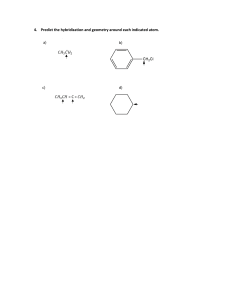
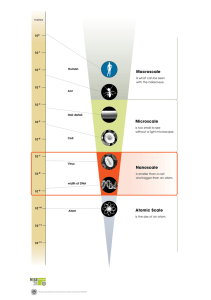
PERIODIC TRENDS IN THE P E R I O D I C TA B L E P E R I O D I C T R E N D S I N T H E P E R I O D I C TA B L E AT O M I C R A D I U S I n c r e a s e s Decreases 1 2 1 2 3 4 5 6 7 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 P E R I O D I C T R E N D S I N T H E P E R I O D I C TA B L E I O N I Z AT I O N E N E R G Y D e c r e a s e s Increases 1 2 1 2 3 4 5 6 7 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 P E R I O D I C T R E N D S I N T H E P E R I O D I C TA B L E E L E C T R O N E G AT I V I T Y D e c r e a s e s Increases 1 2 1 2 3 4 5 6 7 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 P E R I O D I C T R E N D S I N T H E P E R I O D I C TA B L E IONIC RADIUS • Cations are smaller than the atom they are formed from. • Anions are larger than the atom they are formed from.