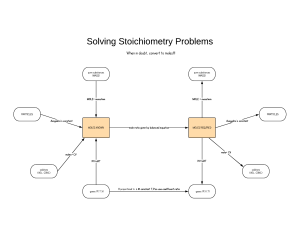
The mole concept Why did people start using kilograms, meters, liters etc? A mole is simply a unit of measurement. Units are invented when existing units are inadequate. Chemical reactions often take place at levels where using grams wouldn't make sense, yet using absolute numbers of atoms/molecules/ions would be confusing, too. So how much is a mole? A mole is the amount of a substance which contains the same number of chemical species as there are atoms in exactly 12 grams of the isotope carbon – 12. In short – 1 mole = 6.02 x 1023 particles This is called Avogadro’s constant (more on him in a second) The mole is a SI unit with the symbol mol. (The word comes from the Latin for heap or pile) Avogadro’s Constant (L) Lorenzo Romano Amedeo Carlo Avogadro di Quaregna e di Cerreto, Count of Quaregna and Cerreto (9 August 1776, Turin, Piedmont – 9 July 1856) was an Italian scientist. He is most noted for his contributions to molecular theory, including what is known as Avogadro's law. In tribute to him, the number of elementary entities (atoms, molecules, ions or other particles) in 1 mole of a substance, 6.02214179(30)×1023, is known as the Avogadro constant. Avogadro’s Constant (L) Avogadro's number is the number of particles found in one mole of a substance. – Number of atoms in 1 mole of an element – Number of molecules in 1 mole of a compound In the periodic table The relative atomic mass in the periodic table is the mass of 1 mole of that atom. So, in the case of Hydrogen, 1 mole weighs 1.01 g. When working with molecules, the composition is given by its molecular formula. - The relative molecular mass (Mr) is calculated by adding the relative atomic masses of the atoms that make up the molecule (What would the Mr of water be??) Molecular mass of Water So how do you calculate the number of moles? Example Problem • Determine the number of moles of CO2 in 454 grams. Solution First, look up the atomic masses for carbon and oxygen from the Periodic Table. The atomic mass of C is 12.01 and the atomic mass of O is 16.00. Manipulating the equation Here’s an easy way to figure out how to use the equation if mass or molar mass is asked instead of number of moles: Using Avogadro’s constant Example Calculate how many molecules are present in 3.00 moles of H2O. What is given: •n= 3.00 moles •L= 6.02 x 1023 What is asked: •N – number of particles. References • Brown, C & Ford , M (2009) Chemistry, Pearson, Malaysia. • http://chemistry.about.com