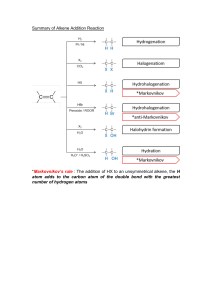
II. Homogeneous catalysis 1. Oxoprocess Oxo process, also known as hydroformylation or, is an industrial process for the production of aldehydes from alkenes. This chemical reaction entails the net addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. In other words it is the reaction of an alkene with carbon monoxide and hydrogen, catalyzed by cobalt or rhodium salts (Co or Rh catalyzed reaction) to form an aldehyde. For example, An olefin (e.g. propylene), H2, and carbon monoxide react together in presence of dicobalt-octacarbonyl to give butyraldehydes. Scheme 1 Structure of dicobalt-octacarbonyl: Figure 1 Many of the industrially useful compounds such as long carbon chain alcohols (detergents) were produced by hydroformylation. Mechanism of cobalt-catalyzed hydroformylation: The process begins with dissociation of CO from cobalt tetracarbonyl hydride A to give the 16-electron species (B, step 1). Subsequent binding of alkene gives an 18electron species (C, step 2). In step 3, the olefin inserts to give the 16electron alkyl tricarbonyl followed by the coordination of another equivalent of CO give alkyl tetracarbonyl (D, step 4). Migratory insertion of CO gives the 16 electron acyl which is followed by CO addition resulted the formation of intermediate E (step 5). In step 6, oxidative addition of hydrogen gives a dihydrido complex, which then releases aldehyde by reductive elimination. The mechanism of the reaction is shown in scheme 2. hydrogen migration acyl complex Addition of CO followed by CO insertion Scheme 2 2. Wacker Oxidation of alkenes The Wacker process or the Hoechst-Wacker process refers to the oxidation of ethylene to acetaldehyde in the presence of palladium(II) chloride as the catalyst (Scheme 3). This chemical reaction was one of the first homogeneous catalysis with organopalladium chemistry applied on an industrial scale. The catalyst is a two-component system consisting of palladium chloride, PdCl2, and copper chloride, CuCl2 (promotor). Note that all catalysts are regenerated and only the alkene and oxygen are consumed. Scheme 3 Note: β-Hydride elimination : It is a reaction in which an alkyl group bonded to a metal centre is converted into the corresponding metal-bonded hydride and an alkene. The alkyl must have hydrogens on the β-carbon (Scheme 4). Scheme 4 The mechanism of the reaction is shown in scheme 5. Scheme 5 3. Suzuki cross coupling reactions The Suzuki cross-coupling reaction is the organic reaction of an organohalide with an organoborane to give the coupled product using a palladium catalyst and base (Scheme 6). Scheme 6 The mechanism of the reaction is depicted in scheme 7. Scheme 7 4. Ziegler-Natta Catalyst It is a reagent or a mixture of reagents used in the production of polymers of 1- alkenes (α-olefins), Scheme 8. Ziegler-Natta catalysts are typically based on titanium compounds and organometallic aluminium compounds (Co-catalyst), for example triethylaluminium, (C2H5)3Al. In other words a typical Ziegler-Natta catalyst (ZN catalyst) system usually contains two parts: a transition metal (Group IV metals, like Ti, Zr, Hf) compound and an organoaluminum compound. The common examples of ZN catalyst systems include TiCl4 + Et3Al and TiCl3 + AlEt2Cl. Ziegler-Natta catalysts are used to polymerize terminal 1-alkenes. Scheme 8 Mechanism of Ziegler-Natta catalytic polymerization: (i) Activation of Ziegler-Natta catalyst Eg: TiCl4 + Et3Al The titanium chloride compound has a crystal structure in which each Ti atom is coordinated to 6 chlorine atoms. On the crystal surface, a Ti atom is surrounded by 5 chlorine atoms with one empty orbital to be filled. When Et3Al comes in, it donates an ethyl group to Ti atom and the Al atom is coordinated to one of the chlorine atoms. Meanwhile, one chlorine atom from titanium is kicked out during this process. Thus, the catalyst system still has an empty orbital (Scheme ( 9). ). The catalyst is activated by the coordination of AlEt3 to Ti atom. Scheme 9: The he activation of ZN catalyst system by coordination of AlEt3 to Ti atom. (ii) Initiation step The polymerization merization reaction is initiated by forming alkene-metal alkene metal complex. When a vinyl monomer like propylene comes to the active metal center, it can be coordinated to Ti atom by overlapping their orbitals. As shown in Figure 3,, there is an empty dxy orbital and a filled dx2-y2 orbital in Ti’s outermost shell (the other four orbitals are not shown here). The carbon-carbon carbon double bond of alkene has a pi bond, which consists of a filled pi pibonding orbital and an empty pi-antibonding pi antibonding orbital. So, the alkene's pi pi-bonding orbital and the Ti's dxy orbital come together and share a pair of electrons. Once they're together, that Ti’s dx2-y2 orbital comes mighty close to the pi-antibonding pi antibonding orbital, sharing another pair of electrons. Figure 3: Molecular orbitals representation of monomer coordinating to metal center. The formed alkene-metal complex (1) then goes through electron shuffling, with several pairs of electrons shifting their positions: The pair of electrons from the carbon-carbon pibond shifts to form Ti-carbon bond, while the pair of electrons from the bond between Ti and AlEt3’ ethyl group shifts to form a bond between the ethyl group and the methylsubstituted carbon of propylene (Scheme 10). After electron shuffling, Ti is back with an empty orbital again, needing electrons to fill it (2). Scheme 10: The formation process of alkene-metal complex. (iii) Propagation step: When other propylene molecules come in, this process starts over and over, giving linear polypropylene (Scheme 11). Scheme 11: Propagation towards polymeric chains. (iv) Termination step: It is the final step of a chain-growth polymerization, forming “dead” polymers (desired product, Scheme 12). The suggested mechanism of termination is shown by path (a) and (b). Scheme 12 Stereo-selectivity: Tacticity: The relative stereochemistry of adjacent chiral centers within a macromolecule is defined as tacticity. Stereochemistry of polymers made from ZN-catalyst can be well regulated by rational design of ligands. By using different ligand system, either syndiotactic or isotactic polymers can be obtained. Classification: Isotactic polymers: Substituents are located on the same side of the polymer backbone Figure 4 Syodiotactic polymers: Substituents located on alternative positions Figure 5 Atactic polymers: Substituents located randomly along the chain Figure 6