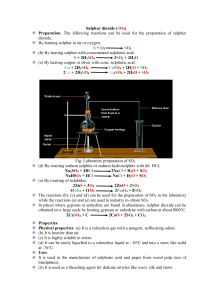
Sulphuric acid is an oily, colorless liquid with no odor. It is a key component in batteries, wastewater treatment, ore production, and fertilizer creation. Sixtyfive percent of all fertilizers available on the market today are created from a combination of this acid and other chemicals. It is created from inorganic materials through a process that utilizes a special type of chemical reaction. A variety of substances can be mixed with sulfuric acid in order to create new products. Sox refers to all sulphur oxides , the two major ones being sulphur dioxide ( 𝑆𝑂2 ) and sulphur trioxide (𝑆𝑂3 ). Soluble in water Diprotic acid Dense Nonvolatile acid Properties of Sulphuric Acid Highly corrosive Viscous colourless liquid Oily liquid *Diprotic Acid: - Acids that contain two hydrogen ions For eg, 𝐻2 𝑆𝑂4 *Viscous: - A fluid which does not flow easily Property of dilute sulphuric acid 1. Ions in the acid Sulphate 𝑆𝑂42− , and hydrogen ions, 𝐻 + (the 𝐻+ 𝑖𝑜𝑛𝑠 𝑔𝑖𝑣𝑒 𝑠𝑢l𝑝ℎ𝑢𝑟𝑖𝑐 𝑎𝑐𝑖𝑑 𝑖𝑡𝑠 𝑎𝑐𝑖𝑑𝑝𝑟𝑜𝑝𝑒𝑟𝑡𝑖𝑒𝑠) 2. Reaction with metals Products: a salt and hydrogen gas Eg: Fe + 𝐻2 𝑆𝑂4 → 𝐹𝑒𝑆𝑂4 + 𝐻2 3. Reaction with carbonates Products: a salt, water and carbon dioxide gas Eg: 𝐶𝑢𝐶𝑂3 + 𝐻2 𝑆𝑂4 → 𝐶𝑢𝑆𝑂4 + 𝐻2 𝑂 + 𝐶𝑂2 4. Reaction with metal oxides and hydroxides (bases) Products: a salt and water Eg: 𝐻2 𝑆𝑂4 + 𝐶𝑢𝑂 → 𝐶𝑢𝑆𝑂4 + 𝐻2 𝑂 5. Types of salts produced with alkalis Two salts: products of reaction with NaOH are 𝑁𝑎𝐻𝑆𝑂4 𝑎𝑛𝑑 𝑡ℎ𝑒𝑛 𝑁𝑎2 𝑆𝑂4 6. Effect of Indicators Blue litmus turns red Universal indicator turns red (shows a pH of 1 or 2) H2SO4 • commercially important raw material In the surface, molten sulphur is burnt in dry air to produce sulphur dioxide,𝑆𝑂2 . The gas produced is purified and cooled. In the converter, 𝑆𝑂2 and excess oxygen gas, 𝑂2 are passed over a few plates of Vanadium (V) oxide, 𝑉2 𝑂5 catalyst at 450°C to produce sulphur trioxide, 𝑆𝑂2 . In the absorber, the sulphur trioxide, 𝑆𝑂3 is first reacted with concentrated sulphuric acid, 𝐻2 𝑆𝑂4 to form a product called oleum, 𝐻2 𝑆2 𝑂7 . The oleum, 𝐻2 𝑆2 𝑂7 is then diluted with water to produce concentrated sulphuric acid, 𝐻2 𝑆𝑂4 in large quantities. Main source of Sulphur Dioxide : Burning of fossil fuels like petroleum and from manufacture of plastic or rayon. Other industrial processes such as manufacturing of sulphuric acid also contribute to the presence of 𝑆𝑂2 in the air.