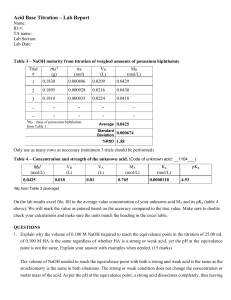
CHM 110 Lab 9 Lab Titrations Lab Report Worksheet Lab 9 Part A: Standardization of NaOH Acid-Base Titration Simulation Activity Open the acid-base titration simulation by clicking on the following link https://pages.uoregon.edu/tgreenbo/acid_base.html This simulation requires that flash be enabled. You will have to tell your web browser to allow flash. For google chrome that is done by going into your browser setting, go to site settings and make sure the option for flash is set to “ask first”. You then have to click on the simulation screen and tell your browser to allow flash. You may see a pop up asking if you want it to allow flash or you might have to click on the flash icon. To run the simulation you will need to follow the following steps in exact order. They are labeled as such on the simulation. 1. Select Type of Reaction a. For this activity we will be looking at Strong Acid vs Strong Base Titration reactions. Select that option. 2. Fill the Burette with a. Select Base 3. Select the acid and Base a. Click on the box. It will then display a column of Acids and Bases. In the problems below you we will asked to select certain acid and base pairs. 4. Select the Indicator a. Select Phenolphthalein 5. Push slider up to add a Volume of a. Pushing the slide is just like opening your burette up all the way, larger volumes will come out more quickly. Recall than in a titration we typically add volumes quickly until we start to see pink show up and disappear. We then add slowly until the pink remains for at 30 seconds. You will see the same thing in the simulation. When you start to see pink starting adding dropwise. If you add too much volume on the first try reset the simulation. 6. You will run several trials to get an average and calculate error and deviation. The simulation starts off by giving you a random volume and molarity of your acid. Running multiple trials should be done with the same concentration. To make sure you have the same acid concentration for each trial you will want to select the Concordant Values button in between trials. This will refill the burette and keep the same acid concentration. Experiment: 1. Use the titration simulation to work through a titration of HCl and NaOH which react according to the following balanced equation 𝐻𝐶𝑙 + 𝑁𝑎𝑂𝐻 → 𝑁𝑎𝐶𝑙 + 𝐻2 𝑂 a. The simulation will give you the known volume and concentration of your acid. It will be located next to the bottom of the Erlenmeyer flask. Record all data in the data table below. Use the volume of the base added along with acid volume and concentration information to determine the M of your base. Check your answer by entering it into block 6 in the simulation and selecting OK. Table 1: Titration of NaOH with a standard HCl Solution Volume of HCl Molarity of HCl Volume of NaOH used from Burette Moles of NaOH M of NaOH Deviation Trail 1 25.00 mL .1184 M 14.86 mL Trial 2 25.00 mL .1184 M 14.86 mL Trial 3 25.00 mL .1184 M 14.86 mL .002968 mol .1662M 0 .002968 mol .1662 M 0 .002968 mol .1662 M 0 Average M of NaOH: ________.1662 M___________ b. Show your work for your Molarity Calculation below. To do this in Microsoft word you will need to go to insert and select equation. A new equation menu should open up. Set up any fractions using the fraction menu option. Subscripts and superscripts can be accessed using the ex option. If you have the ability to write you on a touch screen you may do that as well. Otherwise you should scan or take a phot of hand written work and insert below. 2. Use the titration simulation to work through a titration of H2SO4 and NaOH which react according to the following balanced equation 𝐻2 𝑆𝑂4 + 2𝑁𝑎𝑂𝐻 → 𝑁𝑎2 𝑆𝑂4 + 2𝐻2 𝑂 a. The simulation will give you the known volume and concentration of your acid. It will be located next to the bottom of the Erlenmeyer flask. Record all data in the data table below. Use the volume of the base added along with acid volume and concentration information to determine the M of your base. Check your answer by entering it into block 6 in the simulation and selecting OK. Table 2: Titration of NaOH with a standard H2SO4 Solution Volume of H2SO4 Molarity of H2SO4 Volume of NaOH used from Burette Moles of NaOH M of NaOH Deviation Trail 1 25.00 mL .2181 M 41.06 mL Trial 2 25.00 mL .2181 M 41.06 Trial 3 25.00 mL .2181 41.06 .0010905 mol .02656 M 0 .0010905 .02656 0 .0010905 .02656 0 Average M of NaOH: ______.02656 M_____________ b. Show your work for your Molarity Calculation below. To do this in Microsoft word you will need to go to insert and select equation. A new equation menu should open up. Set up any fractions using the fraction menu option. Subscripts and superscripts can be accessed using the ex option. If you have the ability to write you on a touch screen you may do that as well. Otherwise you should scan or take a phot of hand written work and insert below. Part B: Volumetric Analysis of Vinegar (Acetic Acid) In Part B of this experiment you will collect data by watching the following video. You will need to pause periodically to record the measurements. You will calculate both the molarity and the mass percent of the vinegar solution. The manufacturer claim is that the Vinegar solution is 5.0% by mass. You will use this as your true value to calculate the percent error and make comparisons to the manufacturer claim. All work should be shown in the area below the data table. 1. This reaction occurs according to the following balanced chemical equation: 𝑁𝑎𝑂𝐻(𝑎𝑞) + 𝐶𝐻3 𝐶𝑂𝑂𝐻 → 𝐶𝐻3 𝐶𝑂𝑂𝑁𝑎(𝑎𝑞) + 𝐻2 𝑂(𝑙) 2. In this case the NaOH is the standard solution and it’s Molarity is known. Record the Molarity given in the table below. 3. Record the initial burette reading in the table below 4. Record the mass of the empty Erlenmeyer flask. 5. Record the mass of the Erlenmeyer flask with Vinegar in it. 6. Determine the mass of just the vinegar solution and record in the table below. 7. Record the final burette reading after NaOH has been added to the endpoint 8. Determine the volume of NaOH used to titrate the acetic acid 9. Use the volume and concentration of the NaOH added to determine the moles of NaOH added. 10. Based on the sodium hydroxide added, determine the moles of HC2H3O2 present in the vinegar solution 11. Determine the mass percent of HC2H3O2 in the vinegar solution 12. Determine the molarity of the vinegar solution 13. Calculate the deviation and percent error 14. Answer the questions at the end Table 2: Titration of standard NaOH with a HC2H3O2 Solution M of NaOH Volume of Vinegar Solution Mass of Erlenmeyer flask: Mass of Erlenmeyer flask plus vinegar solution: Mass of Vinegar solution Initial Burette Reading: Final Burette Reading: Volume of NaOH used: Moles of NaOH added: Moles of HC2H3O2 Mass of HC2H3O2 Mass percent of Vinegar solution Molarity of HC2H3O2 Deviation Percent error Trail 1 .4936 M 25.00 mL 134.347 g 159.248 g Trial 2 .4936 M 25.00 mL 134.347g 159.428g Trial 3 .4936 M 25.00 mL 134.347g 159.428g 25.081g 1.4 mL 44.3mL 42.9 mL .0212 mol .0212 mol 1.24 g 5.06% 25.081g 1.6 mL 44.4 mL 42.8 mL .0211 mol .0211 mol 1.24 g 5.06% 25.081g 1.2mL 44.3 mL 43.1 mL .0213 mol .0213 mol 1.28g 5.10% .848 M 0 1.2% .844 M 0 1.2% .852 M .04 2.0% Average Mass Percent of Vinegar:____5.06%_________ Average Molarity of Vinegar: _____________.848 M__ Show your work for your calculations below. To do this in Microsoft word you will need to go to insert and select equation. A new equation menu should open up. Set up any fractions using the fraction menu option. Subscripts and superscripts can be accessed using the ex option. If you have the ability to write you on a touch screen you may do that as well. Otherwise you should scan or take a phot of hand written work and insert below. Problems: 1. Determine the molarity of a phosphoric acid solution if 20.00 mL of phosphoric acid was titrated with 15.9 mL of 0.200 M sodium hydroxide. You will need a balanced chemical equation. Show work! M1×20.00 mL=0.200 ×15.9 mL M1=0.200×15.9 mL/20.00 =0.159 M 2. Why must NaOH be standardized? Solid NaOH is very hygroscopic, that is it absorbs water from the air, so it is very hard to weigh exactly. That is why NaOH must be standardized 3 In part B, how did the mass percent you found compare to the manufacturer claim. What can we conclude about the manufacturers claim? Explain by supporting with experimental evidence. Compared to the manufacturer claim and what I found it was very close only off by .06 percent. We canclaim that the manufacturers claim is accurate because the mass percent I obtained was so close to 5.00 %.With a percent error of 1.2 % and 2 % one can claim that the 5.00 % the manufacturers give us is prettyaccurate and could be accountable 4 In the Volumetric Analysis of Acetic Acid Video the demonstrator did something that is generally considered poor chemical hygiene. Meaning he created opportunity for contamination. What was it, and what should he have done instead? When using the volumetric pipet to obtain the vinegar solution he inserted the pipet into the original container and when measuring too much would put it back in the original container. Instead he should oftransfers some of the vinegar solution into I separate container to obtain it and when obtaining too much should’ve had a separate container to dispose of the extra.