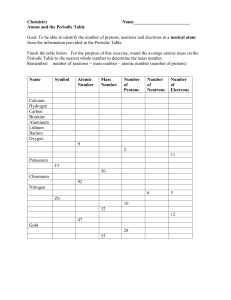
Protons, Neutrons, and Electrons Practice Worksheet Fill in the blanks in the following worksheet. Please keep in mind that the isotope represented by each space may NOT be the most common isotope or the one closest in atomic mass to the value on the periodic table. Atomic symbol Atomic number Protons B Neutrons Electrons Atomic mass 6 11 24 31 37 39 29 89 35 43 100 207 Pb 102 89 Mo 70 225 53 81 100 206 159 No Yb 261 172 106 159 Finding Protons, Neutrons, and Electrons of Elements One of the things students frequently have problems with is determining how many protons, neutrons, and electrons an element has, based only on some sparse information given to you on the periodic table or in a problem. This review will give you everything you need to figure it out. First, let's examine one of the entries in the periodic table: This is what the entry for helium is on a normal periodic table. The atomic number is found at the top, and is equal to the number of protons that an element has. The reason that all atoms of the element have the same number of protons is that the number of protons determines what element is present - if there were a different number of protons, you would have a different element. For neutral atoms, the atomic number is also equal to the number of electrons present because the number of electrons equals the number of protons in neutral atoms. The atomic symbol is different for every element. As you might guess, the atomic symbol and atomic number are very closely related to one another. After all, any element that has the atomic number "2" will have the atomic symbol "He". As a result, if you don't know the atomic number of an element, you can look up the atomic symbol on the periodic table to find it. As a result, you can determine the number of protons an element has from the atomic symbol if you have a periodic table handy. The atomic mass is also different for every element, and also for isotopes of the same element. The atomic mass is equal to the number of protons that an element has plus the number of neutrons that an element has. This is because protons have a mass of approximately 1 amu (atomic mass unit) and neutrons have a mass of approximately 1 amu. Electrons don't count in this calculation, because their mass is small enough that it can usually be ignored. Atomic number = number of protons (which, for neutral atoms, is equal to the number of electrons). Atomic symbol allows us to find the atomic number because you can just look it up on the periodic table. Atomic mass = number of protons + number of neutrons.