Gas Laws Worksheet: Kinetic Theory, Pressure, Volume, Temperature
advertisement

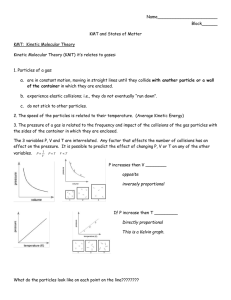
Name: Nickname: You can use the simulator found at https://ch301.cm.utexas.edu/simulations/js/idealgaslaw/ to help you answer the following questions. Chemistry Inquiry – Introduction to the Gas Laws Review of concepts from a previous class: 1. Kinetic Molecular theory According to the Kinetic Molecular Theory: particles All matter is compose of tiny ______________ These particle are in constant ______________ motion 2. This movement of particles has a great impact on the properties of matter. motion Kinetic Energy is the energy of _______________________. ___________________ is a measure of the average Kinetic Energy of the Temperature particles. An increase of ________________ will increase the Kinetic Energy. temperature 1/2mv^2 The formula to calculate Kinetic energy is Ke = _______________. If particles are moving in a confined place, then they will ultimately collide ______________ with one and other. Gas Pressure and Volume: The box below represents a canister designed to hold a gas and measure the gas pressure. Inside the box, place 10 gas particles, use small circles to represent them. Use arrows to indicate the random motion of the particles. P What causes gas Pressure? Collisions with walls If there were no particles of gas in the box, what would the gas pressure be? zero In the diagram below, the size of the box is decreased by ½. Place the 10 particles of gas into the smaller volume. Draw arrows to indicate random motion. 2atm P 0 atm 10 atm What has happened to the number of collisions? Explain. increase What has happened to the interior surface area? Explain. decrease less volume What has happened to the number of collisions against the side of the box? Explain. increase less volume Does this cause a change in pressure? If so, what change should be observed? Explain. yes, increase more collions with walls On the pressure dial above, indicate what you think the change in pressure would be. Would go to 1 atm if started 2 atm Using what you have learned, state a general rule for the relationship between pressure and the volume of a gas. PV=k P1V1=P2V2 The diagram below represents a balloon that is partially inflated at a specific temperature. Inside balloon A, place 10 gas particles, use small circles to represent them. Use arrows to indicate the random motion of the particles. A B T = 200 K T= 100K What are the gas particles doing inside the balloon to give it the shape (and volume) it has? Colliding with the sides In diagram B, the temperature is doubled. What happens to the kinetic energy of the gas particles inside the balloon? increase What happens to the velocity of the particles inside the balloon? Increase What happens to the collisions between the particles and the walls of the balloon? Increase frequence and more energetic collisions In diagram B, the original balloon volume is drawn for your reference. Estimate the new volume of the balloon, and draw it using the reference provided. 2 times volume State a general rule for the relationship between the volume of a gas and the temperature. T=kV or V1/T1=V2/T2 or directly proportional