketones, b y e x c h a n g e
of a p h o s p h o r u s - s u l p h u r d o u b l e b o n d with a c a r b o n - o x y g e n d o u b l e b o n d .
F o r i n s t a n c e , in t h e s y n t h e s i s of 2 . 1 9 , t h e 1 , 4 - d i k e t o n e is c o n v e r t e d i n t o
the c o r r e s p o n d i n g 1 , 4 - d i t h i o k e t o n e f o l l o w e d b y loss of h y d r o g e n s u l p h i d e .
p2s5
Ph
^
O
T h e m e c h a n i s m of the cyclisation
step is similar to that of t h i o p h e n e
2.17.
H,S
^ P h
O
Ph
Ph
S
Ph
S
Ph
S
2.19
O u r r e t r o s y n t h e t i c a n a l y s i s of t h e P a a l - K n o r r s y n t h e s i s l e a d s to a p r o b l e m
w h e n a p p l i e d to f u r a n , as it i m p l i e s a d d i t i o n of a w a t e r m o l e c u l e , f o l l o w e d b y
e l i m i n a t i o n of t w o w a t e r m o l e c u l e s . In p r a c t i c e , s i m p l e d e h y d r a t i o n of a 1,4d i c a r b o n y l c o m p o u n d l e a d s to f u r a n s as in t h e p r e p a r a t i o n of 2.21.
oO
Ph
Ph
2.21
R e t u r n i n g again to pyrroles, p r o b a b l y the m o s t widely-used m e t h o d f o r
their p r e p a r a t i o n is t h e K n o r r p y r r o l e s y n t h e s i s , w h i c h is t h e c o n d e n s a t i o n o f
a k e t o n e 2 . 2 2 w i t h a n a - a m i n o k e t o n e 2 . 2 3 to g i v e p y r r o l e 2 . 1 3 , v i a e n a m i n e
2 . 2 4 . A r e a s o n a b l e m e c h a n i s m is s h o w n b e l o w , a l t h o u g h n o n e o f t h e
i n t e r m e d i a t e s is isolated.
K
1
V
2.22
s
k
^
i
v
R
2.23
R2
R3
OH
1
H
N
2.13
T h e a - a m i n o k e t o n e s a r e o f t e n p r e p a r e d b y n i t r o s a t i o n of a n
active
m e t h y l e n e g r o u p f o l l o w e d b y r e d u c t i o n of t h e o x i m e to t h e a m i n e (e.g. 2 . 2 5
t o 2 . 2 6 to 2 . 2 7 ) .
O
W?
C0 2 Et
2.25
, C0 2 Et
-
C 5 H h OH
V
AcOH
°
Jl,C0
H
O
2
U N : 0
C0 2 Et
Et
«
^
N
2.26
c -
A s a - a m i n o k e t o n e s are p r o n e to self-condensation (see p a g e 2 2 for a
d i s c u s s i o n of a - a m i n o k e t o n e s ) , t h e initial c o n d e n s a t i o n s t e p is f a c i l i t a t e d b y
R-2 in 2 . 2 2
being an electron-withdrawing
group. This enhances
the
e l e c t r o p h i l i c n a t u r e of t h e k e t o n e c a r b o n y l g r o u p t h e r e b y i n c r e a s i n g t h e r a t e
of t h e d e s i r e d r e a c t i o n , a n d f a v o u r s e n a m i n e t a u t o m e r 2 . 2 4 o v e r t h e i m i n o
CO,Et
Zn
AcOH
NH 2
2.27
t a u t o m e r b e c a u s e of c o n j u g a t i o n w i t h t h e e l e c t r o n - w i t h d r a w i n g g r o u p . A
s e l e c t i o n of K n o r r p y r r o l e s y n t h e s e s , s h o w i n g t h e k e y i n t e r m e d i a t e e n a m i n e s ,
is s h o w n b e l o w .
COjEt
CO,Et
X
The Knorr pyrrole synthesis
consists of a k e t o n e a n d a m i n e
c o n d e n s i n g to give an e n a m i n e ,
f o l l o w e d by i n t r a m o l e c u l a r
cyclisation of this e n a m i n e onto
the remaining ketone.
H2N
X
:o 2 Et
CO,Et
0
N
H
^ C0 2 Et
COzEt
y -
C0 22Et
/
N-
C0 2 Et
H
o
H
J '
H,N
W
V
H
C(
COjEt
COjEt
\Et
0^,C5H
C0 2 Et
C0 2 Et
n
-
r i
C0 22Et
H
N
H
Ph
Ph
Ph
l
2.3
Note that pyrrole reacts w i t h
e l e c t r o p h i l e s o n c a r b o n , like a n
enamine.
H,N
Ph
O^-Ph
° I Ph
//
-N^^ph
C5Hn
%
H
Electrophilic substitution of pyrrole, thiophene,
and furan
All t h r e e h e t e r o c y c l e s u n d e r g o a r o m a t i c s u b s t i t u t i o n r e a c t i o n s , t h o u g h t h e i r
reactivities vary considerably. Let us consider a generalised m e c h a n i s m and
h o w t h e s t a b i l i t y of t h e t w o p o s s i b l e i n t e r m e d i a t e s a f f e c t s t h e p o s i t i o n of
substitution.
CY_ -CX—
v >
o
X
>
X=NH,S,0
X
H
2.28a
H
"X
2.28b
2.28c
H
H
E
1
-H
<—1>
// w
v
X
2.29a
(&'H
x'
2.29b
T h e i n t e r m e d i a t e d e r i v e d f r o m a t t a c k at t h e C 2 p o s i t i o n h a s g r e a t e r
d e l o c a l i s a t i o n of t h e p o s i t i v e c h a r g e ( m e s o m e r i c f o r m s 2 . 2 8 a , b , c ) t h a n that
d e r i v e d f r o m a t t a c k at t h e C 3 p o s i t i o n ( m e s o m e r i c f o r m s 2 . 2 9 a , b ) . A s t h e
c h a r g e is m o r e e x t e n s i v e l y d e l o c a l i s e d in t h e f o r m e r , this i n t e r m e d i a t e is at
l o w e r e n e r g y . T h i s in t u r n is r e f l e c t e d in a l o w e r a c t i v a t i o n e n e r g y f o r this
p a t h w a y a n d m a n i f e s t e d in a s e l e c t i v i t y f o r e l e c t r o p h i l i c s u b s t i t u t i o n at t h e
C 2 position over the C 3 position. T h e actual isomer ratio depends on the
h e t e r o c y c l e , t h e e l e c t r o p h i l e , a n d t h e p r e c i s e c o n d i t i o n s , a l t h o u g h in m a n y
cases such reactions are virtually regiospecific, and only the C 2 substitution
p r o d u c t s a r e i s o l a t e d . V e r y r e a c t i v e e l e c t r o p h i l e s ( s u c h as t h e n i t r o n i u m ion
N 0 2 + ) exhibit lower selectivity b e c a u s e they tend to b e less discriminating
as t o w h e r e t h e y a t t a c k t h e h e t e r o a r o m a t i c n u c l e u s .
T h e e a s e of e l e c t r o p h i l i c s u b s t i t u t i o n is p y r r o l e > f u r a n > t h i o p h e n e >
benzene.
T o give a quantitative feel for
t h e s e d i f f e r e n c e s in reactivity,
d a t a for the b r o m i n a t i o n of three
r e p r e s e n t a t i v e d e r i v a t i v e s are
shown below.
P y r r o l e is e x t r e m e l y r e a c t i v e t o w a r d s e l e c t r o p h i l e s w h i l e t h i o p h e n e , t h e
most
aromatic
o f t h e t r i o , is m u c h
less reactive.
At a very
rough
a p p r o x i m a t i o n , t h e r e a c t i v i t y of t h i o p h e n e is of t h e o r d e r of a h e t e r o a t o m s u b s t i t u t e d b e n z e n e d e r i v a t i v e s u c h as p h e n o l .
D e s p i t e l a r g e d i f f e r e n c e s in
t h e r a t e s of e l e c t r o p h i l i c s u b s t i t u t i o n s t h e r e a r e s o m e i m p o r t a n t a r o m a t i c
s u b s t i t u t i o n r e a c t i o n s c o m m o n to all t h r e e h e t e r o c y c l e s .
CO,Me
T h e V i l s m e i e r r e a c t i o n is t h e f o r m y l a t i o n of r e a c t i v e a r o m a t i c c o m p o u n d s
by
using
a
combination
of
phosphorus
oxychloride
and
N,N-
Br,
dimethylformamide, followed by a hydrolytic workup.
o
1. Me2NCHO / POCI3
//
"X
2. H , 0
X
X=NH,S,0
CO,Me
H
T h e r e a c t i o n p r o c e e d s b y f o r m a t i o n of t h e e l e c t r o p h i l i c V i l s m e i e r c o m p l e x
X
2 . 3 0 , f o l l o w e d b y e l e c t r o p h i l i c s u b s t i t u t i o n of t h e h e t e r o c y c l e . T h e f o r m y l
g r o u p is g e n e r a t e d in t h e h y d r o l y t i c w o r k u p . P y r r o l e , t h i o p h e n e , a n d f u r a n all
u n d e r g o this f o r m y l a t i o n w h i c h is h i g h l y s e l e c t i v e f o r t h e C 2 p o s i t i o n .
a
1
1.2 x 102
5.6 x 108
Cl°
\
Cl-P.O
CP
C l ^
V-NMe2
H k j
CI
v ©
ci
H"^NMe2
e
2.30
H i ^ J
NMe,
o
o
s
o
NH
O
11
Cl- P - CI
</ X
Relative Rate
-H
F
//
V
Q
NMe,
NMe2 CI
H
All three heterocycles u n d e r g o sulphonation with the p y r i d i n e - s u l p h u r
trioxide c o m p l e x . This b e h a v e s like a mild source of sulphur trioxide,
enabling the sulphonation to be carried out under essentially
neutral
conditions.
o
N-
O
II
s-
e
II
o
( X
X
X=NH,S,0
SO,H
F u r a n a n d p y r r o l e a r e n o t s t a b l e to m i n e r a l a c i d s , b u t a c e t y l n i t r a t e c a n b e
u s e d f o r t h e n i t r a t i o n of all t h r e e h e t e r o c y c l e s .
NO,
—H
cx
NO,
e
H,0
•HNMe 2
// V
^X
H
T h e f o r m a t i o n of 2 . 3 2 raises a n
i m p o r t a n t theoretical point:
b e c a u s e f u r a n is not very a r o m a t i c
a n d the driving f o r c e to 'rearomatise' by loss of a proton is
not v e r y s t r o n g , cation 2 . 3 1 c a n
be i n t e r c e p t e d to give 2 . 3 2 . T h i s
b e h a v i o u r is not o b s e r v e d w i t h
pyrrole and thiophene.
W h i l s t t h e m e c h a n i s m s h o w n a b o v e a p p l i e s to p y r r o l e a n d t h i o p h e n e , t h e
n i t r a t i o n of f u r a n w i t h a c e t y l n i t r a t e g i v e s t h e 2 , 5 - a d d i t i o n p r o d u c t 2 . 3 2 ,
a r i s i n g f r o m a t t a c k of a c e t a t e i o n o n t h e i n t e r m e d i a t e c a t i o n 2 . 3 1 . T r e a t m e n t
of 2 . 3 2 w i t h p y r i d i n e e l i m i n a t e s t h e e l e m e n t s of a c e t i c a c i d p r o d u c i n g
nitrofuran 2.33.
o
o
Q
C
Xr
N0!
CV
f o ffi H
NO,
-AcOH
NO,
© v
2.3
AcO
2.31
2.32
m d
2.33
Thiophene, alkyl-substituted furans, and especially pyrrole,
undergo
Mannich reactions.
CH 2 0/HNEt 2
Q
AcOH
NEt,
N
H
H
CH20/HNMe2 •
HC1
CH20/HNMe2 •
AcOH
J J
NMe,
/ M
NMe,
T h i s i n v o l v e s c o n d e n s a t i o n of t h e h e t e r o c y c l e , f o r m a l d e h y d e , a n d a n a m i n e
( u s u a l l y a s e c o n d a r y a m i n e ) to g i v e a n a m i n o m e t h y l d e r i v a t i v e .
Me,NH + C H , 0
AcOH
• H,0
eOAc
CH-p= NMe,
(h
O
r
CH7—— NMe, r X
NMe,
NMe,
X
H
T h e F r i e d e l - C r a f t s acylation and alkylation reactions are f u n d a m e n t a l
p r o c e s s e s in a r o m a t i c c h e m i s t r y . P y r r o l e s a n d f u r a n s a r e n o t s t a b l e t o t h e
L e w i s a c i d s n e c e s s a r y f o r t h e s e r e a c t i o n s , b u t t h i o p h e n e s a r e s t a b l e to L e w i s
acids, a n d d o u n d e r g o F r i e d e l - C r a f t s acylation and alkylation.
Q
o
PhCOCl
A1C1,
V
O
Ph
/ / \
SnCL
O b s e r v e that electrophilic
substitution o c c u r s at the C 3
position w h e n both t h e C 2 a n d C5
p o s i t i o n s are b l o c k e d .
EtBr
s
A1C1,
//
V
,0
T h e r e a c t i v i t y o f all t h r e e h e t e r o c y l e s is c o n s i d e r a b l y r e d u c e d
when
e l e c t r o n - w i t h d r a w i n g g r o u p s a r e p r e s e n t o n t h e r i n g . T h i s is i m p o r t a n t in t h e
s y n t h e s i s of p y r r o l e d e r i v a t i v e s as it a d d s c h e m i c a l s t a b i l i t y t o t h e r i n g ,
e n a b l i n g r e a c t i o n s to b e p e r f o r m e d in the p r e s e n c e of L e w i s acids.
T h e r e g i o c h e m i s t r y of t h e s e
r e a c t i o n s is easily e x p l a i n e d by
rationalisations f r o m classical
b e n z e n e c h e m i s t r y , i.e. e l e c t r o n w i t h d r a w i n g g r o u p s direct meta.
o
jtC
AcONO,
NO2
O
2.4 Anion chemistry of pyrroles, thiophenes, and
furans
P y r r o l e h a s a w e a k l y a c i d i c h y d r o g e n a t o m a t t a c h e d to t h e n i t r o g e n ( p ^ a =
17.5) a n d c a n b e d e p r o t o n a t e d b y s t r o n g b a s e s . T h e s o d i u m a n d p o t a s s i u m
salts a r e i o n i c in c h a r a c t e r a n d t h e n a k e d a n i o n t e n d s to r e a c t o n n i t r o g e n as in
t h e p r e p a r a t i o n of N - m e t h y l p y r r o l e 2 . 3 4 . T h e c o r r e s p o n d i n g m a g n e s i u m
d e r i v a t i v e 2 . 3 5 h a s m o r e c o v a l e n t c h a r a c t e r a n d t e n d s to r e a c t m o r e o n c a r b o n
t h a n n i t r o g e n , as in t h e p r e p a r a t i o n of p y r r o l e a l d e h y d e 2 . 3 6
f
\
NaNH^
N
H
f
\
Mel
>
N
e
© Na
f
\
2.34
N
Me
2.1
( f—TV H ' V ^ O E t
r=\
f
N
O
o
N
H
2.1
N
H
N
H
MgBr
2.35
«
2.36
N - m e t h y l p y r r o l e 2 . 3 4 , t h i o p h e n e , a n d f u r a n c a n b e m e t a l l a t e d at t h e C 2
p o s i t i o n w i t h a l k y l l i t h i u m r e a g e n t s . T h i s p o s i t i o n is m o r e a c t i v a t e d t o
deprotonation than the C 3 position b e c a u s e of the e l e c t r o n - w i t h d r a w i n g
i n d u c t i v e e f f e c t of t h e h e t e r o a t o m . T h e n u c l e o p h i l i c 2 - l i t h i o s p e c i e s c a n t h e n
b e r e a c t e d w i t h v a r i o u s e l e c t r o p h i l e s , a s in t h e p r e p a r a t i o n of 2 . 3 7 , 2 . 3 8 ,
a n d 2 . 3 9 . L e t u s s e e h o w this m e t h o d o l o g y c a n b e a p p l i e d t o t h e s y n t h e s i s o f
2 . 4 2 , a f u r a n - c o n t a i n i n g m i m i c of a l o n g - c h a i n f a t t y a c i d . D e p r o t o n a t i o n o f
f u r a n a n d a l k y l a t i o n p r o d u c e s 2 . 3 9 . A s e c o n d d e p r o t o n a t i o n at t h e C 5
p o s i t i o n a n d a l k y l a t i o n g i v e s b r o m i d e 2 . 4 0 . D i s p l a c e m e n t of t h e b r o m i d e
a f f o r d s nitrile 2 . 4 1 , a n d a c i d i c h y d r o l y s i s y i e l d s the target f u r a n 2 . 4 2 .
o
n-BuLi
•
N
Me
Me
T h e precise n a t u r e of the c a r b o n lithium b o n d is b e y o n d t h e s c o p e
of this b o o k . O r g a n o l i t h i u m
i n t e r m e d i a t e s are here
r e p r e s e n t e d as c a r b a n i o n a n d
c a t i o n to e m p h a s i s e d i f f e r e n c e s in
properties a n d reactivities as
c o m p a r e d w i t h full c o v a l e n t b o n d s .
as ClC0 2 Me
e Li
cx
N
Me
CO,Me
2.37
ClP(0)(0Et) 2
/
r
y
" -
B
u
L
i
»
/
r
\
© Li
S
2.2
T h e alkyl g r o u p at the C 2 position
is not d e p r o t o n a t e d in the s e c o n d
alkylation.
r \
2.38
n-BuLi / CH 3 (CH 2 ) 6 Br
n-BuLi / Br(CH2)6Br
+jr\
'O'
C H 3 ( C H 2 ) 6 ^ 0 - / ^ (CH2)6Br
CH 3 (CH 2 ) 6
2.3
-OEt
OEt
2.40
2.39
NaCN
Note the u s e of " C N as a s y n t h o n
for - C O 2 H .
HCI/H 2 O
J T \
(CH 2 ) 6 C0 2 H <3
CH^CHJs'^-o
CH3(CH2)6-^O
(CH2)6CN
2.41
2.42
2.5 Problems
l.
MeN
2.43
T r i c y c l i c p y r r o l e d e r i v a t i v e 2 . 4 3 is a d r u g c u r r e n t l y u n d e r d e v e l o p m e n t f o r
t h e t r e a t m e n t of s c h i z o p h r e n i a .
It is p r e p a r e d b y a K n o r r p y r r o l e s y n t h e s i s .
W h a t a r e t h e s t r u c t u r e s o f t h e t w o s t a r t i n g m a t e r i a l s r e q u i r e d , a n d that of t h e
intermediate enamine?
2.
W h y is p y r r o l e a l d e h y d e 2 . 4 4 l e s s r e a c t i v e to n u c l e o p h i l e s t h a n , s a y ,
b e n z a l d e h y d e ? W h y is p y r r o l e a l c o h o l 2 . 4 5 r e a d i l y p o l y m e r i s e d o n e x p o s u r e
to acid?
H
2.44
H
H
2.45
3.
N i t r a t i o n of f u r a n w i t h n i t r o n i u m t e t r a f l u o r o b o r a t e p r o d u c e s n i t r o f u r a n
2 . 3 3 d i r e c t l y . C o n t r a s t this r e s u l t to t h e t w o stage r e a c t i o n n e c e s s a r y w i t h
acetyl nitrate, p a g e 16. E x p l a i n t h e s e o b s e r v a t i o n s .
©
^
JO*^
Q
2.2
2.33
4. W h a t is the m e c h a n i s m of this r e a c t i o n ?
o
phC0C1
S'
2.2
,
ti
Ph
AlClj
y
o
2.6 References
Dean,
F.M.
(1982).
Adv.
heterocyclic
chem.,
30,
167; 3 1 ,
231
(furans).
G r o n o w i t z , S. (ed.) (1985).
chemistry
of heterocyclic
In Thiophene
compounds
and
its
derivatives
(The
[ed. A. W e i s s b u r g e r a n d E . C . T a y l o r ] ,
V o l . 44). W i l e y I n t e r s c i e n c e , N e w Y o r k .
Furniss, B.S., Hannaford, A.J., Smith, P.W.G, and Tatchell, A.R. (1989).
Vogel's
textbook
of practical
organic
chemistry
(5th e d n ) , p . 1 1 4 8
(preparation of p y r r o l e 2.16). L o n g m a n , H a r l o w .
Jackson, A.H. (1979).
I n Heterocyclic
( V o l . 4 of Comprehensive
organic
chemistry
chemistry,
(ed. P.G.
Sammes)
ed. D. Barton and W . D .
Ollis) (pyrroles). P e r g a m o n Press, O x f o r d .
J o n e s , R . A . ( e d . ) ( 1 9 9 0 ) . In Pyrroles
compounds
(The
chemistry
of
heterocyclic
[ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , V o l 48, Part 1). W i l e y
Interscience, N e w Y o r k .
J o n e s , R . A . a n d B e a n , G . P . ( 1 9 7 7 ) . The chemistry
of pyrroles.
Academic
Press, L o n d o n .
J o n e s , E . a n d M o o d i e , I . M . ( 1 9 7 0 ) . Org.
syn., 5 0 , 104 ( C 2 m e t a l l a t i o n of
thiophene).
K a t r i t z k y , A . R . a n d R e e s , C . W . (ed.) ( 1 9 8 4 ) . Comprehensive
chemistry,
Vol. 4, Part 3
heterocyclic
(five-membered rings with one heteroatom).
P e r g a m o n Press, O x f o r d .
M e t h - C o h n , O. ( 1 9 7 9 ) .
I n Heterocyclic
( V o l . 4 of Comprehensive
organic
chemistry
chemistry,
(ed. P . G .
Sammes)
ed. D. Barton and W . D .
Ollis), p . 7 3 7 ( t h i o p h e n e s ) . P e r g a m o n P r e s s , O x f o r d .
Sargent, M.V. (1979).
I n Heterocyclic
( V o l . 4 of Comprehensive
organic
chemistry
(ed. P.G.
chemistry,
Sammes)
ed. D. Barton
and
W . D . Ollis), p . 6 9 3 ( f u r a n s ) . P e r g a m o n P r e s s , O x f o r d .
Silverstein, R.M., Ryskiewicz, E.E., and Willard, C. (1963).
syntheses,
Coll. V o l . I V , 8 3 1 ( V i l s m e i e r f o r m y l a t i o n of p y r r o l e ) .
Organic
3. Oxazoles, imidazoles, and
thiazoles
3.1 Introduction
O x a z o l e 3 . 1 , i m i d a z o l e 3 . 2 , a n d t h i a z o l e 3 . 3 a r e t h e p a r e n t s t r u c t u r e s of a
related series of 1,3-azoles containing a nitrogen atom plus a second
h e t e r o a t o m in a f i v e - m e m b e r e d ring.
3.1
3.2
3.3
T h e y are isomeric with the 1,2-azoles isoxazole, pyrazole, and isothiazole
( s e e C h a p t e r 4 ) . T h e i r a r o m a t i c i t y d e r i v e s f r o m d e l o c a l i s a t i o n of a l o n e p a i r
f r o m the second heteroatom, 3.4a-e.
X=0,NH,S
T h e b i o s y n t h e s i s of h i s t a m i n e
i n v o l v e s d e c a r b o x y l a t i o n of t h e
a m i n o acid histidine.
T h e i m i d a z o l e r i n g o c c u r s n a t u r a l l y in h i s t a m i n e 3 . 5 , a n
important
m e d i a t o r of i n f l a m m a t i o n a n d g a s t r i c a c i d s e c r e t i o n . A q u a t e r n i s e d t h i a z o l e
r i n g is f o u n d in t h e e s s e n t i a l v i t a m i n t h i a m i n 3 . 6 . T h e r e a r e f e w n a t u r a l l y
occurring oxazoles, apart f r o m some secondary metabolites f r o m plant and
fungal sources.
H
H
3.5
3.6
Oxazole, imidazole, and thiazole can be formally derived f r o m furan,
pyrrole, and t h i o p h e n e respectively by r e p l a c e m e n t of a C H g r o u p b y a
n i t r o g e n a t o m at t h e 3 p o s i t i o n . T h e p r e s e n c e of this p y r i d i n e - l i k e n i t r o g e n
deactivates the 1,3-azoles towards electrophilic attack and increases their
s u s c e p t i b i l i t y t o w a r d s n u c l e o p h i l i c a t t a c k (see later). T h e s e 1 , 3 - a z o l e s c a n b e
v i e w e d as h y b r i d s b e t w e e n f u r a n , p y r r o l e , o r t h i o p h e n e , a n d p y r i d i n e .
I m i d a z o l e (p/T a =7.0) is m o r e b a s i c t h a n o x a z o l e ( p / f a = 0 . 8 ) o r t h i a z o l e
(p/sT a =2.5). T h i s increased basicity arises f r o m the greater electron-releasing
c a p a c i t y of t w o n i t r o g e n a t o m s r e l a t i v e to a c o m b i n a t i o n of n i t r o g e n and a
h e t e r o a t o m of h i g h e r e l e c t r o n e g a t i v i t y . A l s o n o t e that a s y m m e t r i c a l
resonance-stabilised cation 3 . 7 a , b is f o r m e d .
^
H
o H
0
© H
N'
N
1
0 .
H
H
3.7b
3.7a
F u r t h e r m o r e , certain s u b s t i t u t e d i m i d a z o l e s can exist in t w o t a u t o m e r i c
forms.
>3
N
H
N
H
3.8
F o r i n s t a n c e , t h e i m i d a z o l e s h o w n a b o v e e x i s t s as a r a p i d l y e q u i l i b r a t i n g
m i x t u r e of 4 - m e t h y l 3 . 8 a n d 5 - m e t h y l 3 . 9 t a u t o m e r s , and is r e f e r r e d to as
4 ( 5 ) - m e t h y l i m i d a z o l e . It m u s t a g a i n b e s t r e s s e d that t a u t o m e r i s a t i o n a n d
r e s o n a n c e are totally d i f f e r e n t . M e s o m e r i c r e p r e s e n t a t i o n s 3 . 7 a , b are n o t
interconverting like t a u t o m e r s 3.8 and 3.9; this is simply a m e a n s to describe
an intermediate hybrid structure.
3.2 Synthesis of oxazoles
R e t r o s y n t h e t i c c l e a v a g e of t h e c a r b o n - o x y g e n b o n d in g e n e r a l i s e d o x a z o l e
3.10 p r o d u c e s iminoalcohol 3 . 1 1 (better represented in the a m i d e f o r m
3 . 1 2 ) . S i m i l a r t a u t o m e r i s a t i o n o f t h e e n o l g r o u p g i v e s an a c t u a l
i n t e r m e d i a t e 3 . 1 3 , a n d d i s c o n n e c t i o n of t h e a m i d e l i n k a g e r e v e a l s
a m i n o k e t o n e 3 . 1 5 a n d an acylating species 3 . 1 4 such as an acid chloride. T h e
f o r w a r d p r o c e s s , c y c l o c o n d e n s a t i o n of a m i d e s 3 . 1 3 to yield oxazoles 3.10, is
k n o w n as the R o b i n s o n - G a b r i e l synthesis.
T h e statement that oxazole has a
p K a of 0.8 means that the
protonated form of oxazole is a
very strong acid.
Therefore oxazole (as the free
base) is a very w e a k base indeed.
O
R
"T
A,
NH,
N
Base
1
H
fV\
H,0
O O
3.15
3.13
r3
3.10
In p r a c t i c e t h e d e h y d r a t i o n c a n b e a c h i e v e d w i t h a b r o a d r a n g e of a c i d s o r
acid a n h y d r i d e s , s u c h as p h o s p h o r i c acid, p h o s p h o r u s o x y c h l o r i d e , p h o s g e n e
( C O C I 2 ) , a n d t h i o n y l c h l o r i d e . A n e x a m p l e of t h e m e c h a n i s m is s h o w n
b e l o w f o r t h i o n y l c h l o r i d e a n d i n v o l v e s a c t i v a t i o n of t h e a m i d e to i m i d o l y l
h a l i d e 3 . 1 6 t h e n i n t r a m o l e c u l a r a t t a c k b y t h e e n o l i c f o r m of t h e k e t o n e .
H
R^V
.X
R
o
ou
3
C1
R,
<0
3.13
so-'2
R .I
.R,
Y -
1
Cl
^
D
- HC1
Ri'
^ O H
*o"
3.10
3.16
T h e a m i n o k e t o n e s t h e m s e l v e s c a n b e p r e p a r e d b y a n u m b e r of m e t h o d s . A
t y p i c a l r o u t e is i l l u s t r a t e d b y t h e s y n t h e s i s of a n t i - i n f l a m m a t o r y d r u g 3 . 2 3 .
Ph,
P h X
Ph
Br,
0
3.17
Br
NaN,
Ph
N3
P t A o
Ph^O
3.18
3.19
H2/Pd
HC1
Ph.
. NH,.HC1
Ph^O
3.20
O
Et3N (2 eq.)
Cl'
Ph
Ph
^
Ph"
CO,Et
u
CO,H
2 HC1
Drugs which reduce inflammation
are often u s e d to treat the
s y m p t o m s of arthritis.
Ph-^Q
CO,Et
3.21
3.22
3.23
CO,Et
POC1,
l.NaOH
B r o m i n a t i o n of k e t o n e 3 . 1 7 g i v e s 3 . 1 8 w h i c h c a n b e c o n v e r t e d to a z i d e 3 . 1 9 .
Hydrogenation
of 3 . 1 9 in the p r e s e n c e of h y d r o c h l o r i c acid a f f o r d s
a m i n o k e t o n e h y d r o c h l o r i d e salt 3 . 2 0 . S u c h a m i n o k e t o n e s a r e o f t e n i s o l a t e d as
the corresponding
salts b e c a u s e the f r e e a m i n o k e t o n e s
are prone
to
dimerisation, having both nucleophilic and electrophilic centres. (For a
c o m m o n alternative p r e pa r a t i o n of a m i n o k e t o n e s , see the K n o r r pyrrole
s y n t h e s i s , C h a p t e r 2 . ) L i b e r a t i o n of t h e f r e e b a s e of 3 . 2 0 in t h e p r e s e n c e of
t h e a c i d c h l o r i d e a f f o r d s a m i d e 3 . 2 1 w h i c h is c y c l i s e d to o x a z o l e 3 . 2 2 . E s t e r
hydrolysis then affords the biologically-active carboxylic acid 3.23.
3.3 Synthesis of imidazoles
A l t h o u g h t h e r e a r e s e v e r a l w a y s o f p r e p a r i n g i m i d a z o l e s , t h e r e is n o o n e
o u t s t a n d i n g m e t h o d . O n e u s e f u l synthesis is the c o n d e n s a t i o n of a 1,2-
d i c a r b o n y l c o m p o u n d w i t h a m m o n i u m a c e t a t e a n d a n a l d e h y d e , as in t h e
p r e p a r a t i o n of i m i d a z o l e 3 . 2 5 .
MeO
A r e a s o n a b l e r a t i o n a l i s a t i o n is a c y c l o c o n d e n s a t i o n t y p e of p r o c e s s to g i v e
3.24 followed by irreversible tautomerisation to 3.25.
®
.
R
K
l
R
—
X x .
i
N
H
3.25
D,
R
1
R2
^^
H
H
w NH,
®
/ ^ H
o
/
H
1
2
V f v
RI
N ^ H
r - ^ N
*
H
3.24
3.4 Synthesis of thiazoSes
R e t r o s y n t h e t i c d i s c o n n e c t i o n of t h e n i t r o g e n - c a r b o n b o n d i n t h i a z o l e 3 . 2 6
l e a d s f o r m a l l y t o e n o l 3 . 2 7 w h i c h is e q u i v a l e n t to k e t o n e 3 . 2 8 . T h i s c a n b e
d e r i v e d f r o m h a l o k e t o n e 3 . 2 9 a n d t h i o a m i d e 3.30.
3.26
3.27
3.28
3.29
3.30
X=Cl,Br,I
T h e f o r w a r d p r o c e s s is t h e H a n t z s c h s y n t h e s i s of t h i a z o l e s w h i c h , d e s p i t e
its a n t i q u i t y (it is a r o u n d 1 0 0 y e a r s old), is still v e r y w i d e l y u s e d .
y°
CI
Y l
r
Heat
3.31
T h e m e c h a n i s m f o r t h e f o r m a t i o n of thiazole 3 . 3 1 i n v o l v e s
nucleophilic attack by sulphur followed by a cyclocondensation.
initial
Thiocarbonyl compounds are
much more nucleophilic than
carbonyl compounds because
of the lower electronegativity
of sulphur as compared to
oxygen.
T h e thioamides themselves are conveniently prepared f r o m the corresponding
amides by treatment with phosphorus (V) sulphide (see the P a a l — K n o r r
s y n t h e s i s o f t h i o p h e n e s , C h a p t e r 2 , f o r t h i s t y p e of c o n v e r s i o n ) . A v a r i a t i o n
o f t h e H a n t z s c h r e a c t i o n u t i l i s e s t h i o u r e a s , w h e r e R 3 in 3 . 3 0 i s a n i t r o g e n
a n d n o t a c a r b o n s u b s t i t u e n t . F o r i n s t a n c e , t h i o u r e a i t s e l f is u s e d i n t h e
p r e p a r a t i o n o f 2 - a m i n o t h i a z o l e s s u c h as 3 . 3 2 .
c f
S ^ N H
HEAT
2
%^NH
2
.HCI
S -
NH 2
3.32
3.5 EOectrophiNc substitution reactions of oxazoles,
imidazoles, and thiazoles
A s w i t h pyridine, not o n l y d o e s
the electronegative nitrogen atom
withdraw electron density from
t h e ring, but u n d e r t h e acidic
c o n d i t i o n s of m a n y electrophilic
reactions the a z o l e n i t r o g e n is
p r o t o n a t e d . T h e a z o l i u m c a t i o n is
relatively inert to further attack by
a positively charged electrophile.
T h e 1,3-azoles are not very reactive towards electrophilic attack due to the
deactivating effect of the pyridine-like nitrogen. However, electron-donating
g r o u p s c a n f a c i l i t a t e e l e c t r o p h i l i c a t t a c k , as in t h e p r e p a r a t i o n o f o x a z o l e s
3.34 and 3.35.
D i m e t h y l a m i n o o x a z o l e 3 . 3 3 is e s s e n t i a l l y f u n c t i o n i n g l i k e
a n e n a m i n e i n this r e a c t i o n .
fo
CF
3
o^
^O
cf3
^
Me 2 N. A .o K
ph
MejNji O
3.33
Me
N02
Ph
3.34
>
~ H
% ' ^ O M E
I kOMe
3.35
Imidazole can be nitrated under forcing conditions, nitration remarkably
occurring on the imidazolium cation 3.7a,b, giving nitroimidazole 3.36 after
l o s s of t w o p r o t o n s .
N02
E
©
N02
Hi
XJf
H,
P
©
/T%>
HNO3/h2so4
N
H
Heat
NO-2
H
3.7ab
—
H
C
//
A 3.36
N
H
_H
\
N02 ©
© H
N'
C1^
H
V N '
- H
'
i N/>
H
H
3.6 Anion chemistry of oxazoies, imidazoles, and
thiazoles
T h e C 2 p o s i t i o n of 1 , 3 - a z o l e s is p a r t i c u l a r l y e l e c t r o n - d e f i c i e n t b e c a u s e of t h e
e l e c t r o n - w i t h d r a w i n g e f f e c t of t h e a d j a c e n t h e t e r o a t o m s . T h e a c i d i t y of t h e
p r o t o n s at t h i s p o s i t i o n is s u c h t h a t d e p r o t o n a t i o n c a n b e a c h i e v e d w i t h
s t r o n g b a s e s to g i v e n u c l e o p h i l i c c a r b a n i o n s 3 . 3 7 w h i c h c a n b e q u e n c h e d
w i t h e l e c t r o p h i l e s p r o d u c i n g s u b s t i t u t e d 1,3-azoles 3 . 3 8 .
_I\\
" - B u L i t>
Li©
X
/ f x
3.37
3'38
x
E
S i m i l a r l y , a l k y l g r o u p s at t h e C 2 p o s i t i o n s ( b u t n o t t h e C 4 o r C 5 p o s i t i o n s )
can be deprotonated giving carbanions 3.39a,b which can also be quenched
w i t h e l e c t r o p h i l e s to a f f o r d 1 , 3 - a z o l e s 3 . 4 0 .
^
N©
-N
n-BuLi
w
Li
0
A
.
RO
X
X
X=0,NR,S
3.39b
3.39a
S o m e e x a m p l e s of b o t h t h e a b o v e t y p e s of r e a c t i v i t y are g i v e n b e l o w
Cv>
1. n-BuLi
CPh,
1. n-BuLi
2.CH3CHO
3.HC1 / H 2 0
1. n-BuLi ^
2.Ph2CO
3.HC1/H 2 0
S
v\
N
^
Me
V N
// \\
»-B"Li„
2.PhCHO
3.HC1/H 2 0
1. »-BuLi ^
2.Mel
"
H
> . vO H
OH
f t
r
Ph
Ph
a
0H
^Ph
Me
\ - N
/ / \\
S
^
^
1
o®
RO A ,
Ph
Ph
T h e r e is a useful a n a l o g y
between resonance-stabilised
a n i o n 3.39a,b a n d an ester
enolate anion. Note that in both
c a s e s the negative c h a r g e c a n be
d e l o c a l i s e d onto a h e t e r o a t o m .
©
3.7 Nucleophilic aromatic substitution of oxazoles,
imidazoles, and thiazoles
W e h a v e p r e v i o u s l y d i s c u s s e d t h e r e d u c e d r e a c t i v i t y t o e l e c t r o p h i l e s of
o x a z o l e , i m i d a z o l e , and thiazole, as c o m p a r e d to f u r a n , p y r r o l e ,
and
t h i o p h e n e , w h i c h r e s u l t s f r o m t h e p r e s e n c e of t h e p y r i d i n e - l i k e n i t r o g e n
a t o m . T h i s b e h a v i o u r is p a r a l l e l e d b y i n c r e a s e d r e a c t i v i t y to n u c l e o p h i l e s .
Nucleophilic attack on furan, pyrrole, and thiophene derivatives only occurs
w h e n a n a d d i t i o n a l a c t i v a t i n g g r o u p is p r e s e n t , as in t h e
displacement
reaction giving thiophene 3.41.
H
^
N
R
N
Br
^N
• HBr
>
©N
I
©O
e,
O n c e a g a i n this reactivity parallels
c e r t a i n f e a t u r e s of c a r b o n y l
c h e m i s t r y . C o m p a r e t h e reaction of
aniline w i t h c h l o r o f o r m a t e s , b e l o w .
w h i c h a l s o illustrates t h e f a c t that i m i d a z o l e is a g o o d n u c l e o p h i l e . H o w e v e r ,
n u c l e o p h i l e s , as s h o w n b y t h e p r e p a r a t i o n s of 3 . 4 2 a n d 3 . 4 3 .
Ph
A
Ph
Ph
Ph
NHPh
3.42
C x OMe
3.43
H 2 NPh
f \
S^j
NPh
H
©
• CI
N J.
-3s
O
NHPh
Heat
1 1
I H 2 NPh
RO
S'
3.41
n o a c t i v a t i o n is n e c e s s a r y w i t h 2 - h a l o - 1 , 3 - a z o l e s , w h i c h c a n r e a c t w i t h
P h ^ o ^ C l
O
I ^
NO 2
T h e n i t r o g r o u p p l a y s a k e y r o l e as a n e l e c t r o n - a c c e p t o r in t h i s r e a c t i o n ,
Ph
RO^Cl
^S
Br
^L
-Br
OMe
OMe
3.8 Problems
1. S u g g e s t a s y n t h e s i s o f o x a z o l e 3 . 3 3 .
A
Me2N'^o
Ph
3.33
2. A l e s s g e n e r a l s y n t h e s i s of o x a z o l e s is t h e c o n d e n s a t i o n of b r o m o k e t o n e s
w i t h a m i d e s . W h a t is t h e m e c h a n i s m f o r t h e f o r m a t i o n of o x a z o l e 3 . 4 4 ? H o w
does 3.44 relate to the oxazole w h i c h might b e prepared f r o m the s a m e
bromoketone
by
conversion
to
formylation, and cyclocondensation?
O
the
corresponding
aminoketone,
N-
3.
C a r b o x y l i c acid 3 . 4 6 h a s b e e n e x t e n s i v e l y u s e d in t h e p r e p a r a t i o n of
s e m i - s y n t h e t i c p e n i c i l l i n s a n d c e p h a l o s p o r i n s . D e v i s e a s y n t h e s i s of 3 . 4 6
f r o m ester 3 . 4 5 .
3.45
3.46
3.9 References
C a m p b e l l , M . M . ( 1 9 7 9 ) . In Heterocyclic
chemistry
(ed. P . G .
Sammes)
(Vol. 4 of Comprehensive
organic
chemistry,
ed. D. Barton and W . D .
Ollis), p. 9 6 2 (oxazoles) and p. 9 6 7 (thiazoles). P e r g a m o n Press, O x f o r d .
Furniss, B . S . , H a n n a f o r d , A.J., S m i t h , P . W . G . , a n d T a t c h e l l , A . R . (1989).
Vogel's
textbook
of practical
organic
chemistry
( 5 t h e d n ) , p. 1 1 5 3
(preparation of a m i n o t h i a z o l e 3.32). L o n g m a n , H a r l o w .
G r i m m e t t , M . R . ( 1 9 7 0 ) . Adv. heterocyclic
chem., 1 2 , 103 ( i m i d a z o l e s ) .
G r i m m e t t , M . R . ( 1 9 7 9 ) . In Heterocyclic
chemistry
(ed. P . G . S a m m e s )
(Vol. 4 of Comprehensive
organic
chemistry,
ed. D . B a r t o n a n d W . D .
Ollis), p . 3 5 7 (imidazoles). P e r g a m o n Press, O x f o r d .
G r i m m e t t , M . R . ( 1 9 8 0 ) . Adv. heterocyclic
chem., 2 7 , 2 4 1 ( i m i d a z o l e s ) .
L a k h a n , R . a n d T e r n a i , B . ( 1 9 7 4 ) . Adv.
heterocyclic
chem., 1 7 , 9 9
(oxazoles).
M e t z g e r , J . V . ( 1 9 7 9 ) . In Thiazole
and its derivatives
(The chemistry
of
heterocyclic
compounds
[ed. A . W e i s s b u r g e r and E.C. T a y l o r ] , V o l . 34).
W i l e y Interscience, N e w York.
Turchi, I.J. (1986). In Oxazoles
{The chemistry
of heterocyclic
compounds
[ed. A . W e i s s b u r g e r and E . C . Taylor], V o l . 45). W i l e y Interscience, N e w
York.
T u r c h i , I.J. a n d D e w a r , M . J . S . ( 1 9 7 5 ) .
Chem.
rev. , 7 5 , 3 8 9 ( o x a z o l e s ) .
4. Isoxazoles, pyrazoles, and
isothiazoles
I s o x a z o l e 4.1, p y r a z o l e 4.2, and isothiazole 4 . 3 are the parent structures of
t h e 1 , 2 - a z o l e f a m i l y of h e t e r o c y c l e s , h a v i n g a n i t r o g e n a t o m p l u s o n e o t h e r
h e t e r o a t o m in a 1 , 2 - r e l a t i o n s h i p in a f i v e - m e m b e r e d r i n g .
4
5
3
ft
o
4
)N
2
3
4
5 FT VL 2
5
N
1
3
ft
x
V
s
2
1
H
4.1
;
4.2
4.3
T h e a r o m a t i c s e x t e t is c o m p l e t e d b y d e l o c a l i s a t i o n of t h e l o n e p a i r f r o m t h e
s e c o n d h e t e r o a t o m , 4 . 4 a - e . C o n s e q u e n t l y , as in p y r i d i n e , t h e n i t r o g e n a t o m s
of t h e 1 , 2 - a z o l e s h a v e a l o n e p a i r a v a i l a b l e f o r p r o t o n a t i o n . H o w e v e r t h e 1,2a z o l e s a r e s i g n i f i c a n t l y l e s s b a s i c t h a n t h e 1 , 3 - a z o l e s b e c a u s e of t h e e l e c t r o n w i t h d r a w i n g e f f e c t of t h e a d j a c e n t h e t e r o a t o m . I s o x a z o l e a n d i s o t h i a z o l e a r e
e s s e n t i a l l y n o n - b a s i c h e t e r o c y c l e s (pA" a s < 0 ) , a n d e v e n p y r a z o l e (pAT a =2.5) is
a m u c h w e a k e r b a s e than t h e c o r r e s p o n d i n g 1,3-azole i m i d a z o l e (pA' a =7).
—>
?n
X
X
4.4a
—
—
C
?
n
—>
0
ffi
X
©
X
4.4b
4.4c
4.4d
ffi
e
x
G
©
4.4e
X = 0,NH,S
A s w i t h s u b s t i t u t e d i m i d a z o l e s , s u b s t i t u t e d p y r a z o l e s m a y e x i s t as a
m i x t u r e of t a u t o m e r s . 5 - M e t h y l p y r a z o l e 4 . 5 a n d 3 - m e t h y l p y r a z o l e 4 . 6 e x i s t
as a r a p i d l y e q u i l i b r a t i n g m i x t u r e in s o l u t i o n .
A
3
5// v
N
H1
4.5
4
^
5
34
N
2
4.6
A l t h o u g h t h e r e a r e a f e w e x a m p l e s of n a t u r a l l y - o c c u r r i n g 1,2-azoles, m a n y
totally s y n t h e t i c d e r i v a t i v e s h a v e f o u n d p h a r m a c e u t i c a l a p p l i c a t i o n .
4.1 Synthesis of isoxazoles and pyrazoles
R e t r o s y n t h e t i c d i s c o n n e c t i o n of g e n e r a l i s e d 1 , 2 - a z o l e 4 . 7 g i v e s initially 4 . 8
w h i c h w o u l d e x i s t as k e t o n e 4 . 9 . T h i s in t u r n is c l e a r l y d e r i v e d f r o m 1,3diketone 4.10.
R,
*3
R2
R3
x
N
RI
r
Ri
XH
O
"OH XH
X=0,NH,S
T h i s a n a l y s i s s u g g e s t s t h a t c o n d e n s a t i o n of 4 . 1 0 w i t h h y d r o x y l a m i n e 4 . 1 1 ,
hydrazine 4.12, or thiohydroxylamine 4 . 1 3 should give the corresponding
1,2-azole. . This a p p r o a c h represents an important route to isoxazoles and
p y r a z o l e s , b u t t h i o h y d r o x y l a m i n e 4 . 1 3 , a l t h o u g h k n o w n , is f a r t o o u n s t a b l e
f o r s y n t h e t i c p u r p o s e s . T h e s y n t h e s i s of i s o t h i a z o l e s will b e m e n t i o n e d later.
T h e m e c h a n i s m of t h e f o r w a r d p r o c e s s is i l l u s t r a t e d b y t h e p r e p a r a t i o n of
i s o x a z o l e 4 . 1 4 a n d is s i m p l y t w o c o n s e c u t i v e c o n d e n s a t i o n s .
O
O V
O
(OH
-H,o
O
O
4.11
H 2 NOH
4.12
H 2 NNH 2
4.13
H 2 NSH
In practice h y d r o x y l a m i n e a n d
h y d r a z i n e are v e r y reactive
nucleophiles, far m o r e so t h a n
might be e x p e c t e d f r o m
c o n s i d e r a t i o n of s i m p l e p h y s i c a l
parameters. The inceased
nucleophilicity of a h e t e r o a t o m
w h e n b o n d e d to a s e c o n d
h e r e o a t o m is k n o w n as t h e a
effect. For a theoretical
rationalisation of the a effect in
t e r m s of frontier obitals s e e
Fleming, 1976.
•H,0
- M s H'NVOH
H,N-OH
/
4.10
4.9
4.8
4.7
*
k2
V'
4.14
H
N o t e that if h y d r o x y l a m i n e o r a s u b s t i t u t e d h y d r a z i n e is c o n d e n s e d w i t h a n
unsymmetrical
diketone
( 4 . 1 0 , w h e r e R] a n d R 3 a r e d i f f e r e n t ) t h e n a
r e g i o i s o m e r i c m i x t u r e of i s o x a z o l e s o r p y r a z o l e s m a y r e s u l t . H o w e v e r a
s i n g l e r e g i o i s o m e r m a y p r e d o m i n a t e w h e r e t h e r e is an i n h e r e n t bias,
o
o
R2
H,NOH
r,
Y
Rl
R2
o
o
R3
H
O
r„
R,
o
N
R,
N'
R2
H,NNHR
l
f/
S
vN
N'
R
+
^
N
,NR
F o r i n s t a n c e , t h e p r e p a r a t i o n of i s o x a z o l e 4 . 1 7 is v i r t u a l l y r e g i o s p e c i f i c
b e c a u s e t h e r e a c t i o n c o m m e n c e s w i t h t h e m o r e n u c l e o p h i l i c h e t e r o a t o m (i.e.
nitrogen) attacking the m o r e electrophilic ketone (activated by the electronw i t h d r a w i n g i n d u c t i v e e f f e c t of t h e a d j a c e n t e s t e r g r o u p ) . T h e r e a d e r is
e n c o u r a g e d to c o n s i d e r t h e r e g i o c h e m i c a l b i a s in t h e p r e p a r a t i o n of i s o x a z o l e
4.15 and pyrazole 4.16.
T h e g e n e r a l reactions of H 2 N O H
a n d H 2 N N H R with u n s y m m e t r i c a l
d i k e t o n e s a r e s h o w n here.
4.16
HO.
H,NOH
C0 2 Et
CO,Et
N
C0 2 Et
-H20
-H20
h7 VN
o'
4.17
The other important isoxazole synthesis involves the concerted
[3+2]
cycloaddition reaction of nitrile oxides 4 . 1 8 with either alkynes 4 . 1 9 or
alkyne equivalents 4.20.
X=0Ac,NMe 2 ,N0 2
A w i d e r a n g e of n i t r i l e o x i d e s is k n o w n ( R 3 = H , aryl, a l k y l , e s t e r , h a l i d e ,
etc). T h e m e t h o d of c h o i c e f o r t h e p r e p a r a t i o n of s i m p l e n i t r i l e o x i d e s ( R 3 =
a l k y l , aryl) is o x i d a t i o n of t h e c o r r e s p o n d i n g o x i m e :
o
11
A
R3-^H
N H2NOH
H20
•
R
X
^H
3
o h
(-2H)
© e
R3—= N - O
4.18
Several
oxidising
agents
can
be
used
(lead
tetraacetate,
N-
b r o m o s u c c i n i m i d e , c h l o r i n e , e t c ) . A m e c h a n i s m is i l l u s t r a t e d b e l o w f o r
alkaline sodium hypochlorite.
ft
N'
N'
3
CI-OH
f
OH
N
©
Wn
CI
*3
©
-EEN - O
^
4.18
L e t us n o w c o n s i d e r the synthesis of isoxazole 4 . 2 8 , a drug f o r the
t r e a t m e n t of b r o n c h i a l a s t h m a . T h e m o s t d i r e c t p r e p a r a t i o n of i s o x a z o l y l
ketone 4.24
is t h e c y c l o a d d i t i o n of u n s t a b l e b r o m o n i t r i l e o x i d e
4.22
( p r e p a r e d in situ b y d e h y d r o b r o m i n a t i o n of 4 . 2 1 ) w i t h a c e t y l e n i c k e t o n e 4 . 2 3 .
O b s e r v e the regioselectivity of this reaction. Both electron-donating and
electron-withdrawing
groups
on
the
acetylenic
components
in
such
c y c l o a d d i t i o n s t e n d to o c c u r at t h e C 5 p o s i t i o n in t h e f i n a l i s o x a z o l e a n d n o t
at C 4 . B r o m i n a t i o n of k e t o n e 4 . 2 4 a f f o r d s b r o m o k e t o n e 4 . 2 5 w h i c h is
4.23
Br
Y
N
Br
n
Br
K2C°3
Br
Br.
//
ill
//
Br,
N
NFFI
OH
Oft
©
4.21
4.24
O
Br
4.25
4.22
NaBH4 .
Br.
HjN-f-Bu
<
NH-«-Bu
//
N
s
4.28
NaH
O
4.27
r e d u c e d w i t h s o d i u m b o r o h y d r i d e to g i v e b r o m o h y d r i n 4.26. T r e a t m e n t w i t h a
strong base p r o d u c e s e p o x i d e 4 . 2 7 via the intermediate alkoxide,
and
n u c l e o p h i l i c o p e n i n g of this e p o x i d e at t h e least s t e r i c a l l y h i n d e r e d p o s i t i o n
a f f o r d s t h e t a r g e t d r u g 4.28.
4.2 Synthesis of isothiazoles
Isothiazoles are usually p r e p a r e d by routes involving f o r m a t i o n of the
n i t r o g e n - s u l p h u r b o n d in t h e c y c l i s a t i o n s t e p . T h i s is o f t e n s e t u p b y
o x i d a t i o n of t h e s u l p h u r a t o m , as in t h e c o n v e r s i o n of t h i o a m i d e 4 . 2 9 t o
isothiazole 4.30.
NH,
4.29
N
N
'/
\\
4.30
NH 2
I
.
Cl-NH,
\
H
€1. H
Q
N
C1NH,
®s~c>
N,
-H
^N
S
— HC1
tS
N
NH
4.3 Electrophilic substitution of isoxazoles,
pyrazoles, and isothiazoles
T h e p r e s e n c e of a p y r i d i n e - l i k e nitrogen in the 1,2-azoles m a k e s t h e m
markedly less reactive towards electrophilic substitution than f u r a n , pyrrole,
and thiophene. (The same effect was noted for the 1,3-azoles in Chapter 3.)
Nevertheless, electrophilic substitution is k n o w n in 1,2-azoles, occurring
principally at the C 4 position. This selectivity is reminiscent of p y r i d i n e
chemistry w h e r e the position meta to the electronegative nitrogen a t o m is
the 'least deactivated' (see Chapter 5).
®r
B^-Br
Br
H
-J^
I O'W
PN
Cy©
i j > '
N
Br
^Br^-Br
If
Br
—" V
^
Nitration and sulphonation of 1,2-azoles under vigorous conditions are
also k n o w n , as in the preparation of 4-nitropyrazole 4 . 3 1 .
NO2
J ° 2
o
N
H
- 3-36
See the related preparation of
nitroimidazole 3.36.
n
^
cv
N'
H
NR
H
H
^
-H
NO 2
rv
T\
©N'
H
V
431
H
A s w e h a v e s e e n with o t h e r e l e c t r o n - d e f i c i e n t h e t e r o c y c l e s , the
i n t r o d u c t i o n of an e l e c t r o n - d o n a t i n g g r o u p p r o m o t e s e l e c t r o p h i l i c
substitution, as in the facile bromination of aminoisothiazole 4 . 3 2 .
Br—B
5
?r
Ph
^
NH 2
S'
4.32
Ph
,*M ^
H Z N
V
S
'
Br
NH
2
H
Ph
s'
4.4 Anion chemistry of isoxazoles, pyrazoles, and
isothiazoles
Isothiazoles and nitrogen-blocked pyrazoles can be deprotonated at the C5
position with alkyl lithium reagents, and the resultant carbanions q u e n c h e d
with a wide range of electrophiles, as in the preparation of 4.33 and 4.34.
\\
N
n-BuLi
H-BuLi
N
N
ffi
Li
•
• Li
//
\\
N
e(*
©
(C0 2 Et) 2
s'
eti.
1
Mel
N
N'
'N'
Ph
Ph
4,3
O
4.34
Ph
This useful m e t h o d o l o g y (complementary to the C 4 selectivity of normal
e l e c t r o p h i l i c s u b s t i t u t i o n ) is n o t a p p l i c a b l e t o i s o x a z o l e c h e m i s t r y b e c a u s e
the i n t e r m e d i a t e a n i o n s ( s u c h as 4 . 3 5 ) a r e r a t h e r u n s t a b l e a n d d e c o m p o s e via
oxygen-nitrogen cleavage.
Ph
Ph
Ph
V
»- B "Li » Li
\N
Ph
Ph
y /
vN
+
PhCN
o
°©
4.35
Li
H o w e v e r , a l k y l g r o u p s at the C 5 p o s i t i o n of i s o x a z o l e s c a n b e d e p r o t o n a t e d
a n d r e a c t e d w i t h electrophiles.
'/ VN
Note the analogy to the anions
derived f r o m crotonate esters.
n-BuLi
Li
©
OR
D i m e t h y l i s o x a z o l e 4 . 1 4 c a n b e s e l e c t i v e l y d e p r o t o n a t e d at t h e C 5 m e t h y l
group, nearer the more electronegative oxygen atom. Although
simple
©
d e p r o t o n a t i o n c a n n o t a f f o r d a n e n t r y i n t o C 4 s u b s t i t u t i o n in this s y s t e m , it is
p o s s i b l e t o g e n e r a t e a c a r b a n i o n at t h e C 4 p o s i t i o n in a r o u n d a b o u t f a s h i o n .
B r o m i n a t i o n of 4 . 1 4 a f f o r d s t h e C 4 - f u n c t i o n a l i s e d i s o x a z o l e 4 . 3 6 . M e t a l -
OR
h a l o g e n e x c h a n g e \fcith n - b u t y l l i t h i u m at l o w t e m p e r a t u r e ( - 7 8 ° C ) g e n e r a t e s
c a r b a n i o n 4 . 3 7 w h i c h c a n b e q u e n c h e d w i t h e l e c t r o p h i l e s to g i v e i s o x a z o l e s
s u c h as 4 . 3 8 .
O
N©
©
Interestingly, 1, 3, 5 - t r i m e t h y l
p y r a z o l e is d e p r o t o n a t e d o n the
N - m e t h y l g r o u p , facilitating
reaction w i t h electrophiles at this
position.
ICO,
2.HC1
// WN
N
4.5 Problems
Me
n-BuLi
— 78°C
1. W h a t is t h e m e c h a n i s m f o r t h e f o r m a t i o n of i s o t h i a z o l o n e 4 . 3 9 ?
o
NH,
NEC — S
,0
K»
EtOH / H 2 0
S
4.39
N"
Mel
NH
N'
Et
N
I ©
a
CH 2 Li
2. W h a t g e n e r a l strategy m i g h t b e e m p l o y e d to c o n v e r t p y r a z o l e to a l c o h o l
4 . 4 0 , a p o t e n t i n h i b i t o r of steroid b i o s y n t h e s i s .
Q
H O ^ - 1
^
H
Ar
Ar
^
4 4 (
H
3. W h a t is the p r o d u c t resulting f r o m oxidation of 4.41?
NaOCl
NaOH
9
4.41
4. A s y n t h e s i s of 2 - c y a n o c y c l o h e x a n o n e 4 . 4 5 f r o m c y c l o h e x a n o n e is s h o w n
b e l o w . F o r m y l a t i o n of c y c l o h e x a n o n e p r o d u c e s a m i x t u r e of k e t o / e n o l
t a u t o m e r s 4 . 4 2 a n d 4 . 4 3 , the e q u i l i b r i u m lying to the side of the e n o l 4 . 4 2 .
T r e a t m e n t with h y d r o x y l a m i n e affords isoxazole 4.44, and base-induced
f r a g m e n t a t i o n of t h e i s o x a z o l e ring a f f o r d s 4.45. E x p l a i n the regioselectivity
of t h e i s o x a z o l e f o r m a t i o n , and the m e c h a n i s m of the f r a g m e n t a t i o n p r o c e s s .
°
o
n r 0 " ^ cx^'^o*
NaOEt
4.42
—
V A
4.44
4.43
°
>
• rr"
2.NH4Q / H 2 0
4.45
4.6 References
Campbell, M.M.
(1979).
In Heterocyclic
( V o l . 4 of Comprehensive
organic
chemistry
chemistry,
(ed. P . G . S a m m e s )
ed. D. Barton and W . D .
O l l i s ) , p . 9 9 3 ( i s o x a z o l e s ) a n d p. 1 0 0 9 ( i s o t h i a z o l e s ) .
Pergamon Press,
Oxford.
F l e m i n g , I. ( 1 9 7 6 ) .
Frontier
orbitals
and
organic
chemical
reactions,
p.77. Wiley, Chichester.
Furniss,
B.S.,
(1989).
Hannaford,
In Vogel's
A.J.,
textbook
p. 1149 ( p r e p a r a t i o n
Smith,
P.W.G.,
of practical
organic
of 3 , 5 - d i m e t h y l p y r a z o l e ) .
G r i m m e t t , M . R . ( 1 9 7 9 ) . In Heterocyclic
( V o l . 4 of Comprehensive
organic
chemistry
chemistry,
and Tatchell,
chemistry
A.R.
(5th e d n ) ,
Longman,
Harlow.
(ed. P . G .
Sammes)
e d . D. B a r t o n a n d W . D .
Ollis), p . 3 5 7 ( p y r a z o l e s ) . P e r g a m o n P r e s s , O x f o r d .
Kochetkov, N . K . and Sokolov, S.D.
(1963).
Adv.
heterocyclic
chem.,
2,
3 6 5 (isoxazoles).
Kost, A.N.
a n d G r a n d b e r g , I.I.
(1966).
Adv.
heterocyclic
chem.,
6, 3 4 7
chem.,
4, 107
(pyrazoles).
Slack, R. and Wooldrige, K.R.H.
( 1 9 6 5 ) . Adv.
heterocyclic
(isothiazoles).
W a k e f i e l d , B.J. a n d W r i g h t , D . J .
147 (isoxazoles).
(1979).
Adv.
heterocyclic
chem.,
25,
5. Pyridines
5.1 Introduction
P y r i d i n e 5 . 1 is a p o l a r liquid (b.p. 1 1 5 ° C ) w h i c h is m i s c i b l e w i t h b o t h
o r g a n i c s o l v e n t s a n d w a t e r . It c a n f o r m a l l y b e d e r i v e d f r o m b e n z e n e b y
r e p l a c e m e n t of a C H g r o u p b y a nitrogen atom. P y r i d i n e is a highly aromatic
h e t e r o c y c l e , b u t t h e e f f e c t of t h e h e t e r o a t o m m a k e s its c h e m i s t r y q u i t e
distinct f r o m that of b e n z e n e . T h e a r o m a t i c sextet of six n e l e c t r o n s is
c o m p l e t e w i t h o u t i n v o k i n g p a r t i c i p a t i o n of the l o n e pair o n the nitrogen.
T h i s is in direct contrast with the situation in p y r r o l e ( C h a p t e r 2) w h e r e the
a r o m a t i c sextet includes the lone p a i r on the nitrogen. H e n c e the lone pair of
p y r i d i n e is a v a i l a b l e f o r b o n d i n g w i t h o u t d i s t u r b i n g the a r o m a t i c i t y of the
ring. P y r i d i n e is m o d e r a t e l y b a s i c (p/T a =5.2) and can be q u a t e r n i s e d with
a l k y l a t i n g a g e n t s to f o r m p y r i d i n i u m s a l t s 5 . 2 . P y r i d i n e a l s o f o r m s
c o m p l e x e s with L e w i s acids such as s u l p h u r trioxide. T h i s c o m p l e x 5 . 3 is a
mild source of sulphur trioxide f o r sulphonation reactions (see C h a p t e r 2).
R-X
x
©N
I
H
SO,
N'
•T
e
5.2
R
5.3
SO©
T h e e f f e c t of the h e t e r o a t o m is to m a k e t h e p y r i d i n e ring very u n r e a c t i v e
to n o r m a l e l e c t r o p h i l i c a r o m a t i c s u b s t i t u t i o n . C o n v e r s e l y , p y r i d i n e s are
susceptible to nucleophilic attack. T h e s e topics are discussed later.
5.2 Synthesis of pyridines
O u r r e t r o s y n t h e t i c analysis of g e n e r a l i s e d p y r i d i n e 5 . 4 c o m m e n c e s with an
a d j u s t m e n t of t h e o x i d a t i o n l e v e l to p r o d u c e d i h y d r o p y r i d i n e 5 . 5 . T h i s
m o l e c u l e c a n n o w b e d i s c o n n e c t e d very r e a d i l y . C l e a v a g e of t h e c a r b o n h e t e r o a t o m b o n d s in the usual w a y leaves dienol 5.6 w h i c h exists as diketone
5.7. T h e 1,5-dicarbonyl relationship can be d e r i v e d f r o m a M i c h a e l reaction
of k e t o n e 5 . 8 a n d e n o n e 5 . 9 , w h i c h in turn can arise f r o m c o n d e n s a t i o n of
aldehyde 5 . 1 0 and ketone 5.11.
T h e s e p r o c e s s e s are facilitated w h e n R 2 and R 4 are e l e c t r o n - w i t h d r a w i n g
g r o u p s such as esters. F u r t h e r m o r e , w h e n k e t o n e s 5.11 and 5 . 8 are the s a m e ,
w e h a v e the basis f o r the classical H a n t z s c h pyridine svnthesis.
5.12
S.13
F o r i n s t a n c e , c o n d e n s a t i o n of ethyl a c e t o a c e t a t e , f o r m a l d e h y d e , a n d
a m m o n i a gives dihydropyridine 5 . 1 2 which is readily oxidised with nitric acid
to g i v e p y r i d i n e 5 . 1 3 . A l t h o u g h t h e p r e c i s e details of this m u l t i c o m p o n e n t
c o n d e n s a t i o n are not k n o w n , a reasonable p a t h w a y is s h o w n below,
o
Note that in this example R 2 and
R4 are ethyl esters, so the
adjacent carbon is actually an
active methylene group. T h e
higher acidity a n d hence
nucleophilicity of these cent
facilitates the reaction sequence.
N
H
5.12
HO.
HS
S o m e e x a m p l e s of dihydropyridines p r e p a r e d in this w a y are s h o w n b e l o w .
(The student is encouraged to work out the aldehydes used in each case.)
N ^
N
A s well as b e i n g i n t e r m e d i a t e s f o r t h e s y n t h e s i s of p y r i d i n e s , t h e s e
d i h y d r o p y r i d i n e s are t h e m s e l v e s an i m p o r t a n t class of h e t e r o c y c l e s . F o r
instance, d i h y d r o p y r i d i n e 5 . 1 4 is a d r u g f o r l o w e r i n g b l o o d pressure. In the
s y n t h e s i s of 5 . 1 4 n o t e that c a r r y i n g o u t t h e H a n t z s c h s y n t h e s i s s t e p w i s e
allows f o r the preparation of an u n s y m m e t r i c a l d i h y d r o p y r i d i n e , h a v i n g both
a m e t h y l and an ethyl ester.
H ^O
piperidine
-H,0
gtQ
N
H
5.14
5.3 Electrophilic substitution of pyridines
P y r i d i n e is virtually inert t o a r o m a t i c e l e c t r o p h i l i c s u b s t i t u t i o n . C o n s i d e r
nitration of p y r i d i n e b y nitric acid. First, as p y r i d i n e is a m o d e r a t e b a s e , it
will b e a l m o s t c o m p l e t e l y p r o t o n a t e d b y t h e acid, m a k i n g it m u c h less
susceptible to electrophilic attack. S e c o n d , addition of the electrophile to the
s m a l l a m o u n t of u n p r o t o n a t e d p y r i d i n e p r e s e n t in s o l u t i o n is n o t a f a c i l e
process.
A t t a c k o f t h e e l e c t r o p h i l e at t h e C 2 o r C 4 p o s i t i o n r e s u l t s in an
intermediate cation with partial positive charge on the electronegative
n i t r o g e n a t o m . T h i s is clearly n o t energetically f a v o u r a b l e w h e n c o m p a r e d to
C 3 substitution, w h e r e n o partial positive c h a r g e resides o n nitrogen. In f a c t
t h e p r o d u c t of C 3 substitution, n i t r o p y r i d i n e 5 . 1 5 , can b e isolated f r o m the
e x h a u s t i v e nitration of pyridine, but only in p o o r yield.
A consequence of the asymmetry
of 5.14 is that C 4 is a stereogenic
centre. Hence the product is formed
as a racemic mixture.
NO,
N
NO,
>
<
<
NO,
•
"N
H
H
N02
X
H
N02
NO,
H
H
,N°2
H
<
C-alkylation of a stericallyhindered phenolate anion.
C3-attack
N
N
H
C2-attack
,N02
>-
C4-attack
A l t h o u g h b e t t e r r e s u l t s h a v e b e e n a c h i e v e d w i t h t h e s u l p h o n a t i o n of
p y r i d i n e to g i v e t h e s u l p h o n i c a c i d 5 . 1 6 , e l e c t r o p h i l i c s u b s t i t u t i o n s o n an
i n a c t i v a t e d p y r i d i n e r i n g a r e in g e n e r a l n o t p r e p a r a t i v e l y u s e f u l .
NO,
^ / S 0
3
H
N
N
5.16
5.15
P y r i d i n e c a n b e a c t i v a t e d t o e l e c t r o p h i l i c s u b s t i t u t i o n b y c o n v e r s i o n to
p y r i d i n e N - o x i d e 5 . 1 7 . A t f i r s t s i g h t it is c u r i o u s to c o n s i d e r o x i d a t i o n (i.e.
e l e c t r o n l o s s ) as a m e a n s of a c t i v a t i n g a s y s t e m to e l e c t r o p h i l i c s u b s t i t u t i o n ,
b u t 5 . 1 7 c a n act r a t h e r l i k e a s t e r i c a l l y - h i n d e r e d p h e n o l a t e a n i o n t o w a r d s
electrophiles, producing i n t e r m e d i a t e 5.18 which then loses a p r o t o n to
give s u b s t i t u t e d N - o x i d e 5 . 1 9 . F o r t h i s m e t h o d o l o g y t o b e u s e f u l it is of
course necessary to r e m o v e the activating oxygen atom. This can b e d o n e
with phosphorus trichloride, which becomes oxidised to
phosphorus
oxychloride.
[O]
N
o
5.17
e
OH
-H
5.17
5.19
5.18
- POCl,
5.19
PCI,
F o r i n s t a n c e , 4 - n i t r o p y r i d i n e 5 . 2 0 can be p r e p a r e d f r o m p y r i d i n e in three
steps b y this m e t h o d o l o g y .
5.17
oe
oe
5.20
P y r i d i n e N - o x i d e s c a n a l s o b e c o n v e r t e d into s y n t h e t i c a l l y u s e f u l 2 chloropyridines 5.21 (see later).
A n o t h e r a p p r o a c h to electrophilic substitution i n v o l v e s t h e c h e m i s t r y of
2 - p y r i d o n e 5 . 2 2 and 4 - p y r i d o n e 5.23. T h e s e are the t a u t o m e r i c f o r m s of 2 <3y\ t j
and 4 - h y d r o x y p y r i d i n e r e s p e c t i v e l y . T h e y exist exclusively in the p y r i d o n e
f o r m , the h y d r o g e n a t o m b e i n g a t t a c h e d to the n i t r o g e n a t o m , n o t the
o x y g e n . T h e i r electronic structures are not a d e q u a t e l y described b y a single
v a l e n c e representation, the l o n e pair f r o m the nitrogen a t o m being delocalised
t o a c o n s i d e r a b l e e x t e n t o n t o t h e o x y g e n a t o m , as in m e s o m e r i c
representations 5.22a and 5.23a.
OH
o
B o t h p y r i d o n e s can react with electrophiles at positions ortho a n d para to
the activating oxygen atom. For instance, 4-pyridone reacts with
e l e c t r o p h i l e s at t h e C 3 p o s i t i o n (the m e c h a n i s m can b e f o r m u l a t e d f r o m
either m e s o m e r i c representation) to g i v e intermediate 5.24. A s with pyridine
N-oxides,
reaction
with
phosphorus
oxychloride
gives
useful
c h l o r o p y r i d i n e s 5.25. W e shall see the utility of 2- and 4 - c h l o r o p y r i d i n e s in
the n e x t section.
5.23a
5.4 Nucleophilic substitution of pyridines
P y r i d i n e c a n b e a t t a c k e d b y n u c l e o p h i l e s at t h e C 2 / C 6 a n d C 4 p o s i t i o n s in a
m a n n e r a n a l o g o u s t o t h e a d d i t i o n o f n u c l e o p h i l e s to a c a r b o n y l g r o u p in a
1,2 o r 1,4 f a s h i o n . A t t a c k at t h e C 3 / C 5 p o s i t i o n s is n o t f a v o u r e d b e c a u s e t h e
negative
charge
on the intermediate
cannot be delocalised
onto
the
electronegative nitrogen atom.
l
C
O
H
X = Nucleophile
T h e actual m e c h a n i s m is rather
c o m p l i c a t e d . H y d r o g e n g a s is
e v o l v e d , but in reality free s o d i u m
h y d r i d e is n e v e r g e n e r a t e d . S e e
McGill a n d R a p p a (1988).
U n d e r conditions of high temperatures the intermediate anion can rea r o m a t i s e b y l o s s of a h y d r i d e i o n , e v e n t h o u g h it is a v e r y p o o r l e a v i n g
g r o u p . T h i s is i l l u s t r a t e d b y t h e C h i c h i b a b i n r e a c t i o n o f p y r i d i n e
and
s o d a m i d e to p r o d u c e 2-aminopyridine 5.26. T h e immediate product of the
r e a c t i o n is 5 . 2 7 , t h e s o d i u m salt of 5 . 2 6 , as t h e e l i m i n a t e d h y d r i d e ion is
v e r y b a s i c . P r o t o n a t i o n of this s o d i u m salt d u r i n g t h e a q u e o u s w o r k u p t h e n
r e g e n e r a t e s 5 . 2 6 . A s i m p l i s t i c r a t i o n a l e is s h o w n b e l o w .
N'VhV
NaH (—H
—2) •
•
NH 2
-
N a H
N
5.26
Aq. workup
NH,
N
5.27
e
©
NH Na
These nucleophilic substitution reactions are m u c h more facile w h e n better
l e a v i n g g r o u p s (e.g. h a l i d e i o n s i n s t e a d of h y d r i d e i o n s ) are e m p l o y e d .
e
x
^
x
-a!
o
©
e -- 7
x
-ci
( h
•
3
e
•.
X = Nucleophile
N u c l e o p h i l i c s u b s t i t u t i o n s a r e w i d e l y u s e d in p y r i d i n e c h e m i s t r y . S o m e
e x a m p l e s are s h o w n below.
NH3
I
(|
^
HN(CH 2 CHCH 2 ) 2
CI
CI
Na® 0 SEt
H 2 NNH 2
"N'
^ci
"~N-NH2
H
H,NMe
N
SEt
CI
1
"N
^n'
CI
HNPh
H,NPhs>
rf
N
NMe
H
N
N'
CI
HN-™2
N'
"N
© ©
Na
OMe
i>
N
ci
N
OMe
F i n a l l y , b e f o r e l e a v i n g t h i s s e c t i o n , w e s h a l l c o n s i d e r t h e s y n t h e s i s of
pyridotriazine 5 . 3 2 , a potential anti-fungal drug. This synthesis illustrates
f e a t u r e s of b o t h e l e c t r o p h i l i c a n d n u c l e o p h i l i c p y r i d i n e c h e m i s t r y .
N i t r a t i o n of 4 - p y r i d o n e 5 . 2 3 gives 5 . 2 8 , a n d r e a c t i o n w i t h p h o s p h o r u s
oxychloride affords chloropyridine 5.29. This
pyridone-chloropyridine
conversion activates the system to nucleophilic attack by hydrazine, affording
5 . 3 0 . T h e n i t r o g r o u p a l s o f a c i l i t a t e s n u c l e o p h i l i c a t t a c k b y d e l o c a l i s a t i o n of
n e g a t i v e c h a r g e in t h e i n t e r m e d i a t e .
H,N
H 2 N - N H 2 CI
1
CI
NH
H,N
n
&
/P
•CX, NO,
N
5.29
N-Acylation, reduction
HN
N. e
© o
-CI
©
.NH,
NO-
N
5.30
of nitro to a m i n o , and c o n d e n s a t i o n
produce
d i h y d r o t r i a z i n e 5 . 3 1 . T h i s s y s t e m is r e a d i l y d e h y d r o g e n a t e d w i t h m a n g a n e s e
dioxide to afford the fully aromatic heterocycle 5.32. N o t e h o w relatively
s i m p l e c h e m i s t r y c a n b e u s e d to f o r m a q u i t e c o m p l e x h e t e r o c y c l e .
O
CI
NO,
HN03
1
H 2 SO 4
H
N
NO,
POC1,
H 2 NNH 2
N
H
5.23
HN
.NH,
CI.
HN'
fir
NO,
NO,
N
5.29
5.30
5.28
Pd/C
H2
11
HN'
HN'
MnO,
O
NH 2
Heat ( - H 2 0 )
N
N
5.32
5.31
H
.N.
N
5.5 Anion chemistry of pyridines
W e earlier drew a parallel between nucleophilic attack on the C 2 / C 6 and C 4
p o s i t i o n s of p y r i d i n e a n d 1,2 a n d 1,4 a d d i t i o n of n u c l e o p h i l e s to a c a r b o n y l
g r o u p . T h i s a n a l o g y c a n b e e x t e n d e d to d e p r o t o n a t i o n of a l k y l s u b s t i t u e n t s at
the C 2 / C 6 and C 4 positions.
J u s t as a c a r b o n y l g r o u p s t a b i l i s e s a n a d j a c e n t n e g a t i v e c h a r g e as a n
e n o l a t e a n i o n , s o t h e a n i o n d e r i v e d f r o m 2 - m e t h y l p y r i d i n e is s t a b i l i s e d by
d e l o c a l i s a t i o n of t h e n e g a t i v e c h a r g e o n t o t h e e l e c t r o n e g a t i v e n i t r o g e n a t o m .
A similar a r g u m e n t h o l d s f o r 4 - m e t h y l pyridine. T h e s e stabilised anions can
then react with the usual r a n g e of electrophiles.
T h e negative c h a r g e resulting
from deprotonation of the ethyl
methylene group of 5.33 c a n n o t
be delocalised onto the nitrogen
atom.
D i a l k y l p y r i d i n e 5 . 3 3 is selectively d e p r o t o n a t e d at the C 4 alkyl g r o u p ,
illustrating t h e g r e a t e r acidity of this p o s i t i o n o v e r the C 3 p o s i t i o n . W i t h
r e g a r d to ring d e p r o t o n a t i o n , h o w e v e r , t h e r e are relatively f e w e x a m p l e s
k n o w n f o r s i m p l e pyridines, in contrast to the extensive c h e m i s t r y d e v e l o p e d
f o r t h e f i v e - m e m b e r e d r i n g h e t e r o c y c l e s . T h i s is b e c a u s e t h e r e s u l t a n t
o r g a n o m e t a l l i c species are g o o d n u c l e o p h i l e s , and b e c a u s e pyridines are also
m o d e r a t e electrophiles, p o l y m e r i s a t i o n p r o b l e m s are often encountered. M o r e
s u c c e s s h a s b e e n a c h i e v e d w i t h s u b s t i t u t e d p y r i d i n e s h a v i n g an
ortho
activating
substituent
(e.g.
-CONHR,
-NHCOR,
-OMe,
- C H 2 N R 2 etc). T h e s e substituents increase the rate of kinetic d e p r o t o n a t i o n
and stabilise the intermediate o r g a n o l i t h i u m species by coordination.
F o r instance, 4 - a m i n o p y r i d i n e 5 . 3 4 can b e c o n v e r t e d to a m i d e 5.35 which,
o n t r e a t m e n t w i t h t w o e q u i v a l e n t s of b u t y l l i t h i u m , g i v e s o r g a n o m e t a l l i c
species 5.36. F o r m y l a t i o n of the m o r e reactive anion (the c a r b a n i o n ) then rep r o t o n a t i o n of the a m i d e a n i o n g i v e s 5 . 3 7 . A c i d i c h y d r o l y s i s r e m o v e s the
activating g r o u p to release pyridine aldehyde 5.38.
T h e m e t a l a t i o n p r o c e e d s by initial
d e p r o t o n a t i o n of the a m i d e
f o l l o w e d by o r f f r o - d i r e c t e d
d e p r o t o n a t i o n at the C3 position to
p r o d u c e the p s e u d o sixm e m b e r e d ring o r g a n o l i t h i u m
species 5.36.
f-BuCOCl
Et,N
1
n-BuLi
.
( 2 equivalents)
4
N
5.35
O
„
N
,
5.36
11
1.H
NMe 2
2.HC1 / H 2 0
NH 2
HN'^O
O
O
H
HC1 (conc.)
Heat
N
5.38
IN
5.37
5.6 Problems
1. W h a t is t h e m e c h a n i s m of this r e a c t i o n ?
r
C0 2 Et
CO z Et
C0 2 Et
NaOEt/EtOH
N
N
C0 2 Et
Hint. Start by acetylating the
pyridine to give a quaternary
cationic species. How can
deprotonation afford a
nucleophilic enamine-like system?
2. T h e c o n d e n s a t i o n of a c t i v e m e t h y l g r o u p s w i t h a l d e h y d e s c a n b e c a t a l y s e d
w
j t h a c e t i c a n h y d r i d e as w e l l as b a s e . S u g g e s t a p o s s i b l e m e c h a n i s m ,
Ph
PhCHO
Ac20/Ac0H
3. R a t i o n a l i s e t h e f o r m a t i o n of l a c t o n e 5 . 4 0 f r o m p y r i d y l a m i d e 5 . 3 9 .
OMe
DMP
1. 2 eq. n-BuLi
2.p-MeOC 6 H 4 CHO
3.H2S04/H20
4. S o m e p y r i d i n e N - o x i d e s a r e n o t j u s t s y n t h e t i c i n t e r m e d i a t e s , b u t a r e of
i n t e r e s t in t h e i r o w n r i g h t . F o r i n s t a n c e , p y r i d i n e N - o x i d e 5 . 4 1 is a n e w d r u g
c l a i m e d to b e u s e f u l f o r the t r e a t m e n t of senile d e m e n t i a . W h a t a r e t h e
m e c h a n i s m s of the p y r i d o n e - f o r m i n g step and the final displacement?
H
5
"1
oe
o©
5.7 References
A b r a m o v i t c h , R . A . (1974). In Pyridine and its derivatives
(The
chemistry
of heterocyclic
compounds
[ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , Vol.
14, S u p p l e m e n t Parts 1 - 4). W i l e y Interscience, N e w York.
E i s n e r , V . a n d K u t h u m , J. ( 1 9 7 2 ) . Chem. rev., 7 2 , 1 ( d i h y d r o p y r i d i n e s ) .
Furniss, B . S . , H a n n a f o r d , A.J., S m i t h , P . W . G . , and Tatchell, A . R . (1989).
Vogel's
textbook
of practical
organic
chemistry
( 5 t h e d n ) , p. 1168
(preparation of pyridine 5.13). L o n g m a n , H a r l o w .
K l i n s b e r g , E. ( 1 9 7 4 ) . In Pyridine
and its derivatives
(The chemistry
of
heterocyclic
compounds
[ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , Vol. 14,
Parts 1 - 4). W i l e y Interscience, N e w Y o r k .
M c G i l l , C . K . and R a p p a , A . ( 1 9 8 8 ) . Adv. heterocyclic
chem., 4 4 , 3.
S m i t h , D . M . (1979). In Heterocyclic
chemistry
(ed. P . G . S a m m e s ) (Vol.
4of Comprehensive
organic chemistry,
ed. D . B a r t o n and W . D . Ollis), p.3.
P e r g a m o n Press, O x f o r d .
6= Quinolines and isoquinolsnes
6.1 Introduction
Quinoline and isoquinoline can
also be viewed as being formally
derived from naphthalene
Q u i n o l i n e 6 . 1 and i s o q u i n o l i n e 6 . 2 are t w o i s o m e r i c h e t e r o c y c l i c s y s t e m s ,
w h i c h can b e e n v i s a g e d as b e i n g c o n s t r u c t e d f r o m the f u s i o n of a b e n z e n e
ring at t h e C 2 / C 3 and C 3 / C 4 p o s i t i o n s of p y r i d i n e r e s p e c t i v e l y . T h e y are
both ten 7t-electron a r o m a t i c heterocycles. L i k e pyridine, they are m o d e r a t e l y
b a s i c ( p ^ a q u i n o l i n e = 4.9, p K a i s o q u i n o l i n e = 5.1). I n d e e d q u i n o l i n e is
s o m e t i m e s used as a high boiling-point ( 2 3 7 ° C ) basic solvent.
Note the numbering system for
isoquinoline
A s w i t h p y r i d i n e , the n i t r o g e n a t o m s of quinoline and i s o q u i n o l i n e e a c h
b e a r a l o n e p a i r of electrons n o t i n v o l v e d in aromatic b o n d i n g w h i c h can be
p r o t o n a t e d , alkylated, or c o m p l e x e d to L e w i s acids. This c h a p t e r s h o u l d b e
read in c o n j u n c t i o n with the c h a p t e r o n pyridines as several points discussed
at l e n g t h t h e r e a r e a l s o r e l e v a n t to t h e c h e m i s t r y of q u i n o l i n e a n d
isoquinoline.
6.2 Synthesis of quinolines and isoquinolines
T h e classical S k r a u p synthesis of quinolines is e x e m p l i f i e d by the reaction of
aniline 6 . 3 with g l y c e r o l 6 . 4 u n d e r a c i d i c / o x i d a t i v e c o n d i t i o n s to p r o d u c e
quinoline 6.1.
At f i r s t sight this r e a c t i o n a p p e a r s to b e a n o t h e r o n e of t h o s e a n c i e n t
h e t e r o c y c l i c s y n t h e s e s that o w e m o r e to a l c h e m y than to logic, b u t in f a c t
the processes involved are relatively straightforward.
H
HO'
H
OH
OH
-H,0
OH
6.4
H
HO^^OH
/
H
-H,0
6.5
H
6.7
Protonation of glycerol 6.4 catalyses dehydration via secondary c a r b o n i u m
i o n 6 . 5 to g i v e enol 6 . 6 . A c i d c a t a l y s e d e l i m i n a t i o n of a s e c o n d w a t e r
m o l e c u l e a f f o r d s acrolein 6.7. T h u s glycerol acts essentially as a p r o t e c t e d
f o r m of acrolein, slowly releasing this unstable a , p - u n s a t u r a t e d aldehyde into
t h e r e a c t i o n m e d i u m . B e t t e r yields are r e a l i s e d w i t h this a p p r o a c h than if
acrolein itself is p r e s e n t f r o m the start. T h e reaction p r o c e e d s with a M i c h a e l
addition of aniline 6 . 3 to acrolein, p r o d u c i n g saturated a l d e h y d e 6 . 8 w h i c h
cyclises via an a r o m a t i c substitution reaction to alcohol 6.9. A c i d - c a t a l y s e d
dehydration to 6 . 1 0 then oxidation yields quinoline 6.1. N i t r o b e n z e n e can b e
used as a m i l d oxidant, as can iodine and ferric salts.
S o m e e x a m p l e s of the S k r a u p synthesis are s h o w n b e l o w .
OMe
OMe
T h e k e y i n t e r m e d i a t e s in t h e s y n t h e s i s of i s o q u i n o l i n e s a r e P a r y l e t h y l a m i n e s . F o r instance, a c y l a t i o n of P - p h e n y l e t h y l a m i n e 6 . 1 1 gives
a m i d e s of g e n e r a l s t r u c t u r e s 6.12 w h i c h c a n b e cyclised w i t h p h o s p h o r u s
o x y c h l o r i d e to p r o d u c e d i h y d r o i s o q u i n o l i n e 6.13. Better yields are o b t a i n e d
Acrolein is a highly reactive olefin
that is prone to polymerisation.
w i t h e l e c t r o n - d o n a t i n g g r o u p s o n t h e a r o m a t i c ring f a c i l i t a t i n g this a r o m a t i c
substitution cyclisation.
x.
X\
RCOC1
NH,
X = Electron-donating
substituent
Base
6.11
POC1
Pdu
-2H
6.13
6.14
T h i s d e h y d r o g e n a t i o n is
the reverse of a n o r m a l
hydrogenation reaction. T h e
d e h y d r o g e n a t i o n c a n be c a r r i e d
out u n d e r milder c o n d i t i o n s w h e n
a h y d r o g e n a c c e p t o r ( s u c h as
c y c l o h e x e n e ) is p r e s e n t .
A s in t h e S k r a u p q u i n o l i n e s y n t h e s i s , l o s s of t w o h y d r o g e n a t o m s is
n e c e s s a r y t o r e a c h t h e f u l l y a r o m a t i c s y s t e m . H o w e v e r , t h i s is u s u a l l y
a c c o m p l i s h e d in a s e p a r a t e s t e p , u t i l i s i n g p a l l a d i u m c a t a l y s i s t o
give
g e n e r a l i s e d i s o q u i n o l i n e 6 . 1 4 . T h i s is k n o w n as t h e B i s c h l e r - N a p i e r a l s k i
s y n t h e s i s . T h e m e c h a n i s m p r o b a b l y i n v o l v e s c o n v e r s i o n o f a m i d e 6 . 1 2 to
protonated
imidoyl
chloride 6.15
followed by electrophilic
aromatic
s u b s t i t u t i o n t o g i v e 6 . 1 3 . ( F o r a s i m i l a r a c t i v a t i o n of a n a m i d e t o a n
e l e c t r o p h i l i c s p e c i e s s e e t h e V i l s m e i e r r e a c t i o n , C h a p t e r 2.)
©
N-_
H
0 =PTClR
r O = PR- C I
ci-
a
R
R
6.13
6.15
a
X = Electron-donating substituent
f a
T h e P i c t e t - S p e n g l e r s y n t h e s i s is
usually u s e d w h e n t h e
tetrahydroisoquinoline oxidation
level is r e q u i r e d .
C l o s e l y r e l a t e d t o t h e B i s c h l e r - N a p i e r a l s k i s y n t h e s i s is t h e
Pictet-
Spengler synthesis, which utilises aldehydes rather than acylating species.
C o n d e n s a t i o n of P - a r y l e t h y l a m i n e s w i t h a l d e h y d e s p r o d u c e s i m i n e s s u c h as
6.16 which can be cyclised with acid to give tetrahydroisoquinoline 6.17. As
with the Bischler-Napieralski synthesis, electron-donating groups (typically
m e t h o x y g r o u p s ) f a c i l i t a t e t h e c y c l i s a t i o n step. T h e l o w e r o x i d a t i o n s t a t e of
6 . 1 7 as c o m p a r e d to 6 . 1 3 is a d i r e c t c o n s e q u e n c e of u s i n g a c a r b o n y l g r o u p at
t h e a l d e h y d e r a t h e r than c a r b o x y l i c a c i d o x i d a t i o n level. F o u r h y d r o g e n a t o m s
h a v e t o b e r e m o v e d f r o m t e t r a h y d r o i s o q u i n o l i n e s b y o x i d a t i o n to p r o d u c e the
fully aromatic isoquinoline.
O.
<
CH,0
NH,
6.16
HC1
H
NH
O
6.17
O
O O ' V j s I ,
H
6.3 Electrophilic substitution of quinoline and
isoquinoline
Quinoline and isoquinoline undergo electrophilic substitution reactions m o r e
easily than pyridine, though not surprisingly the i n c o m i n g
electrophile
a t t a c k s t h e b e n z e n o i d r i n g . A s w i t h p y r i d i n e , t h e n i t r o g e n a t o m s of q u i n o l i n e
a n d i s o q u i n o l i n e a r e p r o t o n a t e d u n d e r t h e t y p i c a l l y a c i d i c c o n d i t i o n s of
nitration or sulphonation, m a k i n g the heterocyclic ring resistant to attack.
T h e C 5 a n d C 8 p o s i t i o n s a r e m o s t s u s c e p t i b l e to e l e c t r o p h i l i c a t t a c k .
6.18a H
6.18b H
H
E p S
©
N;
r
6.19
H
A t t a c k of an e l e c t r o p h i l e at C 5 of p r o t o n a t e d q u i n o l i n e g i v e s c a t i o n
6 . 1 8 a , b w h i c h is s t a b i l i s e d b y r e s o n a n c e as s h o w n w i t h o u t d i s t u r b i n g t h e
aromaticity
of t h e a d j a c e n t p y r i d i n i u m
ring. However,
attack
of
an
e l e c t r o p h i l e at C 6 p r o d u c e s c a t i o n 6 . 1 9 w h i c h d o e s n o t p o s s e s s t h e s a m e
resonance stabilisation of cation 6.18a,b. (The student should p e r f o r m the
s a m e exercise f o r the C 7 and C 8 positions and c o n f i r m that the s a m e
a r g u m e n t s c a n b e applied.)
NO 2
HNO,
6.1
+
NO,
6.20
6.21
F o r i n s t a n c e , n i t r a t i o n of q u i n o l i n e g i v e s an e q u a l m i x t u r e of r e g i o i s o m e r s
6 . 2 0 a n d 6 . 2 1 . H o w e v e r , n i t r a t i o n of i s o q u i n o l i n e is r e a s o n a b l y s e l e c t i v e
(10:1) f o r the C 5 position over the C8, a f f o r d i n g mainly 6.22.
NO.
HNO,
H 2 SO„
6.22
6.4 Nucleophilic substitution of quinoline and
isoquinoline
Q u i n o l i n e a n d i s o q u i n o l i n e u n d e r g o n u c l e o p h i l i c s u b s t i t u t i o n r e a c t i o n s , like
pyridine.
1.KNH,/Heat
2. Aq. Workup
1
OEt
6.25
1. KNH 2 /Heat
NaOEt
6
2. Aq. Workup
6.24
CI
NH,
6.26
For instance, both quinoline and isoquinoline undergo the
OEt
Chichibabin
reaction (with f o r m a l h y d r i d e e l i m i n a t i o n , see C h a p t e r 5) to give
2-
aminoquinoline 6.23 and 1-aminoisoquinoline 6.24 respectively. Halogen
s u b s t i t u e n t s ortho
t o t h e n i t r o g e n a t o m s a r e e a s i l y d i s p l a c e d , as in t h e
preparations of 6.25 and 6.26.
X = Nucleophile
J
CI
N o t e t h a t n u c l e o p h i l i c d i s p l a c e m e n t in i s o q u i n o l i n e s o c c u r s m o r e e a s i l y at
t h e C I p o s i t i o n t h a n at t h e C 3 p o s i t i o n ( e v e n t h o u g h t h e y a r e b o t h ortho
to
n i t r o g e n ) b e c a u s e d i s p l a c e m e n t at C 3 i n v o l v e s t e m p o r a r y d i s r u p t i o n of t h e
benzenoid ring.
6.5 Anion chemistry of quinoline and isoquinoline
A l k y l g r o u p s at t h e C 2 a n d C 4 p o s i t i o n s of q u i n o l i n e c a n b e d e p r o t o n a t e d b y
s t r o n g b a s e s . T h i s is b e c a u s e (as w i t h p y r i d i n e ) t h e n e g a t i v e c h a r g e o n t h e
resultant carbanions c a n b e delocalised onto the electronegative nitrogen
a t o m , as in c a r b a n i o n 6 . 2 7 a , b .
N
^ ^
6.27a
N
6.27b ®
S u c h c a r b a n i o n s c a n b e alkylated, acylated, or c o n d e n s e d w i t h a l d e h y d e s :
l.KNH,
2. EtBr
O
l.KNH 2
2. PhC0 2 Et
N
Ph
1. NH4OH
Ar = />MeOC 6 H 4
2. ARCHO
T h i s t y p e of c h e m i s t r y is a l s o o b s e r v e d w i t h 1 - m e t h y l i s o q u i n o l i n e 6 . 2 8 .
H o w e v e r 3 - m e t h y l i s o q u i n o l i n e is m u c h less a c t i v a t e d b e c a u s e d e l o c a l i s a t i o n
of c h a r g e in 6 . 2 9 a , b i n v o l v e s d i s r u p t i o n of a r o m a t i c i t y of t h e b e n z e n o i d r i n g .
T h i s p h e n o m e n o n is c l o s e l y r e l a t e d to t h e r e l u c t a n c e of 3 - h a l o i s o q u i n o l i n e s
to undergo nucleophilic substitution.
6.29b
6.29a
As with pyridine, activated alkyl groups can be condensed with aldehydes
u n d e r a c i d i c as w e l l as b a s i c c o n d i t i o n s , as in t h e p r e p a r a t i o n o f 6 . 3 0 a n d
6.31.
PhCHO
T h e r e a d e r is referred to t h e
p r e v i o u s c h a p t e r ( P r o b l e m 2) for a
m e c h a n i s t i c e x p l a n a t i o n of s u c h
condensations.
ZnCl, / Heat
PhCHO
Ac,0/Heat
6.31
6.6 Problems
1. T h e s y n t h e s i s of t h e i m p o r t a n t q u i n o l o n e a n t i b i o t i c 6 . 3 3 is s h o w n . T h e
k e y s t a g e s a r e t h e G o u l d - J a c o b s o n q u i n o l o n e s y n t h e s i s to g i v e 6 . 3 2 , a n d t h e
d i s p l a c e m e n t r e a c t i o n t o a f f o r d 6 . 3 3 . W h a t a r e t h e m e c h a n i s m s of t h e s e
reactions?
O
o
EtO
O
OEt
OEt
NH,
Heat
o
CO,Et
l.NaH
cr
6.32
N
2. EtI
cr
H
1. NaOH
NH
CO,H
1.HN
2. HC1
HC1.HN
6.33
CO,Et
F
2. HC1
O
CO,H
2. A s y n t h e s i s of the n a t u r a l l y - o c c u r r i n g i s o q u i n o l i n e alkaloid 6 . 3 4 is s h o w n
b e l o w . W h a t r e a g e n t s m i g h t b e u s e d to a c c o m p l i s h each t r a n s f o r m a t i o n ?
O
R = CH2Ph
6.7 References
Adams, R. and Sloan, A . W .
(1941).
Organic
syntheses,
Coll. V o l . I, 4 7 8
(a real b l o o d - a n d - t h u n d e r preparation of quinoline).
Claret, P.A.
( 1 9 7 9 ) . In Heterocyclic
of Comprehensive
organic
chemistry
chemistry,
(ed. P . G . S a m m e s ) ( V o l . 4
ed. D. Barton and W . D . Ollis),
p. 155 (quinolines) a n d p . 2 0 5 (isoquinolines). P e r g a m o n Press, O x f o r d .
F u r n i s s , B . S . , H a n n a f o r d , A.J., S m i t h , P . W . G . , a n d T a t c h e l l , A . R .
Vogel
's textbook
of practical
organic
chemistry
(1989).
(5th e d n ) , p . 1 1 8 5 (a
rather m o r e s a f e t y - c o n s c i o u s preparation of quinoline). L o n g m a n , H a r l o w .
G r e t h e , G . (ed.) ( 1 9 8 1 ) . In Isoquinolines
compounds
(The
chemistry
of
heterocyclic
[ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , V o l . 3, Part 1). W i l e y
Interscience, N e w Y o r k .
Jones, G.
( 1 9 7 7 , 1990).
compounds
In Quinolines
(The
chemistry
of
heterocyclic
[ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , V o l . 3 2 , P a r t s 1, 2,
a n d 3). W i l e y Interscience, New*York.
K a t h a w a l a , G . F . , C o p p o l a , G . M . , a n d S c h u s t e r , H . F . ( e d . ) ( 1 9 8 9 ) . In
Isoquinolines
(The
chemistry
of
heterocyclic
compounds
[ed. A .
W e i s s b u r g e r a n d E . C . T a y l o r ] , V o l . 3, P a r t 2). W i l e y I n t e r s c i e n c e , N e w
York.
Manske, R.H.F. and Kalka, M.
( 1 9 5 3 ) . Organic
reactions,
7, 59 (Skraup
synthesis).
W h a l e y , W . M . a n d G o v i n d a c h a r i , T . R . ( 1 9 5 1 ) . Organic
( P i c t e t - S p e n g l e r synthesis).
reactions,
6, p.151
7. Indoles
7.1 Introduction
F u s i o n of a b e n z e n e r i n g o n t o t h e C 2 / C 3 p o s i t i o n s of p y r r o l e f o r m a l l y
p r o d u c e s the c o r r e s p o n d i n g b e n z o p y r r o l e 7.1 k n o w n as indole. A n a n a l o g o u s
t h e o r e t i c a l t r a n s f o r m a t i o n c a n b e e n v i s a g e d to f o r m b e n z o f u r a n 7 . 2 a n d
b e n z o t h i o p h e n e 7.3. T h i s c h a p t e r will c o n c e n t r a t e e x c l u s i v e l y o n indole, b y
f a r the m o s t i m p o r t a n t m e m b e r of this series.
7
H
Ou
1
Cm
Indole is a ten-7t electron a r o m a t i c system. A s with pyrrole, delocalisation
of t h e l o n e p a i r of e l e c t r o n s f r o m t h e n i t r o g e n a t o m is n e c e s s a r y f o r
a r o m a t i c i t y . T h e s i n g l e o v e r a l l e l e c t r o n i c s t r u c t u r e of i n d o l e is n o t
c o m p l e t e l y described by structure 7.1, b e c a u s e this implies localisation of t h e
l o n e pair o n the n i t r o g e n a t o m . M e s o m e r i c r e p r e s e n t a t i o n 7 . 1 a m a k e s a
c o n t r i b u t i o n to t h e e l e c t r o n i c s t r u c t u r e of indole, as to a l e s s e r e x t e n t d o
m e s o m e r i c representations w h e r e the negative charge occurs o n the b e n z e n o i d
ring.
[cu L
I
a?]
I®
H
7.1
J
H
7.1a
A c o n s e q u e n c e of this delocalisation is that t h e l o n e pair is n o t a v a i l a b l e
f o r p r o t o n a t i o n u n d e r m o d e r a t e l y acidic c o n d i t i o n s so, like p y r r o l e , indole is
a n o t h e r w e a k l y basic heterocycle. A n o t h e r similarity to p y r r o l e is that b e i n g
an 'electron-rich' h e t e r o c y c l e i n d o l e easily u n d e r g o e s a r o m a t i c electrophilic
s u b s t i t u t i o n , a n d is a l s o r a t h e r u n s t a b l e to o x i d a t i v e ( e l e c t r o n - l o s s )
c o n d i t i o n s . H o w e v e r , an i m p o r t a n t d i f f e r e n c e e m e r g e s here, in that w h e r e a s
p y r r o l e preferentially reacts with electrophiles at the C 2 / C 5 positions, indole
s u b s t i t u t e s s e l e c t i v e l y at t h e C 3 p o s i t i o n . T h e r e a s o n s f o r this w i l l b e
discussed later.
Historically,
interest
in
indoles
arose
with
the
isolation
and
c h a r a c t e r i s a t i o n o f m e m b e r s of t h e e n o r m o u s f a m i l y of i n d o l e a l k a l o i d s , s u c h
as l y s e r g i c a c i d 7.4. M a n y i n d o l e a l k a l o i d s p o s s e s s i n t e r e s t i n g a n d s o m e t i m e s
u s e f u l b i o l o g i c a l a c t i v i t i e s . A l t h o u g h n a t u r a l p r o d u c t c h e m i s t r y is still a n
a c t i v e a r e a of p r i m a r i l y a c a d e m i c r e s e a r c h , c o n s i d e r a b l y m o r e e f f o r t is
N e u r o t r a n s m i t t e r s are naturallyo c c u r r i n g s u b s t a n c e s w h i c h effect
chemical communication between
n e r v e cells by b i n d i n g at specific
sites o n the cell s u r f a c e called
receptors.
e x p e n d e d n o w a d a y s i n t h e p r e p a r a t i o n of i n d o l e d e r i v a t i v e s as p o t e n t i a l
drag candidates. Following on f r o m the observations that certain indole
alkaloids or their semi-synthetic derivatives (e.g. lysergic acid d i e t h y l a m i d e ,
L S D 7 . 5 ) h a v e p o t e n t c e n t r a l n e r v o u s s y s t e m a c t i v i t y , it w a s e s t a b l i s h e d t h a t
t h e s i m p l e i n d o l e 5 - h y d r o x y t r y p t a m i n e 7 . 6 is a m a j o r
Many
indole
derivatives
which
mimic
or
block
neurotransmitter.
the
binding
of
this
n e u r o t r a n s m i t t e r t o its r e c e p t o r s h a v e b e e n s y n t h e s i s e d a n d a r e b e g i n n i n g
t o f i n d u s e i n t h e t r e a t m e n t of v a r i o u s p s y c h o l o g i c a l d i s o r d e r s .
NH,
7.4 x = OH
7.5 X = NEt 2
7.6
7.2 Synthesis of indoles
A s m i g h t b e e x p e c t e d f o r a l a r g e b r a n c h of h e t e r o c y c l i c c h e m i s t r y , m a n y
s y n t h e s e s of i n d o l e s h a v e b e e n d e v e l o p e d . W e shall restrict o u r d i s c u s s i o n to
two, c o m m e n c i n g with the widely-used Fischer synthesis.
T h e F i s c h e r s y n t h e s i s is t h e c o n d e n s a t i o n of a n a r y l h y d r a z i n e w i t h a
k e t o n e f o l l o w e d b y c y c l i s a t i o n of t h e r e s u l t a n t h y d r a z o n e u n d e r a c i d i c
c o n d i t i o n s to g i v e t h e c o r r e s p o n d i n g i n d o l e , as i l l u s t r a t e d b y t h e p r e p a r a t i o n
of 2 - p h e n y l i n d o l e 7.9.
O
.A
Ph'
Ph - N - NH 2
H
7.7
-H20
Ph - N - N = <
H
Ph
AcOH or
JLPh
ZnCl2
7.8
7.9
T h e a c t u a l c y c l i s a t i o n s t a g e is n o t as i m p o n d e r a b l e as it a p p e a r s . T h e f i r s t
s t e p is t h e a c i d - c a t a l y s e d e q u i l i b r a t i o n b e t w e e n h y d r a z o n e 7 . 8 a n d e n e
hydrazine
7 . 1 ® . T h e n e x t s t e p , w h i c h i s i r r e v e r s i b l e , is a
concerted
electrocyclic reaction, f o r m i n g a strong c a r b o n - c a r b o n bond, and breaking a
weak nitrogen-nitrogen bond. T h e resulting imine 7 . 1 1 immediately rea r o m a t i s e s by t a u t o m e r i s a t i o n to aniline 7 . 1 2 . Finally,
acid-catalysed
e l i m i n a t i o n of a m m o n i a f o r m s i n d o l e 7.9, r e m i n i s c e n t of t h e last s t e p of t h e
K n o r r p y r r o l e s y n t h e s i s ( C h a p t e r 2).
T h e e l e c t r o c y c l i c r e a c t i o n is v e r y s i m i l a r to t h e C l a i s e n r e a r r a n g e m e n t of
p h e n y l allyl e t h e r 7 . 1 2 to g i v e p h e n o l 7 . 1 3
^ ^
H
LJ
J
J
0
OH
7.12
7.13
S o m e e x a m p l e s of t h e F i s c h e r i n d o l e s y n t h e s i s a r e s h o w n b e l o w .
OMe
,OMe
Ph - N - NH 2
H
41 OMe
Ph - N - NH 2
-f-
Ph-N-NH,
J
MeO
Y l
NMe2
+
H
F
F
F
An
"N-NH2
H
interesting
+
o
regioselectivity
question
arises
with
the
use
of
unsymmetrical ketone 7.14 to prepare indole 7.15. T w o ene hydrazines 7.16
Co
Pe
rearrangement
and 7 . 1 7 can f o r m , w h i c h w o u l d give rise to indoles 7 . 1 5 and 7 . 1 8
respectively.
SPh
SPh
NH
7.15
N'
jr
Ph - N - NH 2
H
7.7
H
7.16
(major)
SPh
SPh
NH
N
H
7.18
7.17 ( minor)
In s u c h c a s e s t h e m o s t t h e r m o d y n a m i c a l l y s t a b l e e n e h y d r a z i n e , i.e. t h e o n e
w i t h t h e m o r e h i g h l y s u b s t i t u t e d d o u b l e b o n d , f o r m s p r e f e r e n t i a l l y . In t h i s
p a r t i c u l a r e x a m p l e t h e r e is a l s o e x t r a s t a b i l i s a t i o n d e r i v e d f r o m c o n j u g a t i o n
of t h e l o n e p a i r s of e l e c t r o n s o n t h e s u l p h u r a t o m w i t h t h e d o u b l e b o n d . T h i s
regioselectivity
in e n e
hydrazine
f o r m a t i o n is t h e n r e f l e c t e d
in
the
r e g i o s e l e c t i v i t y of i n d o l e f o r m a t i o n .
The
more
recent
Leimgruber
aminomethylenation
of
synthesis
nitrotoluene
is
illustrated
7 . 1 9 to give 7 . 2 0 ,
by
the
followed
by
h y d r o g e n a t i o n to p r o d u c e i n d o l e 7.1.
MeO. , O M e
h
^
N
7.21
^
N
NO,
Heat
7.19
1
Pd/C
NO,
7.20
7.1
T h e c o m b i n a t i o n of f o r m y l p y r r o l i d i n e a c e t a l 7 . 2 1 a n d n i t r o t o l u e n e 7 . 1 9
produces electrophilic cation 7.22 and nucleophilic carbanion 7.23a,b which
r e a c t t o g e t h e r a f f o r d i n g e n a m i n e 7.20.
MeO)°Me
h ^ N
,
7_21
- c e
©
— OMe
5
7.20
H y d r o g e n a t i o n of e n a m i n e 7 . 2 0 r e d u c e s t h e n i t r o g r o u p g i v i n g a n i l i n e
7 . 2 4 , t h e n e l i m i n a t i o n of p y r r o l i d i n e p r o d u c e s i n d o l e 7 . 1 . N o t e t h e s i m i l a r i t y
of this r i n g c l o s u r e s t e p t o t h e last s t e p of t h e F i s c h e r s y n t h e s i s . I n b o t h
c a s e s t h e e v e n t u a l C 2 c a r b o n a t o m is f o r m a l l y at t h e c a r b o n y l o x i d a t i o n
l e v e l , e v e n t h o u g h it o c c u r s as e i t h e r a n i m i n e ( F i s c h e r s y n t h e s i s ) o r a n
e n a m i n e ( L e i m g r u b e r s y n t h e s i s ) . E l i m i n a t i o n of a m m o n i a o r p y r r o l i d i n e
r e s p e c t i v e l y is a n a l o g o u s to a c o n d e n s a t i o n p r o c e s s i n v o l v i n g e l i m i n a t i o n of
w a t e r (as in t h e K n o r r p y r r o l e s y n t h e s i s ) .
D
D
H,
'^CcT
Pd/C
Hen
^ o7.24 c
-
N
H
O u
7.1
7.20
S o m e e x a m p l e s of t h e L e i m g r u b e r s y n t h e s i s a r e s h o w n b e l o w .
MeO
XX
MeO
J
NO,
H2N-
^
-no2
NO,
J
o-
NO,
7.3 Electrophilic substitution of indoles
A s an electron-rich heterocycle, indole easily undergoes
electrophilic
s u b s t i t u t i o n . H o w e v e r w h e r e a s p y r r o l e r e a c t s p r e f e r e n t i a l l y at t h e C 2 / C 5
p o s i t i o n s ( s e e C h a p t e r 2), i n d o l e r e a c t s p r e f e r e n t i a l l y at the C 3 p o s i t i o n .
1
H
J
E®
H
©V
7.25 H
"H
E
N
I
H
O n e e x p l a n a t i o n is t h a t a t t a c k at C 2 r e s u l t s in d i s r u p t i o n of t h e a r o m a t i c i t y
of t h e b e n z e n o i d r i n g , as in i n t e r m e d i a t e 7 . 2 5 . T h i s is t h e r e f o r e a h i g h - e n e r g y
i n t e r m e d i a t e , a n d this r e a c t i o n p a t h w a y is s l o w e r b e c a u s e t h e first s t e p is r a t e d e t e r m i n i n g . A l s o t h e C 3 s e l e c t i v i t y is i n a c c o r d w i t h t h e e l e c t r o p h i l e
a t t a c k i n g t h e site of h i g h e s t e l e c t r o n d e n s i t y o n t h e r i n g . In e s s e n c e , i n d o l e
tends to react like an e n a m i n e t o w a r d s electrophiles, with
O
C0 2 Et
C0 2 Et
c o c
H,N'
substitution
occurring at the C 3 position, although substitution occurs at the C 2 position
w h e n t h e C 3 p o s i t i o n is b l o c k e d .
I n d o l e i t s e l f is u n s t a b l e t o t h e m i n e r a l a c i d c o n d i t i o n s f o r n i t r a t i o n . T h e
n i t r a t i o n of s u b s t i t u t e d i n d o l e s is q u i t e c o m p l e x a n d t h e o u t c o m e is d e p e n d e n t
on the precise reaction conditions.
L i k e pyrrole, indole readily undergoes the M a n n i c h reaction a f f o r d i n g the
a m i n o m e t h y l d e r i v a t i v e 7 . 2 6 . A v a r i e t y of n u c l e o p h i l e s c a n d i s p l a c e t h e
a m i n e via
a n e l i m i n a t i o n f o l l o w e d b y a 1 , 4 - a d d i t i o n r e a c t i o n , as i n t h e
p r e p a r a t i o n of a c e t a t e 7 . 2 7 .
CH 2 0
H
T h i s is t h e r e a c t i v e electrophilic
s p e c i e s of t h e M a n n i c h reaction.
CH 2 =
©
NMe 2
NMe2
HNMe 2
AcOH
H
7.26
7.1
r e , OAc
f^jl
|f^>-NMe2
OAc
NaOAc
v
AcOH
H
7.27
T h e Vilsmeier reaction proceeds extremely well with indoles giving
T h i s is t h e reactive electrophilic
s p e c i e s of the V i l s m e i e r reaction.
a l d e h y d e s s u c h as 7 . 2 8 .
H
^>=NMe2
CI
J
1. P0C13 / HCONMe2
•
2. H 2 0
7.1
7.28
A l d e h y d e 7 . 2 8 is a n o t h e r u s e f u l s y n t h e t i c i n t e r m e d i a t e , r e a d i l y u n d e r g o i n g
condensation reactions with active methylene c o m p o u n d s such as malonic
acid and nitromethane to p r o d u c e 7.29 and 7.30.
^C02H
CO 2 H
Pyridine
H
7.30
,C02H
•
Oc?
7.29
H
A c y l a t i o n of t h e C 3 p o s i t i o n c a n a l s o b e a c c o m p l i s h e d w i t h a c i d c h l o r i d e s ,
as i l l u s t r a t e d i n t h e s y n t h e s i s o f i n d o l e 7 . 3 4 , a d r u g f o r t h e t r e a t m e n t o f
d e p r e s s i o n . R e a c t i o n of i n d o l e 7 . 3 1 with oxalyl c h l o r i d e a f f o r d s C 3 s u b s t i t u t e d p r o d u c t 7 . 3 2 e v e n t h o u g h t h e b e n z e n e r i n g is v e r y e l e c t r o n - r i c h .
C o n v e r s i o n t o a m i d e 7 . 3 3 is f o l l o w e d b y r e d u c t i o n w i t h l i t h i u m a l u m i n i u m
hydride which removes both carbonyl groups, affording the target indole
7.34.
ci
cr
MeO
MeO
MeO
MeO
CI
H
1
7.31
U
7.32
o
H
H
N.
N
Ph
O
MeO
r^^NPh
MeO
1.LiAlH 4
2. H 2 0
MeO
N'
H
MeO
7.34
7.33
7.4 Anion chemistry of indole
T r e a t m e n t o f i n d o l e ( p K a = 1 7 ) w i t h s t r o n g b a s e s s u c h as b u t y l l i t h i u m ,
Grignard reagents, or metal hydrides produces the corresponding indolyl
a n i o n , w h i c h r e a c t s w i t h e l e c t r o p h i l e s e i t h e r o n n i t r o g e n o r at t h e C 3
p o s i t i o n . W i t h l i t h i u m , s o d i u m , o r p o t a s s i u m as c o u n t e r i o n t h e i n d o l y l
a n i o n t e n d s to r e a c t o n n i t r o g e n , as in t h e p r e p a r a t i o n of 7 . 3 5 . H o w e v e r , w i t h
m a g n e s i u m as t h e c o u n t e r i o n t h e i n t e r m e d i a t e h a s a n e s s e n t i a l l y c o v a l e n t
r a t h e r t h a n i o n i c s t r u c t u r e , a n d r e a c t i o n t e n d s to o c c u r at t h e C 3 p o s i t i o n , as
in t h e p r e p a r a t i o n of 7 . 3 6 .
NaH
a
u
Mel
1
N
e
7.1
7.35
Na'
Me
EtMgBr
cx?
H
a
NJ-
— MgBr2
7.36
MgBr
H
W h e n t h e n i t r o g e n is b l o c k e d , d e p r o t o n a t i o n c a n o c c u r at t h e C 2 p o s i t i o n ,
a d j a c e n t to t h e e l e c t r o n e g a t i v e h e t e r o a t o m . T h i s o f f e r s a m e a n s of i n t r o d u c i n g
e l e c t r o p h i l e s at this p o s i t i o n , c o m p l e m e n t i n g t h e C 3 s e l e c t i v i t y s h o w n b y
classical electrophilic substitution. For instance, alcohol 7.37 can be prepared
in t h i s w a y u s i n g e t h y l e n e o x i d e as t h e e l e c t r o p h i l e .
J
N
7.35
Me
n - BuLi
J8
N
Me
Li
2. HC1 / H 2 0
7.37
7.5 Problems
1. D e v i s e a s y n t h e s i s of the a n t i d e p r e s s a n t d r u g 7.38.
7.38
2.
T h e s y n t h e s i s of a m i n o e s t e r 7 . 4 1
is s h o w n b e l o w . W h a t is t h e
m e c h a n i s m of the c o n v e r s i o n of 7 . 3 9 to 7.40.
C0 2 Et
^ ^
NMe2
fto
2
R
0
Heat
7.39
C
/
°
jy
||
N0
^ ^ N ^
H
E
2
H
t
„
•, ,
Raney nickel ^
^
2
C0 22Et
^
^ ^ N
7-40
H
7.41
R = PhCH2
3. It w a s i n t e n d e d to p r e p a r e i m i n e 7 . 4 3 f r o m indole 7 . 4 2 b y d e p r o t o n a t i o n
at the C 2 p o s i t i o n then q u e n c h i n g w i t h b e n z o n i t r i l e f o l l o w e d by an a q u e o u s
w o r k u p . H o w e v e r , the isolated p r o d u c t s w e r e k e t o n e 7 . 4 4 a n d s u l p h o n a m i d e
7.45. A c c o u n t f o r this o b s e r v a t i o n .
7.42
i-
11 ~n i. n-BuLi
|
l|
II
N
l
o = sI =
Ph
\ /
r
-V-+2. P h — C E N / N
3. HCl / H 2 0
o
I
?r i
Ph
II
I
^ - ^ N
I
0 = S =0
^ _ Ii _
Ph
7.43
NH2
7.44
Ph
0 = S=0
Ph
N
H
M
O
7.4 5
7.6 References
Brown, R.T. and Joule, J.A. (1979).
In Heterocyclic
' S a m m e s ) (Vol. 4 of Comprehensive
organic
chemistry,
W . D . Ollis), p.411 (indoles and related systems).
chemistry
(ed. P . G .
e d . D. B a r t o n a n d
Pergamon
Press,
Oxford.
F u r n i s s , B . S . , H a n n a f o r d , A.J., S m i t h , P . W . G . , a n d T a t c h e l l , A . R .
Vogel's
textbook
of practical
organic
chemistry
(1989).
( 5 t h e d n ) , p . 1161
( p r e p a r a t i o n of indole 7.9). L o n g m a n , H a r l o w .
H o u l i h a n , W . J . ( e d . ) ( 1 9 7 2 ) . Indoles
compounds
(The
chemistry
of
heterocyclic
[ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , V o l . 2 5 , P a r t s 1 - 3).
W i l e y Interscience, N e w York.
Leimgruber, W.
Robinson, B.
Saxton,
J.E.
(1985).
(1969).
(ed.)
Organic
Chem.
rev.,
syntheses,
63, 214 (indole synthesis).
69, 227 (Fischer indole synthesis).
( 1 9 7 9 ) . Indoles
(The
chemistry
of
heterocyclic
compounds
[ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , V o l . 25, Part 4). W i l e y I n t e r s c i e n c e ,
N e w York.
Sundberg, R.J.
(1970).
The
chemistry
of
indoles.
Academic
Press,
8. Five-membered ring
heterocycles with three or four
heteroatonris
8.1 Introduction
T h e b r o a d c a t e g o r y of f i v e - m e m b e r e d r i n g h e t e r o c y c l e s c o n t a i n i n g t h r e e o r
four heteroatoms encompasses m a n y heterocyclic systems. Obviously there
is c o n s i d e r a b l e v a r i a t i o n in t h e p h y s i c a l a n d c h e m i c a l p r o p e r t i e s of s u c h a
l a r g e g r o u p of h e t e r o c y c l e s . F o r i n s t a n c e , w i t h r e g a r d t o
aromaticity,
Note the parallel w i t h f u r a n b e i n g
less a r o m a t i c t h a n pyrrole,
C h a p t e r 2.
o x a d i a z o l e 8 . 3 is c o n s i d e r e d to b e l e s s a r o m a t i c than t r i a z o l e 8 . 8 or t e t r a z o l e
8.9.
n
N-N
V
N
8.4
N
N
oxadiazoles
4.,
c
8.3
8.2
8.1
0
U
N-N
V
U
3
8.7
thiadiazoles
N-N
5^ >2
N,
H
8.9
N
8.6
8.5
4 N-N
5
N
H '
tetrazole
N-N
o
>
i
O
oxatriazole
8.10
S
thiatriazole
8.11
N e v e r t h e l e s s , this c o l l e c t i o n of h e t e r o c y c l e s d o e s s h a r e c e r t a i n c h a r a c t e r i s t i c s .
T h e trend w e h a v e seen of d e c r e a s i n g t e n d e n c y t o w a r d s
electrophilic
s u b s t i t u t i o n o n g o i n g f r o m f u r a n , p y r r o l e , a n d t h i o p h e n e t o t h e a z o l e s is
c o n t i n u e d i n t o t h e s e series. T h e p r e s e n c e of a d d i t i o n a l ' p y r i d i n e - l i k e ' n i t r o g e n
atoms renders these systems particularly 'electron-deficient', and electrophilic
s u b s t i t u t i o n is of little i m p o r t a n c e .
C o n v e r s e l y , n u c l e o p h i l i c s u b s t i t u t i o n ( w h i c h w e h a v e s e e n in e a r l i e r
c h a p t e r s o n 1 , 3 - a z o l e s a n d p y r i d i n e s ) d o e s o c c u r in t h e s e s y s t e m s , e s p e c i a l l y
w h e n t h e c a r b o n a t o m c o n c e r n e d is b e t w e e n t w o h e t e r o a t o m s , as in t h e
d i s p l a c e m e n t r e a c t i o n s of o x a d i a z o l e 8 . 1 2 a n d t e t r a z o l e 8.13.
3
V
N
N J
H
triazoles
8.8
2
O n c e a g a i n note the a n a l o g y w i t h
stand
l 11IUU1V1 1111111 Lll 11J V» 1111
AO 11 'Ul U1V1V IUV/ W\ [11I 1J11VI V.' 1 Ut/piuiuijdtion
of a l k y l s u b s t i t u e n t s b e t w e e n t w o h e t e r o a t o m s f o l l o w e d b y q u e n c h i n g t h e
r e s u l t a n t c a r b a n i o n s w i t h e l e c t r o p h i l e s , as in t h e p r e p a r a t i o n of o x a d i a z o l e
8.14.
R i n g d e p r o t o n a t i o n is a l s o k n o w n w i t h c e r t a i n m e m b e r s of t h e s e s e r i e s .
C a r b a n i o n 8 . 1 5 is s t a b l e at l o w t e m p e r a t u r e ( - 7 0 ° C ) a n d c a n b e t r a p p e d w i t h
e l e c t r o p h i l e s , b u t o n w a r m i n g t o r o o m t e m p e r a t u r e it d e c o m p o s e s w i t h r i n g
f r a g m e n t a t i o n a n d e x t r u s i o n o f n i t r o g e n . T h i s f r a g m e n t a t i o n p r o c e s s is
r e m i n i s c e n t of t h e b a s e - c a t a l y s e d c l e a v a g e of i s o x a z o l e s ( C h a p t e r 4).
F o r s i m p l i c i t y w e shall n o w c o n s i d e r t h e s y n t h e s i s of j u s t t h r e e m e m b e r s
of t h e s e s e r i e s , 1 , 2 , 4 - o x a d i a z o l e 8.3, 1,2,3-triazole 8.7, a n d t e t r a z o l e 8.9.
8.2 Synthesis of 1,2,4-oxadiazoles
D i s c o n n e c t i o n o f t h e C 5 - o x y g e n b o n d in 8 . 1 6 l e a d s to i m i n o a l c o h o l 8 . 1 7
w h i c h o c c u r s a s a m i d e 8 . 1 8 . C l e a v a g e of t h e a m i d e l i n k a g e l e a d s t o a n
activated carboxylic acid 8.20 plus the heteroatom-containing
8.19.
amidoxime
R
R
2
R A ' *
R,^
8.16
2
OH
O H
^
R2
R2
N-OH
N.OH
8 1 8
8.17
8 1 9
+
R
A
X
8 2 0
X = Leaving group
A m i d o x i m e s can b e p r e p a r e d b y a c i d - c a t a l y s e d additon of h y d r o x y l a m i n e to
nitriles.
R2-CEN
H 2 NOH
•
HCL
R~V
R 2 — C E N —H
A ®
V .
H 2 N— OH
»•
D
2
A
K T
^
0 H
N
H
A n e x a m p l e of this a p p r o a c h to o x a d i a z o l e s is s h o w n b y t h e c o n v e r s i o n of
e s t e r 8 . 2 1 t o o x a d i a z o l e 8 . 2 2 , p r e p a r e d as a p o t e n t i a l c a n d i d a t e f o r t h e
t r e a t m e n t of s e n i l e d e m e n t i a . S i m p l e e s t e r s a r e m e t a b o l i c a l l y u n s t a b l e in m a n
b e c a u s e of the high activity of esterases. T h e s e e n z y m e s catalyse the
h y d r o l y s i s of e s t e r s to c a r b o x y l i c a c i d s . A c o m m o n t a c t i c in d r u g r e s e a r c h
w h e n c o n f r o n t e d w i t h t h e p r o b l e m o f m e t a b o l i c i n s t a b i l i t y of a b i o l o g i c a l l y
a c t i v e e s t e r is t o r e p l a c e t h e e s t e r g r o u p w i t h a s m a l l h e t e r o c y c l e ( o f t e n
o x a d i a z o l e ) , to try to p r o d u c e a b i o l o g i c a l l y - a c t i v e m o l e c u l e w i t h i m p r o v e d
m e t a b o l i c s t a b i l i t y . T h i s c o n c e p t o f r e p l a c i n g f r a g m e n t s of a m o l e c u l e b y
g r o u p s w i t h b r o a d l y s i m i l a r p h y s i c o c h e m i c a l p a r a m e t e r s in a s y s t e m a t i c
m a n n e r is k n o w n as b i o i s o s t e r i c r e p l a c e m e n t . In this i n s t a n c e o x a d i a z o l e
8 . 2 2 c a n m i m i c b o t h t h e p h y s i c a l a n d b i o l o g i c a l p r o p e r t i e s of 8 . 2 1 , b u t it is
obviously not a substrate for esterases.
NH
J^
M
OMe
M
0
0®
£
__
r C r ^ o -
EtOH, heat
8.21
8.22
8.3 Synthesis of 1,2,3-triazoles
T h e s e a r e b e s t p r e p a r e d b y a 1 , 3 - d i p o l a r c y c l o a d d i t i o n of an a z i d e a n d a n
acetylene.
NaNj
H-CEC-H
•
r— N
//
N
8 8
-
H 2 SO 4
H
F o r instance, triazole 8 . 8 itself h a s b e e n p r e p a r e d b y c y c l o a d d i t i o n of
h y d r a z o i c acid to acetylene.
A l t h o u g h a s i m p l e m e c h a n i s m c a n b e d r a w n f o r t h i s t r a n s f o r m a t i o n , it is
o n l y u s e f u l as a ' b o o k - k e e p i n g e x e r c i s e ' to e n s u r e t h a t t h e c o r r e c t s t r u c t u r e is
d r a w n f o r t h e p r o d u c t . In r e a l i t y t h e r e a c t i o n is a c o n c e r t e d p r o c e s s a n d t h e
u s u a l c o n s i d e r a t i o n s of n u c l e o p h i l i c a n d e l e c t r o p h i l i c attack d o n o t a p p l y .
E x c e l l e n t y i e l d s a r e a c h i e v e d in t h e s e c y c l o a d d i t i o n s w h e n
electron-
w i t h d r a w i n g g r o u p s a r e p r e s e n t o n t h e a c e t y l e n e , as in t h e p r e p a r a t i o n of
triazol
8.23
m
8.4 Synthesis of tetrazoles
T e t r a z o l e itself e x p l o d e s o n
heating with loss of t w o m o l e c u l e s
of N 2
'
T e t r a z o l e s of g e n e r a l s t r u c t u r e 8 . 2 4 c a n b e p r e p a r e d in a v e r y s i m i l a r m a n n e r
tQ t r i a z o l e S )
eXcept
that nitriles are used rather than acetylenes. O n c e again
t h e r e a c t i o n w i t h a z i d e s is a c o n c e r t e d c y c l o a d d i t i o n p r o c e s s .
N
III
N
Jn©
heat
C V . / J
I
R
X
•
N-N
N
N'
K
|
I
8.24
2
R2
L e t u s n o w c o n s i d e r t h e s y n t h e s i s o f t e t r a z o l e 8 . 2 7 , a n i n h i b i t o r of t h e
e n z y m e o r n i t h i n e d e c a r b o x y l a s e , w h i c h c a t a l y s e s t h e c o n v e r s i o n of o r n i t h i n e
8.25 to diamine 8.26.
N-N
„
_
COM
—
NH2
"
C
•
°
H2N^ ^ ^
^
^
,NM2
M
H2N
8.26
2
NH2
8.25
N
"
8.27
T h e t e t r a z o l e m o i e t y is a n e x c e l l e n t b i o i s o s t e r i c r e p l a c e m e n t f o r a
c a r b o x y l i c a c i d , b e i n g a s m a l l , p o l a r , acidic h e t e r o c y c l e .
N
"
N
,
L >
NH
^
\
O
Aoh
v
N-N
N
1
-^N
+
pKa = 5 6 3
0
O
^
+
h
p k a
=
4 7 6
T e t r a z o l e 8 . 2 7 is s u f f i c i e n t l y s i m i l a r to o r n i t h i n e 8 . 2 5 in its p h y s i c a l
p r o p e r t i e s to b i n d to t h e a c t i v e site of t h e e n z y m e . H o w e v e r , as it o b v i o u s l y
c a n n o t u n d e r g o t h e d e c a r b o x y l a t i o n p r o c e s s , it a c t s as a n i n h i b i t o r of t h e
enzyme.
T h e s y n t h e s i s c o m m e n c e s w i t h a l k y l a t i o n of t h e s t a b i l i s e d c a r b a n i o n
derived f r o m cyanoester 8.29 with iodide 8.28 to give adduct 8.30
Cycloaddition with sodium azide followed by acidification during aqueous
w o r k u p a f f o r d s tetrazole 8 . 3 1 .
NaN,
N - N11
7
'
N
N
R-CEN
Na
e
N-N
HCl
11
N
H
8.32
8.31
N o t e t h a t t h e f i r s t - f o r m e d p r o d u c t f r o m t h e c y c l o a d d i t i o n is a c t u a l l y t h e
s o d i u m t e t r a z o l a t e salt 8 . 3 2 . P r o t o n a t i o n a f f o r d s t h e n e u t r a l t e t r a z o l e 8 . 3 1 .
P r o l o n g e d acidic h y d r o l y s i s a c c o m p l i s h e s s e v e r a l t r a n s f o r m a t i o n s : h y d r o l y t i c
r e m o v a l of b o t h t h e p h t h a l i m i d e a n d a c e t y l n i t r o g e n p r o t e c t i n g g r o u p s , a n d
h y d r o l y s i s / d e c a r b o x y l a t i o n of t h e ester. T h e n e t r e s u l t is to p r o d u c e t h e t a r g e t
tetrazole
8.27
as
its
dihydrochloride
salt.
This
tetrazole-assisted
d e c a r b o x y l a t i o n is m e c h a n i s t i c a l l y v e r y s i m i l a r t o t h e d e c a r b o x y l a t i o n of
malonyl half-esters 8.33.
8.33
8.5 Problems
1.
Triazoles and tetrazoles can be alkylated on nitrogen under basic
c o n d i t i o n s , as in t h e s y n t h e s i s of the c l i n i c a l l y - u s e d a n t i f u n g a l d r u g 8 . 3 5 in
w h i c h l|,2,4-triazole is a l k y l a t e d by a c h l o r o m e t h y l k e t o n e a n d an e p o x i d e ,
b o t h g o o d a l k y l a t i n g a g e n t s . W h a t is t h e m e c h a n i s m of f o r m a t i o n of e p o x i d e
8 . 3 4 ? O f c o m p o u n d s 8 . 3 4 a n d 8.35, w h i c h is achiral a n d w h i c h is r a c e m i c ?
F
8.34
2. W h a t is t h e m e c h a n i s m of f o r m a t i o n of o x a d i a z o l e 8 . 2 2 ?
NH
OMe
NNa
—>
H
N
JN
EtOH, heat
N
8.21
8.6
8.22
References
Butler, R.N.
(1977).
Adv.
C l a p p , L . B . ( 1 9 7 6 ) . Adv.
Gilchrist, T.L.
heterocyclic
heterocyclic
chem.,
chem,
( 1 9 8 5 ) . Heterocyclic
21, 323
(tetrazoles).
2 0 , 65, ( 1 , 2 , 4 - o x a d i a z o l e s ) .
chemistry,
p.81
(1,3-dipolar
c y c l o a d d i t i o n s in h e t e r o c y c l i c synthesis). L o n g m a n , H a r l o w .
Gilchrist, T.L. and Gymer, G.E. (1974).
Adv.
heterocyclic
chem.,
16, 33
(1,2,3-triazoles).
Grimmett, M.R.
(1979).
( V o l . 4 of Comprehensive
I n Heterocyclic
organic
chemistry
chemistry,
(ed. P . G . S a m m e s )
ed. D. Barton
W . D . Ollis), p . 3 5 7 (triazoles a n d tetrazoles). P e r g a m o n Press, O x f o r d .
and
Six-membered ring
heterocycles containing one
oxygen atom
9.1 Introduction
T h e p y r i l i u m cation 9.1, 2 - p y r o n e 9.2, 4 - p y r o n e 9.3, and their b e n z o - f u s e d
a n a l o g u e s the b e n z o p y r i l i u m cation 9.4, c o u m a r i n 9.5, c h r o m o n e 9.6, are the
parent structures of a series of s i x - m e m b e r e d ring heterocycles containing one
o x y g e n a t o m . T h e i m p e t u s f o r research in this area c o m e s f r o m the e n o r m o u s
n u m b e r of p l a n t - d e r i v e d n a t u r a l p r o d u c t s b a s e d o n t h e b e n z o p y r i l i u m ,
c o u m a r i n , and c h r o m o n e structures.
i2
9.1
cr ^-o
9.2
9.5
T h e red, violet, and b l u e p i g m e n t s of f l o w e r petals are called anthocyanins,
a n d are g l y c o s i d e s of v a r i o u s b e n z o p y r i l i u m cations. D e l p h i n i d i n c h l o r i d e
9.7, f o r e x a m p l e , is a b l u e p i g m e n t . K h e l l i n 9.8 is a natural p r o d u c t w h i c h
has f o u n d clinical a p p l i c a t i o n in t h e t r e a t m e n t of b r o n c h i a l a s t h m a and has
b e e n t h e starting p o i n t f o r the d e s i g n of m a n y totally synthetic c h r o m o n e s
with i m p r o v e d biological properties.
OH
OMe O
OH
OH
OMe
9.8
C o u m a r i n 9.5 is itself a natural p r o d u c t w h i c h occurs in l a v e n d e r oil and has
been f o u n d in o v e r sixty species of plants.
In natural product chemistry, the
acetal formed between an aliphatic
or aromatic alcohol and a sugar is
termed a glycoside.
T h e p y r y l i u m c a t i o n 9 . 1 is t h e o x y g e n a n a l o g u e of p y r i d i n e a n d is a six
Tc-electron a r o m a t i c s y s t e m . N e v e r t h e l e s s , b e i n g a c a t i o n it is r e a c t i v e
t o w a r d s n u c l e o p h i l e s a n d is r e a d i l y h y d r o l y s e d to g i v e d i a l d e h y d e 9 . 9 . T h e s e
r e a c t i o n s a r e r e v e r s i b l e , a f a c t w h i c h h a s b e e n u s e d in a s y n t h e s i s of 9 . 1 f r o m
9 . 9 . A t l o w p H ( h i g h a c i d i t y ) t h e e q u i l i b r i u m lies t o t h e s i d e of t h e p y r y l i u m
s p e c i e s 9 . 1 b u t if t h e m e d i u m is b a s i f i e d t h e n h y d r o l y s i s of 9 . 1 o c c u r s t o
g i v e 9.9. T h i s is b e c a u s e o n e m o l e of h y d r o x i d e is c o n s u m e d o n g o i n g f r o m
p y r y l i u m cation 9.1 to n e u t r a l a l d e h y d e 9.9. Increasing t h e
hydroxide
c o n c e n t r a t i o n t h e r e f o r e f o r c e s t h e e q u i l i b r i u m f r o m l e f t to right.
H,o.
O
-H,0
OH,
+ H©
9.1
T h e c a r b o n y l g r o u p s of 4 - p y r o n e
a n d 4 - p y r i d o n e a b s o r b at
approximately 1650 cm"1 and
1 5 5 0 c m " 1 respectively. T h e
l o w e r e n e r g y of the p y r i d o n e
a b s o r p t i o n reflects greater single
bond character, and hence
g r e a t e r delocalisation.
9.9
\
In c o n t r a s t , 2 - a n d 4 - p y r o n e s a r e c o n s i d e r e d t o h a v e r e l a t i v e l y little
a r o m a t i c c h a r a c t e r . W h e r e a s in a n a n a l o g o u s n i t r o g e n series 4 - p y r i d o n e 5 . 2 3
has significant aromatic character (mesomeric representation 5.23a m a k i n g a
c o n s i d e r a b l e c o n t r i b u t i o n to the overall electronic distribution), a r o m a t i c
m e s o m e r i c r e p r e s e n t a t i o n 9 . 3 a m a k e s l e s s of a c o n t r i b u t i o n t o t h e o v e r a l l
e l e c t r o n i c s t r u c t u r e of 4 - p y r o n e . A s w i t h f u r a n , t h e h i g h e r e l e c t r o n e g a t i v i t y
of o x y g e n
leads to h e t e r o c y c l e s of little a r o m a t i c i t y
in c a s e s
where
d e l o c a l i s a t i o n o f e l e c t r o n d e n s i t y f r o m t h e h e t e r o a t o m is a p r e r e q u i s i t e f o r
that a r o m a t i c i t y .
9.3
5.23
9.3a
5.23a
L e t u s n o w c o n s i d e r t h e s y n t h e s i s of a p y r y l i u m salt, a c o u m a r i n , a n d a
chromone.
9.2 Synthesis of a pyrylium salt
A t y p i c a l p y r i l i u m salt s y n t h e s i s is i l l u s t r a t e d b y t h e p r e p a r a t i o n of salt
9 . 1 2 . T h e p r e c u r s o r t o 9 . 1 2 is p y r a n 9 . 1 1 , a v a i l a b l e b y d e h y d r a t i o n of 1,5d i k e t o n e 9 . 1 0 . N o t e t h e s i m i l a r i t y of this s e q u e n c e to t h e H a n t z c h p y r i d i n e
s y n t h e s i s , C h a p t e r 5. A l s o , t h e d e h y d r a t i v e c y c l i s a t i o n of a d i k e t o n e to
o x y g e n h e t e r o c y c l e 9 . 1 1 is r e m i n i s c e n t of f u r a n s y n t h e s i s , C h a p t e r 2.
9.10
9.11
9.12
C10 4 °
O n e h y d r o g e n a t o m h a s to b e r e m o v e d f r o m t h e C 4 p o s i t i o n of p y r a n 9 . 1 1
t o p r o d u c e t h e p y r y l i u m c a t i o n , b u t it is i m p o r t a n t t o r e a l i s e t h a t
h y d r o g e n a t o m is l o s t n o t as a p r o t o n b u t as a negatively-charged
ion. T h e p r o c e s s is t h e r e f o r e a n oxidation
NM&j
the
hydride
of p y r a n 9 . 1 1 .
J
Ph
Ph
Ph
9.14a
9.13
Ph
9.14b
9.15
A s u i t a b l e o x i d a n t is c a t i o n 9 . 1 4 a , b , d e r i v e d f r o m a , ( 3 - u n s a t u r a t e d k e t o n e
9 . 1 3 b y p r o t o n a t i o n u n d e r s t r o n g l y a c i d i c c o n d i t i o n s in t h e a b s e n c e of w a t e r .
Q u e n c h i n g o f this c a t i o n w i t h a h y d r i d e i o n ( f r o m t h e C 4 p o s i t i o n of 9 . 1 1 )
p r o d u c e s t h e saturated k e t o n e 9 . 1 5 . T h e b a l a n c e d e q u a t i o n is s h o w n b e l o w .
H
Ph
HCIO,
+
Ph
H
Ph
O
9.13
Ph
Ph
Ph
O,
Ph
p h
CIO®
9.12
9.11
+
9.15
9.3 Synthesis of coumarins
Most pyrylium salts have electrondonating aromatic substituents at
the C2, C4, or C6 positions w h i c h
serve to stabilise the positive charge
by resonance.
Let us consider the synthesis of b r o m o c o u m a r i n 9.16, a c o m p o u n d which
exhibits
biological
activity
against
parasitic
trematodes
s c h i s t o s o m i a s i s , a v e r y c o m m o n d i s e a s e in t h e t r o p i c s .
that
cause
Retrosynthetic
c l e a v a g e of l a c t o n e 9 . 1 6 g i v e s d i e s t e r 9 . 1 7 , w h i c h in p r i n c i p l e c a n b e d e r i v e d
f r o m c o n d e n s a t i o n of o / t / z o - h y d r o x y b e n z a l d e h y d e 9 . 1 8 a n d diethyl m a l o n a t e .
H
CO,Et
Br
C0 2 Et
^oct
C0 2 Et
O-^O
9.18
9.16
In p r a c t i s e a K n o e v e n a g e l c o n d e n s a t i o n r e a c t i o n y i e l d s c o u m a r i n 9 . 1 6
d i r e c t l y , w i t h o u t i s o l a t i o n of d i e s t e r 9 . 1 7 . T h e m e c h a n i s m is s h o w n b e l o w .
-o
Br
3
H
e
OH
r
CO,Et
C0 2 Et
CO,Et
Br
9.16
9.17
H
+r
C0 2 Et
C0 2 Et
9.4 Synthesis of c h r o m o n e s
L e t u s c o n s i d e r t h e s y n t h e s i s of f l a v o n e 9 . 1 9 , w h i c h is t h e p a r e n t of a l a r g e
s e r i e s of n a t u r a l p r o d u c t s . D i s c o n n e c t i o n of t h e c a r b o n - o x y g e n b o n d in t h e
u s u a l w a y r e s u l t s in e n o l 9 . 2 0 w h i c h e x i s t s as 1 , 3 - d i k e t o n e 9 . 2 1 . T h i s 1,3d i c a r b o n y l r e l a t i o n s h i p c a n b e e x p l o i t e d in t h e c l a s s i c a l m a n n e r y i e l d i n g
<?7-//zo-hydroxyacetophenone
9.22. The synthetic problem centres
on
m e t h o d o l o g y f o r t h e C - b e n z o y l a t i o n of t h e e n o l a t e d e r i v e d f r o m 9 . 2 2 w i t h
s o m e a c t i v a t e d b e n z o i c acid d e r i v a t i v e 9.23.
O
o
OH
"Ph
o
OH
OH "Ph
9.20
9.19
O
+
•Ph
O
OH
9.21
In
practice,
the
9.22
Konstanecki-Robinson
O ^ P h
9.23
X = Leaving group
synthesis
of
chromones
c o m m e n c e s w i t h O - b e n z o y l a t i o n n o t C - b e n z o y l a t i o n , to a f f o r d e s t e r 9 . 2 4 .
B a s e - c a t a l y s e d r e a r r a n g e m e n t p r o d u c e s t h e r e q u i r e d 1 , 3 - d i k e t o n e 9 . 2 1 , via
i n t r a m o l e c ul a r b e n z o y l a t i o n of the intermediate enolate.
Acid-catalysed
d e h y d r a t i o n t h e n a f f o r d s f l a v o n e 9.19.
O
O
A
Ph
o
Pyridine
2. AcOH
OH
9. 22
AC0H/H 2 S0 4
l.KOH
CI
9.24
Ph
OH
O
"Ph
9.21
9.5 Reactions with nucleophiles
A l t h o u g h s o m e e x a m p l e s of e l e c t r o p h i l i c s u b s t i t u t i o n a r e k n o w n , t h e
chemistry
o f t h e s e s e r i e s is d o m i n a t e d b y n u c l e o p h i l i c
ring-opening
r e a c t i o n s , s o m e t i m e s f o l l o w e d b y r i n g - c l o s u r e to g i v e n e w h e t e r o c y c l e s . F o r
i n s t a n c e , a m i n o l y s i s of 9.1, 9 . 2 , a n d 9 . 3 l e a d s to p y r i d i n e 5 . 1 a n d p y r i d o n e s
5.22 a n d 5.23.
NH,
H,0
N
5.1
NH,
H,0
N
5.22
NH,
H,0
T h e m e c h a n i s m of t h e c o n v e r s i o n of 4 - p y r o n e to 4 - p y r i d o n e i n v o l v e s a n
initial M i c h a e l r e a c t i o n f o l l o w e d b y r i n g - o p e n i n g . T a u t o m e r i s a t i o n of e n o l
9 . 2 5 to a l d e h y d e 9 . 2 6 , f o l l o w e d b y c y c l i s a t i o n , a f f o r d s 4 - p y r i d o n e 5 . 2 3 .
9.25
9.26
5.23
T h e r e a c t i o n of p y r i l i u m salts w i t h n u c l e o p h i l e s m a y i n v o l v e e l e c t r o c y c l i c
r i n g - o p e n i n g of t h e i n t e r m e d i a t e d i e n e s , as in t h e f o r m a t i o n o f k e t o n e 9 . 2 7 .
Clo4
A s i m i l a r s u s c e p t i b i l i t y to n u c l e o p h i l i c a t t a c k is o b s e r v e d in t h e b e n z o f u s e d s e r i e s . C o u m a r i n 9 . 5 is h y d r o l y s e d b y h y d r o x i d e to c a r b o x y l a t e salt
9.28. T h i s p r o c e s s is r e v e r s i b l e , a n d a c i d i f i c a t i o n r e g e n e r a t e s t h e l a c t o n e .
NaOH
o
HCl
H
9.5
9.28
0
- o
e a
Na
A n i m p o r t a n t d i f f e r e n c e b e t w e e n t h e m o n o c y c l i c a n d b e n z o - f u s e d series is
t h a t r e a c t i o n s w i t h a m i n e s d o n o t l e a d t o t h e c o r r e s p o n d i n g h e t e r o c y c l e s in
t h e b e n z o - f u s e d s e r i e s . F o r i n s t a n c e , a m i n o l y s i s of c h r o m o n e 9 . 2 9 a f f o r d s
p h e n o l 9 . 3 0 . B e n z o p y r i d o n e 9 . 3 2 is n o t p r o d u c e d . T h e f a c i l e t a u t o m e r i s a t i o n
b e t w e e n 9 . 2 5 a n d 9 . 2 6 w o u l d a n a l o g o u s l y g i v e k e t o n e 9 . 3 1 in this s e r i e s .
T h i s h i g h - e n e r g y i n t e r m e d i a t e is n o t a r o m a t i c , a n d t h e r e a c t i o n s t o p s at
phenol 9.30.
O
O
NH,
O
9.29
NH,
9.30
P h e n o l s d o not exist or react in
their t a u t o m e r i c keto forms.
9.6 Problems
1. W h a t is t h e m e c h a n i s m of the c o n v e r s i o n of p y r o n e 9 . 2 to p y r i d o n e 5 . 2 2
by aminolysis?
NH,
^N
o
H
9.2
5.22
2. E x p l a i n the f o r m a t i o n of p y r a z o l e 9.33.
H,N— NH,
OH
9.6
9.33
3. H o w c a n c h r o m o n e 9 . 3 4 b e c o n v e r t e d to 9.35?
o
o
9
OMe
NMe2
9.34
OMe
9.35
4. W h a t is the m e c h a n i s m of this c y c l i s a t i o n ?
o
o
AcOH
L Jl
OH^/^ph
"O
9.21
9.7
Ph
9.19
References
Horing,
E.C.
et al.
( 1 9 5 5 ) . Organic
synthesis,
Coll. Vol. Ill,
165
( e x p e r i m e n t a l details of a K n o e v e n a g e l c o n d e n s a t i o n to g i v e a c o u m a r i n
ester).
L i v i n g s t o n e , R . ( 1 9 7 7 ) . In R o d d ' s Chemistry
of carbon
compounds,
Vol.
I V , p . 2 ( p y r i l i u m salts; 2- a n d 4 - p y r o n e s ) ; p . 6 9 ( b e n z o p y r i l i u m salts); p . 9 6
( c o u m a r i n s ) ; p . 1 3 8 ( c h r o m o n e s ) . Elsevier, A m s t e r d a m .
S t a u n t o n , J. ( 1 9 7 9 ) . I n Heterocyclic
chemistry
of Comprehensive
e d . D . B a r t o n a n d W . D . Ollis), p . 6 0 7
organic
chemistry,
(ed. P . G . S a m m e s ) ( V o l . 4
(pyrilium salts); p . 6 2 9 (2-pyrones and coumarins); p . 6 5 9
(4-pyrones
a n d c h r o m o n e s ) . P e r g a m o n Press, O x f o r d .
W h e e l e r , T . S . ( 1 9 6 3 ) . Organic
synthesis,
details f o r the p r e p a r a t i o n of f l a v o n e ) .
Coll. V o l . IV, 4 7 9 ( e x p e r i m e n t a l
10. Pyrimidines
10.1 Introduction
F o r m a l r e p l a c e m e n t of a C H unit in p y r i d i n e 5.1 by a nitrogen a t o m leads to
the series of three p o s s i b l e diazines, p y r i d a z i n e 10.1, p y r i m i d i n e 10.2, and
p y r a z i n e 10.3. Like p y r i d i n e they are fully a r o m a t i c heterocycles. T h e e f f e c t
of an a d d i t i o n a l n i t r o g e n a t o m as c o m p a r e d to p y r i d i n e a c c e n t u a t e s t h e
essential f e a t u r e s of pyridine chemistry. Electrophilic substitution is difficult
in s i m p l e u n a c t i v a t e d diazines b e c a u s e of b o t h e x t e n s i v e p r o t o n a t i o n u n d e r
strongly acidic conditions and the inherent lack of reactivity of the f r e e base.
Nucleophilic displacements are comparatively easier.
4
4
4
V 4
5 R^^I
n;
3
5 ,-C^N 3
5
N 2
6 L^. J ) 2
6
10.1
10.2
o :
n,
: 0
10.3
5.1
n'J
Interestingly, the second electronegative heteroatom reduces the capacity of
t h e d i a z i n e s to t o l e r a t e t h e p o s i t i v e c h a r g e r e s u l t i n g f r o m p r o t o n a t i o n .
P y r i d a z i n e 10.1 ( p ^ a = 2.24), p y r i m i d i n e 10.2 ( p K a = 1.23), and p y r a z i n e
10.3 ( p ^ a = 0.51) are all far less basic than pyridine ( p ^ a = 5.23).
T h e m o s t i m p o r t a n t of t h e d i a z i n e s is p y r i m i d i n e 1 0 . 2 . P y r i m i d i n e
derivatives uracil 10.4, t h y m i d i n e 10.5, and cytosine 10.6 are the m o n o c y c l i c
'bases' of nucleic acids. T h e bicyclic bases are the p u r i n e s a d e n i n e 10.7 and
g u a n i n e 10.8. T h e p u r i n e ring is essentially a f u s i o n of the p y r i m i d i n e and
imidazole rings.
O
H
O
10.4
10.5
H
NH2
10.6
H
NH2
O
H
10.7
T h e actual b i o s y n t h e s i s of p u r i n e s (illustrated b e l o w in a b b r e v i a t e d f o r m
for the nucleotide adenosine m o n o p h o s p h a t e A M P 1 0 . 9 ) involves
construction of a pyrimidine ring onto a p r e - f o r m e d imidazole.
10.8
Nucleotides are the monomeric
building blocks of
deoxyribonucleic acid (DNA) in
which is stored the genetic
information of the cell.
~nh2
"NH,
T h e e n z y m e s that m a n i p u l a t e n u c l e o t i d e s , n u c l e i c acids, etc. a r e t h e p o i n t s
NH
of t h e r a p e u t i c i n t e r v e n t i o n f o r a n u m b e r of d i s e a s e s i n v o l v i n g cell r e p l i c a t i o n
d i s o r d e r s s u c h as c a n c e r s a n d v i r a l i n f e c t i o n s . F o r i n s t a n c e , A Z T 1 0 . 1 0 , a n
HO— |
^ N - ^ O
i n h i b i t o r of t h e e n z y m e r e v e r s e t r a n s c r i p t a s e , is a n anti-viral d r u g c u r r e n t l y
u s e d in t h e t r e a t m e n t of A I D S .
W e s h a l l n o w g o o n to c o n s i d e r t h e s y n t h e s i s a n d c h e m i s t r y of t h e
10.10
N3
pyrimidine ring system.
10.2 Synthesis of pyrimidines
D i s c o n n e c t i o n of t h e N 1 - C 6 b o n d in g e n e r a l i s e d p y r i m i d i n e 1 0 . 1 1 in t h e
u s u a l w a y p r o d u c e s e n o l 1 0 . 1 2 , w h i c h e x i s t s as k e t o n e 1 0 . 1 3 .
disconnection
of t h e c a r b o n - n i t r o g e n
dicarbonyl compound
Similarly,
d o u b l e b o n d in 1 0 . 1 3 y i e l d s a
1 0 . 1 4 a n d an a m i d i n e 1 0 . 1 5 . T h i s
retrosynthetic
a n a l y s i s , s u g g e s t i n g t h e c o m b i n a t i o n of b i s - e l e c t r o p h i l i c a n d b i s - n u c l e o p h i l i c
c o m p o n e n t s , is t h e b a s i s of a v e r y g e n e r a l p y r i m i d i n e s y n t h e s i s .
R3
R3
R i
10.11
^ o h h n ^ R
H N ^ I
4
10.12
10.13
W h e r e R 4 is a h y d r o g e n o r c a r b o n a t o m , 1 0 . 1 5 is s i m p l y a n a m i d i n e .
NH 2
H o w e v e r , u r e a 1 0 . 1 6 , t h i o u r e a 1 0 . 1 7 , o r g u a n i d i n e 1 0 . 1 8 a n d their d e r i v a t i v e s
m a y be used. T h e s e nucleophiles m a y be condensed with ester and nitrile
f u n c t i o n a l i t i e s as w e l l as w i t h a l d e h y d e s a n d k e t o n e s . S u c h c o n d e n s a t i o n s t o
a f f o r d p y r i d i m i d i n e d e r i v a t i v e s are usually facilitated b y a c i d or b a s e catalysis,
NH2
H
s
NH 2
L
H2N"^NH
a l t h o u g h c e r t a i n c o m b i n a t i o n s of r e a c t i v e e l e c t r o p h i l i c a n d n u c l e o p h i l i c
1017
10.18
c o m p o u n d s r e q u i r e n o c a t a l y s t at all. S o m e e x a m p l e s a r e s h o w n b e l o w .
^
f
L
c
o
+
NH 2
L
H,N
0
HCl
EtOH
Heat
H OEt
•epared by in situ hydrolysis of
H
[
OEt
^ O E t
OEt
N o t e that s e v e r a l of t h e s e e x a m p l e s p r o d u c e p y r i m i d o n e s , a n a l o g o u s to t h e
p y r i d o n e s p r e v i o u s l y e n c o u n t e r e d in C h a p t e r 5. A r e p r e s e n t a t i v e m e c h a n i s m
is s h o w n f o r t h e p r e p a r a t i o n o f 2 - p y r i m i d o n e 1 0 . 1 9 , a n d is s i m p l y t w o
consecutive condensations.
10.3 Electrophilic substitution of pyrimidones
A s m e n t i o n e d earlier, e l e c t r o p h i l i c s u b s t i t u t i o n o n u n a c t i v a t e d p y r i m i d i n e s is
of little i m p o r t a n c e . B u t , as w i t h p y r i d i n e , t h e p y r i m i d i n e n u c l e u s c a n b e
a c t i v a t e d t o w a r d s e l e c t r o p h i l i c a t t a c k b y e m p l o y i n g N - o x i d e s or p y r i m i d o n e s ,
f o r t h e s a m e r e a s o n s as w e r e d i s c u s s e d in C h a p t e r 5.
F o r i n s t a n c e , n i t r a t i o n of 2 - p y r i m i d o n e 1 0 . 2 0 a f f o r d s n i t r o p y r i m i d o n e
1 0 . 2 1 . W i t h d o u b l y - a c t i v a t e d s y s t e m s s u c h as 1 0 . 2 2 , n i t r a t i o n to g i v e 1 0 . 2 3
can occur without heating.
Cl
HNO 3
NO.
NO-
T l
Heat
HNO,
Cz
H
H
10.20
10.21
V
O
A
NH
H
H
10.22
10.23
10.4 Nucleophilic substitution of pyrimidines
L e a v i n g g r o u p s at t h e C 2 , C 4 , a n d C 6 p o s i t i o n s of p y r i m i d i n e s c a n b e
displaced by nucleophiles, with the negative charge of the intermediate
delocalised over both nitrogen atoms.
ac
,e
Y <tN
N
x
X
X = Nucleophile
Y = Leaving group
PhNH,
N
J
Cl
NPh
H
Na © ©OMe
N
Cl
OMe
Chlorinated
pyrimidines
themselves
corresponding
pyrimidones
by
reaction
are often accessible f r o m
with
phosphorus
the
oxychloride.
( A g a i n , s e e C h a p t e r 5 f o r a n e x p l a n a t i o n o f t h i s s o r t of r e a c t i o n . ) F o r
i n s t a n c e , a m i n o p y r i m i d i n e 1 0 . 2 4 c a n b e s y n t h e s i s e d b y the c l a s s i c a l s e q u e n c e
depicted b e l o w .
OEt
HN
10.5 Problems
1.
W r i t e a m e c h a n i s m f o r this n i t r a t i o n , b u t s t a r t i n g f r o m an a l t e r n a t i v e
m e s o m e r i c r e p r e s e n t a t i o n of 1 0 . 2 0 t h a t h e l p s to e x p l a i n the i n c r e a s e d
susceptibility of such p y r i m i d o n e s to electrophilic attack.
N
HNQ3
^
N
°2-vj^N
Heat
N ^ O
H
^ N ^ O
H
10.20
10.21
2. B a r b i t u r a t e s ( p y r i m i d i n e t r i o n e s such as 1 0 . 2 5 ) used to b e w i d e l y u s e d as
s e d a t i v e s , but h a v e n o w largely b e e n s u p e r s e d e d by d r u g s with f e w e r sidee f f e c t s . S u g g e s t a synthesis of 10.25.
O ^ N ^ O
H
10.25
3. T h e r e are several p r e p a r a t i o n s of c y t o s i n e 1 0 . 6 available, o n e of w h i c h is
the c o n d e n s a t i o n of nitrile 1 0 . 2 6 with u r e a 10.16. P r o p o s e a m e c h a n i s m f o r
this reaction.
NHCN
H
NH
2
I
,T X T x t s - n
H2N
O
OEt
\
OEt
10.26
HCL
1
H , 0 / EtOH
10.16
10.6 References
Brown, D.J.
compounds
( 1 9 6 2 ) . In The pyrimidines
(The
chemistry
of
heterocyclic
[ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , V o l . 16). W i l e y
Interscience, N e w York.
Brown, D.J.
compounds
(1970).
In The pyrimidines
(The
chemistry
of
heterocyclic
(ed. A . W e i s s b u r g e r a n d E . C . T a y l o r ] , V o l . 16, S u p p l e m e n t s 1
a n d 2). W i l e y Interscience, N e w Y o r k .
F u r n i s s , B . S . , H a n n a f o r d , A.J., S m i t h , P . W . G . , a n d T a t c h e l l , A . R .
Vogel's
textbook
of practical
organic
chemistry
(1989).
(5th e d n ) , p . 1 1 7 7
(preparation of barbiturate 10.25). L o n g m a n , H a r l o w .
H u r s t , D . T . ( 1 9 8 0 ) . An introduction
pyrimidines,
purines,
and pteridines.
to the chemistry
and biochemistry
Wiley, New York.
of
11. Answers to problems
11.1 Answers to problems in Chapter 2
Note that the reaction p r o c e e d s
with attack of the a m i n o g r o u p o n
t h e least h i n d e r e d k e t o n e .
O
1. R e a c t i o n of d i k e t o n e 2 . 4 6 w i t h a m i n o k e t o n e 2 . 4 7 p r o d u c e s e n a m i n e 2 . 4 8
w h i c h is n o t i s o l a t e d , b u t c y c l i s e s directly to g i v e p y r r o l e 2 . 4 3 .
o
MeN
MeN
4-h2O
H9N
H
2.46
2.47
2. T h e l o n e p a i r of e l e c t r o n s of 2 . 4 4 is d e l o c a l i s e d on to t h e c a r b o n y l g r o u p
as s h o w n , i n c r e a s i n g t h e e l e c t r o n d e n s i t y at t h e a l d e h y d i c c a r b o n a t o m . T h i s
r e n d e r s it less r e a c t i v e to n u c l e o p h i l i c attack.
e
N
H
FFIN
H
H
H
2.44
U n d e r a c i d i c c o n d i t i o n s a l c o h o l 2 . 4 5 r e a d i l y g i v e s c a t i o n 2 . 4 9 a , b w h i c h is
s t a b i l i s e d b y a s i m i l a r d e l o c a l i s a t i o n of t h e n i t r o g e n l o n e p a i r .
-H,0
H
OH"
OH7
fn
kx]
H
2.45
2.49a
2.49b
T h i s h i g h l y e l e c t r o p h i l i c s p e c i e s t h e n r e a c t s w i t h a l c o h o l 2 . 4 5 to g i v e d i m e r
2 . 5 0 . R e p e t i t i o n of this p r o c e s s l e a d s t o p o l y m e r i c m a t e r i a l .
OH-
OH
N'
H
®
polymer
"N
H
2.45
2.50
3. A s d i s c u s s e d in C h a p t e r 2 , i n t e r c e p t i o n of c a t i o n 2 . 3 1 w i t h a n u c l e o p h i l i c
counterion
such
as acetate p r o d u c e s
the 2,5-addition
product
T e t r a f l u o r o b o r a t e is a n o n - n u c l e o p h i l i c c o u n t e r i o n a n d h e n c e t h e
2.32.
only
p a t h w a y a v a i l a b l e to 2 . 3 1 is loss of a p r o t o n to g i v e n i t r o f u r a n 2 . 3 3 d i r e c t l y .
Direct loss of
H
0
4. T h e m e c h a n i s m is a straightforward F r i e d e l - C r a f t s acylation.
11.2 Answers to problems in Chapter 3
1. A p p l i c a t i o n of o u r g e n e r a l i s e d o x a z o l e r e t r o s y n t h e s i s leads to a simple
glycine derivative.
H
Z
T h e f o r w a r d synthesis is s h o w n b e l o w :
ur
2. T h e m e c h a n i s m of this o x a z o l e f o r m a t i o n is identical to that of the
H a n t z c h thiazole synthesis. H o w e v e r , b e c a u s e of the reduced nucleophilicity
of a c a r b o n y l g r o u p as c o m p a r e d to a t h i o c a r b o n y l ( d u e to t h e h i g h e r
e l e c t r o n e g a t i v i t y of o x y g e n ) , this s y n t h e s i s only p r o c e e d s u n d e r v i g o r o u s
conditions (high temperatures, a m i d e c o m p o n e n t as solvent, etc).
T h e alternative s e q u e n c e w o u l d g i v e a positional isomer of oxazole 3.44.
3.
B r o m i n a t i o n o f 3 . 4 5 g i v e s a b r o m o k e t o n e w h i c h is c o n d e n s e d
with
t h i o u r e a to g i v e a m i n o t h i a z o l e e s t e r 3 . 4 7 . T h i s is t h e n h y d r o l y s e d to a c i d
3.46.
CO,R
CO,Et
Br,
MeO.
"N
MeO
NH,
3.45
3.46 R = H
K e t o n e 3 . 4 5 i t s e l f is r e a d i l y p r e p a r e d b y n i t r o s a t i o n of e t h y l a c e t o a c e t a t e
followed by O-methylation.
CO,Et
C0 2 Et
0 = N-0C,H,
C0 2 Et
McI
. HO.
K,CO,
AcOH
O
^ MeO,
1>
N
3.45
11.3 Answers to problems in Chapter 4
1. T h e r e a c t i o n is a M i c h a e l a d d i t i o n f o l l o w e d b y e l i m i n a t i o n of c y a n i d e i o n .
H
\ „
e
N ; C— S
NTH
c K
s
0
3©x
H
J
NH,
~swJ "
NH 2
I
CN
NH
CN
-
S
rA."7 CN
R
4.39
2. T h e o v e r a l l s t r a t e g y is to p r o t e c t t h e n i t r o g e n of p y r a z o l e (as an a c e t a l ) ,
d e p r o t o n a t e , i n t r o d u c e t h e side c h a i n as an e l e c t r o p h i l e , then d e p r o t e c t .
N
H
HC(OMe)3
N
"BuLV
N
Li® © <
N"
4.40
Ar
3.
jn.N
HC1/H 2 0
N'
Ar f j
N
.C-OMe
OMe
H .C-OMe
OMe
oh
N
OH
\\ P- LAr2CO
„N
2.NH4CI / H 2 0
//
At
Ar
'
„ . C-OMe
H
\
OMe
O x i d a t i o n o f o x i m e 4 . 4 1 p r o d u c e s n i t r i l e o x i d e 4 . 4 6 w h i c h c y c l i s e s to
isoxazole 4.47.
N®0
NaOCl
HO,
^N
)
V
[3+2]
NaOH
4.41
4.46
4.47
4. R e a c t i o n w i t h h y d r o x y l a m i n e o c c u r s o n t h e a l d e h y d e g r o u p of t h e m o r e
reactive minor tautomer 4.43 affording isoxazole 4.44. Methoxide-induced
f r a g m e n t a t i o n as s h o w n g i v e s e n o l a t e 4 . 4 8 w h i c h is q u e n c h e d b y a p r o t o n in
t h e w o r k u p to a f f o r d 2 - c y a n o c y c l o h e x a n o n e 4.45.
4.45
4.48
11.4 A n s w e r s to problems in Chapter 5
1. T h e p r o c e s s is e s s e n t i a l l y a n a l o g o u s to a M i c h a e l r e a c t i o n .
CO,Et
CO,Et
I
^
^
CO,Et
©
i
e
COoEt
(— OEt)
CO,Et
2. A r e a s o n a b l e m e c h a n i s m is:-
H.
H
-H
1
-H
N
Ac,0
U
Zc.
O
- Ac,0
O J ^
3.
HOAc
O
P y r i d y l a m i d e 5 . 3 9 is e a s i l y m e t a l l a t e d at t h e C 3 p o s i t i o n . Q u e n c h i n g
w i t h t h e a l d e h y d e , a n d c y c l i s a t i o n of t h e r e s u l t i n g a l c o h o l 5 . 4 2 o n t o t h e
a m i d e group, produces lactone 5.40.
4.
T h e r e a c t i o n p r o b a b l y p r o c e e d s via
enaminoester formation
then
cyclisation.
Fo
'C
In f a c t n u c l e o p h i l i c substitution of p y r i d i n e N - o x i d e s o c c u r s m o r e easily
t h a n o n s i m p l e p y r i d i n e s , as the nitrogen a t o m is positively c h a r g e d .
11.5 A n s w e r s to problems in Chapter 6
1. T h e q u i n o l o n e s y n t h e s i s i n v o l v e s an a d d i t i o n - e l i m i n a t i o n
f o l l o w e d b y an intramolecular aromatic acylation.
reaction
T h e d i s p l a c e m e n t reaction occurs b y initial nucleophilic attack on the
benzenoid ring (with the negative charge being delocalised onto the oxygen
a t o m as s h o w n ) t h e n e l i m i n a t i o n of c h l o r i d e i o n . T h e p r e s e n c e of t h e f l u o r i n e
s u b s t i t u e n t is e s s e n t i a l f o r t h i s d i s p l a c e m e n t , a c t i v a t i n g t h e r i n g t o w a r d s
n u c l e o p h i l i c attack b y its e l e c t r o n - w i t h d r a w i n g i n d u c t i v e e f f e c t .
CO 2 H
2 . ' S t e p 1. C o n d e n s a t i o n o f t h e a l d e h y d e w i t h n i t r o m e t h a n e u n d e r b a s i c
conditions produces the a,(3-unsaturated nitro c o m p o u n d .
£
O
Ar
OH
foil
_NO2 ^
„
Ar
H
'CH,NO,
H
NO,
H
OH
Step 2. L i t h i u m a l u m i n i u m h y d r i d e w a s u s e d , a l t h o u g h h y d r o g e n a t i o n c a n
a l s o e f f e c t this t y p e of r e d u c t i o n .
LiAIH4
NH,
NO,
Ar'
Step
Ar
H,/Pd°
3. A c y l a t i o n of t h e a m i n e w i t h a n a c i d c h l o r i d e in t h e p r e s e n c e o f a n
appropriate base gave the amide.
Ar
NH,
RCH2COCl
NEt,
Ar
O
Step 4. T h i s i s o q u i n o l i n e f o r m a t i o n is of c o u r s e a n e x a m p l e of t h e B i s c h l e r Napieralski synthesis, although p h o s p h o r u s trichloride was actually u s e d in
this e x a m p l e , n o t p h o s p h o r u s o x y c h l o r i d e .
Step 5. S o d i u m b o r o h y d r i d e w a s u s e d t o r e d u c e t h e i m i n e to t h e a m i n e .
r
R,.
^NR3
H
NaBH,
R^NR
R2
H
3
R2
Step 6. T h e c a t e c h o l i c a n d p h e n o l i c e t h e r s w e r e r e m o v e d b y t r e a t m e n t w i t h
h y d r o b r o m i c a c i d . B e n z y l e t h e r s a r e f r e q u e n t l y r e m o v e d b y r e d u c t i o n (e.g.
h y d r o g e n a t i o n ) b u t r e d u c t i o n , of c o u r s e , w o u l d n o t r e m o v e t h e m e t h y l e t h e r .
T h e m e c h a n i s m of t h e d e p r o t e c t i o n is s h o w n b e l o w .
R
OH
H
^©Br
11.6 A n s w e r s to problems in Chapter 7
1. I n d o l e 7 . 3 8 w a s p r e p a r e d b y a F i s c h e r i n d o l e synthesis f o l l o w e d b y Nalkylation as s h o w n .
3. A s i n t e n d e d , C 2 c a r b a n i o n 7 . 4 7 a t t a c k e d t h e nitrile g i v i n g 7 . 4 8 , w h i c h
u n e x p e c t e d l y a t t a c k e d the a d j a c e n t s u l p h o n y l g r o u p g i v i n g indolyl a n i o n
7 . 4 9 . D u r i n g t h e a c i d i c a q u e o u s w o r k u p this a n i o n is q u e n c h e d a n d the
r e a c t i v e N - s u l p h o n y l i m i n e f u n c t i o n a l i t y is r e a d i l y h y d r o l y s e d a f f o r d i n g
7.45
FH
11.7 A n s w e r s to problems in Chapter 8
1. S u l p h u r ylid 8 . 3 6 is the k e y i n t e r m e d i a t e in the f o r m a t i o n of e p o x i d e
8.34. E p o x i d e 8 . 3 4 is r a c e m i c but alcohol 8 . 3 5 is achiral.
c,
O
H
CH3-S(CH3)2
©
NaH
p.
>
(-H2)
U
O
H
CH2-S(CH3)2
e
8.36
Coreysylid
2.
T h i s o x a d i a z o l e f o r m a t i o n i n v o l v e s O - a c y l a t i o n of the
amidoxime
followed by a condensation.
r o
ETO-HA-N.
R
"
NH,
OMe
->
NH,
8.21
/
HO
N-
-H2O
O
M
O-N
\\
N
8.22
11.8 Answers to Problems in Chapter 9
1. T h e m e c h a n i s m is s i m i l a r to t h e 4 - p y r o n e e x a m p l e .
-H2O
9.2
2.
-
OH/^O
NH 2
NH 3
> o V ^
HH2N
HO
- O
T h e f i r s t s t a g e is t h e s a m e as t h e p r e p a r a t i o n of 9 . 3 0 , t h e n c y c l i s a t i o n
a f f o r d s the p y r a z o l e .
H
N
9.33
OH
3. T h i s is a M a n n i c h r e a c t i o n ( s e e C h a p t e r 2 ) a n d is a n u n u s u a l e x a m p l e of
an electrophilic substitution on a chromone.
O
FF,
J"
9.34
4.
o
O
, CH 2 =NMe 2
^•
•
OMe
NMe,
OMe
OMe
9.35
T h e r e a c t i o n is a s t r a i g h t f o r w a r d a c i d - c a t a l y s e d c o n d e n s a t i o n , p a s s i n g
t h r o u g h c a r b o n i u m ion 9 . 3 6 a , b .
O
OH
O
P£
°
-H
O
9.19
Ph
9.36b
N
H
O
"N
O
H
5.22
11.9 A n s w e r s to problems in Chapter 10
1. T h e o v e r a l l e l e c t r o n i c d i s t r i b u t i o n of 2 - p y r i m i d o n e h a s a c o n s i d e r a b l e
contribution f r o m m e s o m e r 1 0 . 2 0 a .
10.20
10.20a
T h e m e c h a n i s m of nitration is s h o w n b e l o w .
2. D i s c o n n e c t i o n of b a r b i t u r a t e 1 0 . 2 5 p r o d u c e s b i s - e l e c t r o p h i l e 1 0 . 2 7 and
urea. In practice m a l o n a t e ester 1 0 . 2 8 (X = O E t ) is used.
3. H y d r o l y s i s of acetal 10.26 leads to reactive aldehyde 10.29 in situ.
C o n d e n s a t i o n of a l d e h y d e 1 0 . 2 9 with u r e a f o l l o w e d b y cyclisation o n t o t h e
nitrile p r o d u c e s cytosine 10.6. O b s e r v e h o w cyclisation o n t o a nitrile a f f o r d s
the a m i n o f u n c t i o n a l i t y directly, as c o m p a r e d with the three step s e q u e n c e
u s e d in t h e synthesis of 1 0 . 2 4 w h e r e an ester is used in the cyclisation step.
Index
a-effect 29
acetyl nitrate 15
acid chlorides 6
a-aminoketone 13
ammonia 5
AMP, biosynthesis of 74
anion chemistry of
furan 17
imidazole 25
indole 59
isoquinoline 50
isothiazole 32
isoxazole 32
pyrazole 32
pyridine 42
pyrrole 17
quinoline 50
thiazole 25
anthocyanins 67
aryl hydrazines 54
1.2 azoles 28
1.3 azoles 20
A Z T 74
barbiturates 77
benzene 2
benzopyrilium cation 67
bioisosteric replacement 63
Bischler-Napieralski synthesis
(isoquioline) 48
Chichibabin reaction 40
chlorophyll 11
chromone 67
Claison rearrangement 55
condensation 3
Cope rearrangement 55
coumarin 67
cytosine 73
delocalisation 3
delphinidin chloricje 67
dihydropyridines 37
disconnection 4
D N A 73
drugs for the treatment of
A I D S 74
asthma 31
bacterial infection 27
depression 60
fungal infection 41, 66
inflammation 22
schizophrenia 18
senile dementia 45,63
sleep disorders 77
trematode infection 69
ulcers 1,11
electrophilic substitution of
furan 14
imidazole 24
indole 57
isoquinoline 49
isothiazole 32
isoxazole 32
oxazole 24
pyrazole 32
pyridine 37
pyridine N-oxide 38
pyridones 39
pyrimidones 76
pyrrole 14
quinoline 49
thiazole 24
thiophene 14
Fischer synthesis (indole) 54
flavone 70
furan 10
Gould-Jacobson synthesis
(quinolone) 51
Hantzsch pyridine synthesis 36
Hantzsch thiazole synthesis 23
heteroaromaticity 1
histamine 20
hydrazine 29
hydrogen sulphide 12
hydroxylamine 29
5-hydroxytryptamine 54
imidazole 20
imidoyl halide 22
indole 53
isoquinoline 46
isothiazole 28
isoxazole 28
Khellin 67
Knorr synthesis (pyrrole) 14
Konstanecki-Robinson synthesis
(chromone) 70
Leimgruber synthesis (indole) 56
lysergic acid 54
Mannich reaction 16,58
neurotransmitters 54
nitrile oxides 30
nucleic acids 73
nucleophilic substitution of
imidazoles 26
isoquinolines 49
oxazoles 26
pyridines 40
pyrimidines 76
quinolines 49
thiazoles 26
ornithine 64
ortho-activating substituents 43
oxadiazoles 61
oxazoles 20
oxazolidinones 8
Paal-Knorr synthesis (pyrrole,
thiophene, furan) 12
phosphorus oxychloride 15, 47
phosphorus sulphide 12
Pictet-Spengler synthesis
(isoquinoline) 48
pyrazole 28
purine 73
pyridine 35
pyridine N-oxide 38
pyridine sulphur trioxide complex 15,
35
pyridones 39
pyridontriazine 41
pyrylium cation 67
pyrimidines 73
pyrones 67
pyrrole 10
Quinoline 46
resonance 2
retrosynthesis 4
Robinson-Gabriel synthesis
(oxazole) 21
Skraup synthesis (quinoline) 46
synthesis of
chromones 70
coumarins 69
furans 11
heterocycles, principles of 3-8
imidazoles 22
indoles 54
isoquinolines 46
isothiazoles 29
oxadiazoles 62
oxazoles 21
pyrazoles 29
pyridines 35
pyrylium salt 68
pyrimidines 74
pyrroles 11
quinolines 46
tetrazoles 64