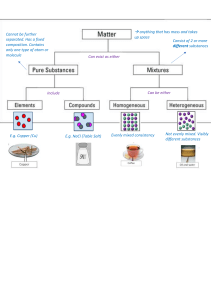
1 2 3 4 5 6 7 8 9 10 11 12 13 Questions 1. A substance has a pH of 5 – How would you describe it? 2. What pH would you expect a strong alkali to have? 3. Water would have a pH of… 4. A substance has a pH of 9.5 – How would you describe it? 5. True or False - Hydrochloric acid is a very strong acid so would have a pH of 3? Score /5 14 Which is which?? You have been given samples of 6 substances A-F. They have been mixed up by accident and you task is to identify which one is which using their pH’s. Read the information given and start your testing! You will be given 10 minutes to identify as many as possible. pH level Type and strength Eg. Strong acid A B C D E F I managed to identify Next time I would /6 of the substances Name of substance