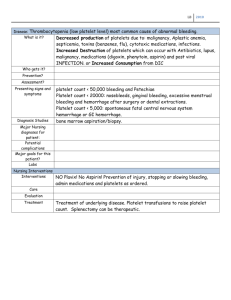
ANTICOAGULANTS and ANTIPLATELET AGENTS 1. Review the coagulation cascade, platelet aggregation and clot formation Platelet GPIb-IX-V receptor complex binds to Von Willebrand Factor on exposed collagen (tethering) This adhesion is inhibited by nitric oxide and prostaglandins on intact endothelial surfaces his binding, as well as release of thrombin from tissue factor, activates the platelets Activated platelets: Signal the activation of integrins that bind strongly to collagen and other collagen ligands (vWf & fibronectin) Integrins signal the release of agonists ADP, ATP, platelet activating factor, platelet factor 4, serotonin, vWf, and thromboxane A2 from platelet granules, which amplifies integrin signaling and thrombus growth/platelet aggregation Aggregation the released agonists and collagen bind to receptors on activated platelets to increase intracytoplasmic calcium platelets change from smooth to a spiny sphere plasma membrane arachidonic acid released and converted to prostaglandin H 2 by COX1 & thromboxane synthase (potent platelet activator) GPIIb/IIIa, resting platelet membrane integrin, undergoes an inside out signaling that initiates an affinity for fibrinogen Platelet aggregation occurs as GPIIb/IIIa bridge between fibrinogen molecules as well as vWf o Fibrinogen is synthesized by the liver and binds to sites on activated platelets o vWf is secreted from platelet granules and endothelial cells Coagulation system involves the sequential activation of inactive clotting factors/proteins circulating in the blood Intrinsic pathway = triggered by damage to endothelium contact between Factor XII (Hageman Factor) and negatively charged substances like collagen, phospholipids, and platelet plug that are located beneath and exposed by injured endothelial tissue. This leads to Factor XIIa which converts FXI to Factor XIa. Factor XIa catalyzes reaction of factor IX to Factor IXa. Factor IXa along w/ cofactor VIIIa form tenase complex, which activates Factor X to Factor Xa. Extrinsic pathway = triggered only if cells surrounding blood vessel are damaged, leading to the leakage of TF. The exposure of factor VII to tissue factor (factor III or thromboplastin) forms an activated TF-FVIIa complex that catalyzes activation of factor IXa & Xa Common pathway: meet at factor Xa …. o Fibrin clot is produced during the final common coagulation pathway during which the tenase complex/plasminogen activator initiates the conversion of prothrombin to thrombin and finally fibrinogen to fibrin Important to know the pathway because important for lab monitoring o PTT (Intrinsic pathway): Heparin, Angiomax (bivalirudin), Argatroban (measure PTT) o Xa (Common pathway): LMWH, fondaparinux (measure anti-Xa) o PT/INR (Extrinsic pathway): Coumadin (warfarin) (measure INR) INR is correction factor for variation between labs 2. Describe medications which affect hemostasis including mechanism of action and adverse effects (♦ = safe in pregnant pts) ANTI-PLATELET MEDICATIONS Aspirin Mechanism of Action: irreversibly inhibits COX inhibits prostaglandin synthesis, including thromboxane A2, which is one agent responsible for irreversible plt activation & aggregation o Hold aspirin 2-3 days before surgery (although some effects of aspirin likely to persist 1-2 weeks) AEs: dyspepsia, bleeding, asthma exacerbation, rare rash, hepatotoxicity, thrombocytopenia Monitor: CBC, platelets, serum Cr Avoid in children < 16 yo with recent/current viral illness or recent live vaccine d/t risk of Reye’s syndrome o Classic features of Reye’s = rash, vomiting, liver damage (fatty liver, hepatomegaly), & life-threatening encephalopathy Drug (Brand) Aspirin Dose 81 – 325 mg QD Comments - recommended in pts w/o contras with unprovoked proximal DVT or PE who are stopping anticoagulation - AHA/ASA: recommend aspirin 81 mg QD ir 100 mg Q48 hrs for pts at high risk for stroke, including in CKD * aspirin should NOT be used to prevent stroke among pts whose sole risk factor is DM or asymptomatic PAD * - ADA: consider aspirin for women ≥ 50 w/at least one additional major risk factor (family hx of premature ASCVD, HTN, smoking, HLD, or albuminemia) & not at increased bleed risk Platelet P2Y12 Inhibitors Mechanism of Action: inhibits ADP-induced platelet aggregation thru interaction w/ P2Y12 receptor AEs: bleeding, dyspepsia, rare rash, headache Monitor: CBC, platelets All are irreversible, except ticagrelor ALL REQUIRE A LOADING DOSE & lots of drug interactions! Effects seen in 3-7 days Drug (Brand) Clopidogrel (Plavix) Prasugrel (Effient) Ticagrelor (Bilinta) Dose 75 mg Q24hr ± 300600mg loading dose 60mg loading dose then 10 mg Q24hr 180 mg loading dose, then 90 mg Q12hr PK - requires activation by liver - irreversible - requires activation by liver - irreversible - does NOT require liver activation - reversible Cagrelor (Kangreal) 30 mcg/kg bolus, then 4 mcg/kg/min infusion - requires activation by liver - irreversible Comments 2C19 & 3A4 substrate Hold for 5 days before surgery 3A4 substrate Prasugrel = CONTRA after TIA or Stroke AE of dyspnea 3A4 substrate & 2C9 inhibitor Ticagrelor = USE IN CIRRHOSIS PTS! PAR-1 Antagonist Mechanism of Action: reversible antagonist of protease-activated receptor-1 (PAR-1) expressed on platelets, inhibits thrombin-induced and thrombin receptor agonist peptide (TRAP)-induced platelet aggregation o Note: due to it’s very long half-life, vorapaxar is effectively irreversible AEs: bleeding, anemia, rash, depression Monitor: CBC AVOID in patients w/hx of stroke, TIA, intracranial hemorrhage or severe hepatic impairment Vorapaxar = for pts w/ prior MI or PAD Drug (Brand) Vorapaxar (Zontivity) Dose 2.08 mg QD in combo w/aspirin or clopidogrel Comments - Vorapaxar = for pts w/ prior MI or PAD Dipyridamole Mechanism of Action: vasodilator that inhibits plt aggregation by inhibiting adenosine uptake & cGMP activity AEs: bleeding, HA, abdominal pain, diarrhea, nausea, dyspepsia Monitor: CBC, platelets Dipyridamole = for stroke prevention after TIA Drug Aggrenox (dipyridamole ER 200 mg / aspirin 25 mg) Dose 1 capsule PO Q12 hr ~ generally taper, often starting w/Qhs dosing for 1st week to prevent HA Comments - Dipyridamole = for stroke prevention after TIA FIBRINOLYSIS IMPACTING MEDICATIONS (fibrinolysis = endothelial cells secrete tissue plasminogen activator or t-PA at sites of injury which binds to fibrin and converts plasminogen to plasmin, which digests fibrin; PAI-1 & PAI-2 inactive t-PA, while α2-antiplasmin inactivates plasmin) Fibrinolytics Mechanism of Action: convert plasminogen to plasmin, which in turn breaks down fibrinogen, destabilizing the clot AEs: bleeding, rare angioedema Alteplase (t-PA, Activase, Cathflo) = most commonly used o dose depends on indication, but NEVER EXCEED 100 mg for any indication Alteplase (t-PA, Activase, Cathflo) = for (1) acute ischemic stroke, (2) severe, total occlusion PE, (3) acute MI (at centeres where PCI is not readily available), (4) catheter clearance, & more Fibrinolysis Inhibitors Mechanism of Action: aminocaproic acid inhibits the activation of plasminogen to plasmin (useful in some bleeding disorders) AEs: thrombosis, myopathy, hypotension, abdominal discomfort, diarrhea, blood dyscrasias Monitor: CBC, fibrinogen, BUN, Cr Aminocaproic acid & tranexamic acid = used to block fibrinolysis in pts w/severe bleeding disorders or in surgery w/severe bleeding HEPARINS (safe in pregos/lactation b/c large molecules unlikely to cross) Unfractionated Heparin (UFH) ♦ Mechanism of Action: directly activates anticlotting factor antithrombin III (ATIII), allowing ATIII to more quickly inactivate factors 9, 10, & 2 (thrombin) AEs: bleeding, rare thrombocytopenia, alopecia, hepatotoxicity, hypersensitivity reactions Monitor: PTT or anti-Xa levels, CBC, platelets, K+ Antidote = protamine 1 mg per 100 units of heparin, max 50 mg Drug UFH ♦ Dose - prophylaxis: 5000 units Q12h or Q8h - treatment: 10-20 units/kg/r on avg, titrated to PTT or anti-Xa Antidote - protamine sulfate 1 mg per 100 units of heparin, max 50 mg Comments - mixture of molecules of diff wt & activity Low Molecular Weight Heparin (LMWH) ♦ Mechanism of Action: primarily binds antithrombin to Xa, inactivating it to prevent clotting More predictable PK, less inter-pt variability than UFH Dosed based on weight ± renal function No routine monitoring required AEs: bleeding, rare thrombocytopenia, skin necrosis, skin hypersensitivity reactions Monitor: K+, CBC, platelets, serum Cr, anti-Xa levels for select pts BLACK BOX WARNING: spinal or epidural hematomas, including subsequent paralysis, may occur w/recent or anticipated epidural or spinal anesthesia or LP in patients coagulated w/LMWH or heparinoide o Hold LMWH before & after spinal procedure o Observe pts closely for s/s of neural impairment Drug Enoxaparin (Lovenox) ♦ Dalteparin (Fragmin) ♦ Prophylaxis 40 mg Q24h or 30 mg Q12h 5000 units Q24h Treatment 1.5 mg/kg Q24h or 1 mg/kg Q12h - 200 units/kg Q24 VTE - 120 units/kg Q12 ACS Antidote - protamine sulfate 1 mg per 1 mg enoxaparin Comments - reduce dose in renal impairment - may be better to do BID option in obese & old - increase dose for overweight pts Heparin-Induced Thrombocytopenia (HIT) Heparin & LMWH can bind to platelet factor 4 and cause immune-mediated HIT (risk lower w/LMWH) 1-4% of all pts exposed to heparin for a week or more Rapid decline in platelets Thrombosis occurs in up to 50% of pts HIT Score: o High (6-8): HIT is likely o Intermediate (4-5): HIT is possible o Low (0-3): HIT is unlikely Management of HIT: o Send HIT antibody, serotonin release assay performed as confirmatory test if antibody is positive o d/c all heparin products o initiate tx w/ direct thrombin inhibitor (argatroban, bivalirudin) at therapeutic dose fondaparinux (Arixtra) can be used in select cases o Once platelet count has recovered, initiate low-dose warfarin o DOCUMENT HEPARIN ALLERGY WARFARIN Warfarin (Coumadin) Mechanism of Action: inhibits the synthesis of Vit K-dependent clotting factors (7, 9, 10, and 2) Warfarin does NOT inhibit activity of existing clotting factors so have to wait for depletion of factors to see effects of warfarin o NOT therapeutic until at least 3-4 days and a couple days of therapeutic INR – should overlap warfarin w/heparin for the first two “therapeutic INRs” b/c can be falsely therapeutic, reflecting depletion of factor 7 rather than body’s ability to halt clot expansion or form new thromboses AEs: bleeding, TERATOGENIC, rare hypersensitivity reaction Monitor: INR (goal 2-3), CBC LOTS of DRUG INTERACTIONS (2C19, 1A2, 2C9, 3A4 substrate) & food interactions (vit K) o High: kale, spinach, turnip greens, collar greens, swiss chard, parsley, mustard greens o Moderate: Brussel sprouts, green leaf lettuce, broccoli, endive lettuce, romaine lettuce Guidelines against vit K for INR < 10 without bleeding (just hold dose with OUT giving vit K if not bleeding & INR < 10) BRIDGE THERAPY: o MUST overlap w/warfarin therapy until INR is within therapeutic range for two consecutive days o Minimum 5 days overlap Drug Usual Initial Dose 5 mg PO daily, titrate w/caution, effect seen in ~72 h, may take 5 days for full anticoag effects Other Doses - Old, small, poor nutrition, hepatic impairment, CHF, coadministration w/drugs that decrease warfarin clearance = initial dose of 2.5 - young, fat, coadministration w/drugs that increase warfarin clearance = initial dose of 7.5 Antidote - vit K (phytonadione) 110 mg PO or IV; large doses can result in warfarin-resistance for up to a week Comments Adjust dose by 10-15% based on weekly total warfarin dose DIRECT ORAL ANTICOAGULANTS - No need for routine lab monitoring Less drug interactions that warfarin Onset w/in hours (no need for IV anticoagulant bridge at initiation) Xa inhibitors & DTIs are recommended over warfarin in patients withOUT cancer who have a lower extremity DVT or PE Compared to warfarin, they reduce risk of stroke and systemic embolism, reduce mortality, reduce risk of hemorrhagic stroke, but increase rate of GI bleeding - Expensive - MOST should be AVOIDED in RENAL impairment - Antidote only available for dabigatran - Offset within hours: o Rapid loss of efficacy w/missed doses, use caution in non-adherent pts o Consider anticoagulant bridge if temporarily held for any reason other than signif bleeding Xa Inhibitors Mechanism of Action: inhibit factor Xa 3A4 & p-glycoprotein substrate AEs: bleeding No need for routine lab monitoring!!! YAY, but may monitor CBC CONTRA: pregos & lactating Drug (Brand) Rivaroxaban (Xarelto) Prophylaxis 10mg q24h Treatment - AF: 20mg q24h - VTE: 15mg q12hr x 2 d then 20mg q24h Comments - reduce dose/avoid in renal or hepatic impairment - 3A4 & P-gp substrate Apixaban (Eliquis) 2.5mg q12h Edoxaban (Savaysa) N/A Fondaparinux (Arixtra) 2.5mg SQ q24h 5 mg q12h - ONLY ONE W/DATA in RENAL IMPAIRMENT REDUCE DOSE of APIXABAN & USE IN RENAL IMPAIRMENT BC BEST ONE!!!! - 3A4 & P-gp substrate - 60 mg daily - 30 mg daily if CrCl 15-30, wt ≤ 69kg or on certain P-gp inhibitors 5-10 mg q24h depending on weight - avoid in CrCl > 95 or < 15 - P-gp substrate - reduce dose/avoid in renal impairment & weight < 50 kg - only available as injection Direct Thrombin Inhibitors (DTIs) Mechanism of Action: inhibit thrombin which prevents thrombin-induced conversion of fibrinogen to fibrin AEs: bleeding, hypotension, gastritis-like sx (dabigatran) Monitor: PTT, INR (Argatroban), CBC CONTRA: pregos & lactating Dabigatran (Pradaxa) preferred over coumadin for Afib, including paroxysmal Afib (but NOT valvular Afib) Drug (Brand) Dabigatran (Pradaxa) Dose 150mg q12h Antidote - Idarucizumab (Praxbind) for dabigatran Comments - Dabigatran = for non-valvular Afib - BB Warning: against use w/mechanical valve - reduce dose/avoid in renal impairment - co-formulated w/acid to enhance absorption dyspepsia - do not crush/open capsule! Bivalirudin (Angiomax) 0.15 to 1.75 mg/kg/h - Angiomax = for procedural anticoagulation OR anticoagulation in setting of HIT - reduce dose in renal impairment Argatroban 2-10 mcg/kg/min, titrate to PTT - Argatroban = for procedural anticoagulation OR anticoagulation in setting of HIT - avoid in hepatic insufficiency