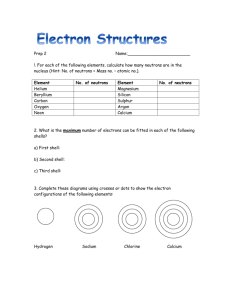
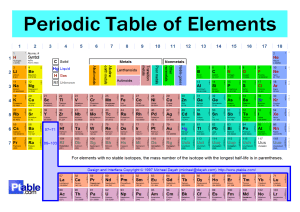
Q1.The diagram below represents different models of the atom. (a) Which diagram shows the plum pudding model of the atom? Tick one box. (1) (b) Which diagram shows the model of the atom developed from the alpha particle scattering experiment? Tick one box. (1) (c) Which diagram shows the model of the atom resulting from Bohr’s work? Tick one box. (1) (d) Define the mass number of an atom. ___________________________________________________________________ ___________________________________________________________________ (1) P.T.O Page 1 of 6 Q2. Figure 2 shows the positions of sodium and chlorine in the periodic table. Figure 2 (a) State one difference and one similarity in the electronic structure of sodium and of chlorine. Difference __________________________________________________________ ___________________________________________________________________ Similarity ___________________________________________________________ ___________________________________________________________________ (2) (b) Sodium atoms react with chlorine atoms to produce sodium chloride (NaCl). In sodium chloride develop into ions. Describe what happens to sodium when it transition into ion. Draw the ion as part of your answer (the outer shell only). ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ (4) Page 2 of 6 Page 3 of 6 Page 4 of 6 Mark schemes Q1. (a) B 1 (b) C 1 (c) A 1 (d) sum of protons and neutrons allow number of protons and neutrons 1 (e) between 69.5 and 70.0 1 (f) Chadwick provided the evidence to show the existence of neutrons allow Chadwick discovered neutrons 1 (this was necessary because) isotopes have the same number of protons allow (this was necessary because) isotopes have the same atomic number or (this was necessary because) isotopes are atoms of the same element ignore isotopes have the same number of electrons 1 but with different numbers of neutrons allow but with different mass (numbers) 1 [8] Q1. (a) (difference) sodium has one and chlorine has seven electrons in outer level / shell or number of electrons number of electrons must be correct if quoted 1 (similarity) both have three / same number of levels / shells or have electrons in third level / shell or both have incomplete (outer) levels / shells Page 5 of 6 allow both have 2 electrons in inner shell or both have 8 electrons in second shell or both are one electron away from full outer level / shell Page 6 of 6