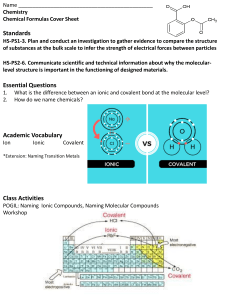
SCH4C1: Unit One Review Assignment Matter and Qualitative Analysis Know the following terms: Energy level: Atomic number: Mass number: Isotope: Emission spectrum: Ionic bond: Covalent bond: Molecular compound: Chemical reaction: Chemical equation: Synthesis reaction: Decomposition reaction: Single displacement reaction: Double displacement reaction: Precipitate: Precipitation reaction: Ionic equation: Spectator ion: Net ionic equation: Name: Assignment Value: 5% upon submission ON THE DAY OF THE TEST, PRIOR TO THE START OF THE TEST. Late assignments will NOT be accepted. 1. Identify the three subatomic particles, their charges and their location within an atom: Subatomic Particle Charge Location 2. Use your knowledge of the periodic table to complete the following charts: Element Symbol Atomic Number Mass Number No. of protons No. of electrons 34 No. of neutrons 45 137 Bi 81 83 3. What can the group number tell you about the atoms within that group? 4. What can the period number tell you about the atoms within that period? 5. What would be the ionic charge of the following atoms? a) Phosphide ion b) Calcium ion c) Bromide ion d) Aluminum ion e) Helium 6. Use your knowledge of chemical nomenclature to provide the names or formulas of the given compounds. Know the rules for ionic vs. molecular compounds. Name Formula Ca3N2 Al2(SO3)3 FeCl3 Pb3(PO4)4 P2O5 CO CCl4 Name Formula Magnesium oxide Potassium nitrate Cobalt (III) nitride Mercury (II) bromate Dinitrogen tetraiodide Iodine heptafluoride 7. Draw the Lewis diagram for the formation of the following ionic compounds. Remember, FIRST determine the formula of the ionic compound. Ba & S C&F Ca & O Mg & N 8. List three (3) things you must remember when drawing Lewis diagrams for ionic compounds. 9. Draw the Lewis diagram for the formation of the following molecular compounds. Remember, the atoms that need the most electrons are often in the centre of the molecule. HCN PO4 SO3 CH2F2 10. List three (3) things you must remember when drawing Lewis diagrams for molecular compounds. 11. Complete the following chart comparing and contrasting ionic vs. molecular compounds. Property Ionic Compounds Molecular Compounds Types of elements involved Transfer or sharing of valence electrons? Physical state at room temperature Melting Point Boiling Point Solubility Electrical Conductivity 12. Why are ionic compounds electrically neutral? 13. Write the word, skeleton and balanced chemical equation for the following reaction. A solution of hydrogen chloride reacts with sodium carbonate to produce carbon dioxide, sodium chloride, and water. Word: Skeleton: Balanced: 14. What is the Law of Conservation of Mass? 15. What are the diatomic elements? 16. Balance the following chemical equations. If the reaction is already balanced, you do not need to write any coefficients in the blanks. Also, identify the type of reaction as a Synthesis, Decomposition, Single Displacement, Double Displacement or Combustion reaction. Chemical Equation Type of Reaction a) HgO → Hg + b) NaCl + AgNO3 → c) Cl2 + d) C2H6 + O2 → CO2 + H2O e) H2O + Fe → Fe2O3 + H2 f) Al2S3 + g) S8 + Fe → FeS h) N2 + H2 → NH3 i) KClO3 → j) Al2(SO4)3 + Ca → O2 NaNO3 + AgCl CaCl2 Ca(OH)2 → KCl + Al(OH)3 + CaS O2 Ca(OH)2 → Al(OH)3 + CaSO4 17. Use the activity series to determine whether the following single displacement reactions will proceed. If it will, complete and balance the chemical equations. If will not proceed, simply write “No rxn.” K+ Ag2S → Br2 + NaCl → Pb4+ + NiCl2 → Al + SnO → F2 + KI → Au2+ + PtBr2 → Mg + Cu(NO3)2 → Cl2 + CaF2 → 18. Use your solubility guidelines to write out the products of the following double displacement reactions. In this exercise, write the symbol (s) beside any precipitates that will form and (aq) beside any soluble products. Balance ALL OF THE EQUATIONS (For practice! ). Al2(SO4)3 (aq) + Na3PO4 (aq) → BaCl2 (aq) + H3PO4 (aq) → K2SO4 (aq) + MgF2 (aq) AlCl3 (aq) + BaSO4 (aq) → → 19. Write the balanced, ionic and net ionic equations for the following double displacement reactions. Identify the spectator ions and the precipitate in each reaction. Reaction: Potassium chloride and lead (II) nitrate Balanced: Ionic: Net Ionic: Spectator Ions: Precipitate: Reaction: Ammonium phosphate and zinc nitrate Balanced: Ionic: Net Ionic: Spectator Ions: Precipitate: