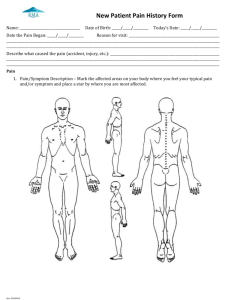
ADVERSE EVENT LOG ____________________________________________________________________________________________________________ Study Title: Protocol No: S.No. Subject ID Adverse Event Site ID: Serious1 Yes No Yes No Yes No Severity2 Mild Moderate Severe Lifethreatening Mild Moderate Severe Lifethreatening Mild Moderate Severe Lifethreatening Relationship to Inv. Agent Unrelated Possible Probably Definitely Unrelated Possible Probably Definitely Unrelated Possible Probably Definitely Project Code: Start Date/Time (dd/mm/yyyy) (24Hrs) Date: End Date/Time (dd/mm/yy yy) (24Hrs) Date: Time: Time: Date: Date: Time: Time: Date: Date: Time: Time: Investigator Initial and Date NOTE: Page 1 of 2 Comments3 ADVERSE EVENT LOG ____________________________________________________________________________________________________________ 1: If the adverse event is considered serious, complete a Serious Adverse Event Form and Log. 2: The definitions of severity are the following: Mild – Transient or mild discomfort; no limitation in activity; no medical intervention/therapy required. Moderate – Mild to moderate limitation in activity, some assistance may be needed; no or minimal medical intervention/therapy required. Severe – Marked limitation in activity, some assistance usually required; medical intervention/therapy required, hospitalizations possible. Life threatening – Extreme limitation in activity, significant assistance required; significant medical intervention/therapy required hospitalization or hospice care probable. 3. Treatment or Procedure or Study Discontinuation (Please Specify) Signature of the Principal Investigator: Date: Page 2 of 2