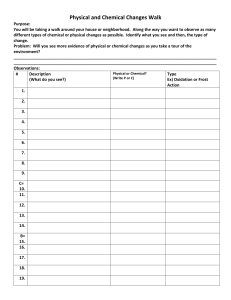
REDOX Worksheet 1. In which substance is oxidation number of nitrogen zero? a. b. c. d. NH3 N2 NO2 N2O a. b. c. d. 2. In the reaction Al + Cr3+ Al3+ + Cr, the reducing agent is? a. b. c. d. 3. What is the oxidation number of carbon in NaHCO3? 4. As an S2- ion is oxidised to an S atom, the number of protons in its nucleus Al Cr3+ Al3+ Cr a. b. c. d. 5. If element X forms the oxides XO and X2O3, the oxidation numbers of element X are: a. +1 and +2 c. +1 and +3 b. +2 and +3 d. +2 and +4 6. Oxygen has a positive oxidation number in the compound: a. H2O c. OF2 b. H2O2 d. IO2 +6 +2 -4 +4 Increases Decreases Stays the same Yes 8. Calculate the oxidation number of chromium in each of the following: a. Cr2O3 b. Na2Cr2O7 c. CrSO4 7. What is the oxidation number of sulfur in H2SO4 a. 0 c. +6 b. -2 d. +4 d. CrO42- (chromate ion) e. Cr2O72- (dichromate ion) REDOX Worksheet 1. In which substance is oxidation number of nitrogen zero? a. b. c. d. NH3 N2 NO2 N2O a. b. c. d. 2. In the reaction Al + Cr3+ Al3+ + Cr, the reducing agent is? a. b. c. d. 3. What is the oxidation number of carbon in NaHCO3? 4. As an S2- ion is oxidised to an S atom, the number of protons in its nucleus Al Cr3+ Al3+ Cr a. b. c. d. 5. If element X forms the oxides XO and X2O3, the oxidation numbers of element X are: a. +1 and +2 c. +1 and +3 b. +2 and +3 d. +2 and +4 6. Oxygen has a positive oxidation number in the compound: a. H2O c. OF2 b. H2O2 d. IO2 +6 +2 -4 +4 Increases Decreases Stays the same Yes 8. Calculate the oxidation number of chromium in each of the following: a. Cr2O3 b. Na2Cr2O7 c. CrSO4 7. What is the oxidation number of sulfur in H2SO4 a. 0 c. +6 b. -2 d. +4 d. CrO42- (chromate ion) e. Cr2O72- (dichromate ion)