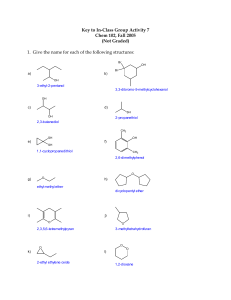
Species Protecting Group (PG) or Reagent (R) Use(s) Notes acetate PG Ester PG removed with OH- benzoate pivalate PG PG Ester PG Ester PG removed with OHreact with 1° alcohol selectively TMS (trimethyl silyl ether) TBS (TBDMS, tert-butyl dimethyl silyl ether) TBDPS (tert-butyl diphenyl silyl ether) PG Silyl Ether PG PG Silyl Ether PG PG Bn (benzyl ether) PMB (para-methoxy benzyl ether) PG PG Tr (triphenyl methyl or trityl ether) Boc (t-butyloxycarbonyl) PG PG Silyl Ether PG Benzyl Ether PG/Amine PG Benzyl Ether PG Benzyl Ether PG/Amine PG Carbamate PG removed by acid, F- ion, or MeOH/NaHCO3 removed by (AcOH, H2O,THF) or F- ion; preferential to 1° alcohol only removed by F- ion; even more preferential to 1° alcohol Cbz (carboxybenzylcarbonyl) Fmoc (fluorenylmethyloxycarbonyl) PG PG Carbamate PG Carbamate PG Ts (para-toluenesulfonyl) PG Sulfonamide PG removed with H2/Pd-C removed with amine base (either morpholine or piperidine) add with 2 equivalents of Ts-Cl; removed with Na, naphthalene Ns (para-nitrophenylsulfonyl) PG Sulfonamide PG removed with PhSH/K2CO3 NaBH4 R reducing agent will reduce aldehydes and ketones, but not esters LiBH4 R will reduce esters LiAlH4 R reducing agent stronger reducing agent will reduce aldehydes, ketones, and esters pyridine (C5H5N) R base/solvent collect H+ ions formed during reaction Et3N (triethylamine) R-MgX THF (tetrahydrofuran) R R R base Grignard Reagent solvent collect H+ ions formed during reaction R= alkyl, allyl, or aryl group; X=halide CH2Cl2 DMF (dimethylformamide) R R solvent solvent removed with DDQ removed with TFA removed with TFA Imidazole (C3H4N2) R base produces mildly basic solution TBAF (n-Bu4NF) Fluoride ion R R salt salt TBS-halide R salt TBDPS-halide NaH Ph-Br (Bn-Br, benzyl bromide) PMB-Cl Tr-Cl R R R R R salt salt salt salt salt H2/Pd-C R hydrogen source H2/Pd(OH)2-C (Pearlman's Catalyst) TFA (trifluoroacetic acid) DDQ (dichloro-dicyanobenzoquinone) chloromethyl ether R R hydrogen source acid provide F- ions removes all silyl ether PG adds TBS PG; also is a Lewis Acid (provides H+ ions) adds TMS PG; also is a Lewis Acid (provides H+ ions) adds TBDPS PG; also is a Lewis Acid (provides H+ ions) deprotonates alcohols adds Bn PG adds PMB PG adds trityl PG saturates multiple bond; removes benzyl ether PG or Cbz PG saturates multiple bond; removes benzyl ether PG or Cbz PG removes trityl PG or Boc PG R R oxidant HOCH2CH2OH (ethylene glycol) R used to create carbonyl acetal PG TMSOCH2CH2OTMS R used to create carbonyl acetal PG NaIO4 TBSOTf R R salt salt cleaves a 1,2 diol into an aldehyde and ketone adds TBS to 2° alcohol! 2,6-lutidine R base collect H+ ions formed during reaction Ph3P/Halogen R Appel Reaction (MeO)2CMe2 PCC (pyridinium chlorochromate) R R carbocation creator milder oxidizing agent will replace alcohol group with halide group creates acetonide (carbocations latch onto neighboring alcohol groups) oxidizes 1° alcohol to aldehyde TsOH (para-toluenesulfonic acid) R acid Ph3P=CH© R Wittig Reaction TMS-halide removes only PMB PG used to create ether acetal PG provides H+ ions for reaction turns carbonyl group into alkene and adds group represented by ©