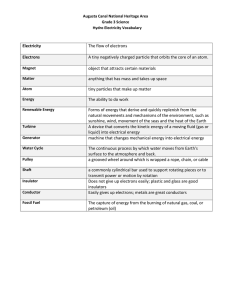
Basic Electrical Engineering Module 1 – Introduction to Basic Electricity What is electricity? The invisible energy that constitutes the flow of electrons in a closed circuit to do work is called ‘electricity’. History of Electricity Long before any knowledge of electricity existed, people were aware of shocks from electric fish. Ancient Egyptian texts dating from 2750 BCE referred to these fish as the "Thunderer of the Nile", and described them as the "protectors" of all other fish. Electric fish were again reported millennia later by ancient Greek, Roman and Arabic naturalists and physicians.[2] Several ancient writers, such as Pliny the Elder and Scribonius Largus, attested to the numbing effect of electric shocks delivered by electric catfish and electric rays, and knew that such shocks could travel along conducting objects. Ancient cultures around the Mediterranean knew that certain objects, such as rods of amber, could be rubbed with cat's fur to attract light objects like feathers. Thales of Miletus made a series of observations on static electricity around 600 BCE Electricity would remain little more than an intellectual curiosity for millennia until 1600, when the English scientist William Gilbert wrote De Magnete, in which he made a careful study of electricity and magnetism, distinguishing the lodestone effect from static electricity produced by rubbing amber.[6] He coined the New Latin word electricus ("of amber" or "like amber", from ἤλεκτρον, elektron, the Greek word for "amber") to refer to the property of attracting small objects after being rubbed.[11] This association gave rise to the English words "electric" and "electricity", which made their first appearance in print in Thomas Browne's Pseudodoxia Epidemica of 1646. Later in the 18th century, Benjamin Franklin conducted extensive research in electricity, selling his possessions to fund his work. In June 1752 he is reputed to have attached a metal key to the bottom of a dampened kite string and flown the kite in a storm-threatened sky.[14] A succession of sparks jumping from the key to the back of his hand showed that lightning was indeed electrical in nature. He also explained the apparently paradoxical behavior of the Leyden jar as a device for storing large amounts of electrical charge in terms of electricity consisting of both positive and negative charges.[13] While the early 19th century had seen rapid progress in electrical science, the late 19th century would see the greatest progress in electrical engineering. Through such people as Alexander Graham Bell, Ottó Bláthy, Thomas Edison, Galileo Ferraris, Oliver Heaviside, Ányos Jedlik, William Thomson, 1st Baron Kelvin, Charles Algernon Parsons, Werner von Siemens, Joseph Swan, Reginald Fessenden, Nikola Tesla and George Westinghouse, electricity turned from a scientific curiosity into an essential tool for modern life. Modern Electron Theory According to modern electron theory, every matter (whether solid, liquid, or gas) consists of tiny divisible particles called ‘molecules’. A molecule is further made up of very minute particles called ‘atoms’; and all atoms consist of electrons, protons, and neutrons. Prepared by: Engr. Bedia Atomic Number All elements are arranged in the periodic table of the elements in order according to their atomic number. The atomic number equals the number of protons in the nucleus. For example, hydrogen has an atomic number of 1 and helium has an atomic number of 2. In their normal (or neutral) state, all atoms of a given element have the same number of electrons as protons; the positive charges cancel the negative charges, and the atom has a net charge of zero, making it electrically balanced. Shells, Orbits, and Energy Levels According to the classic Bohr model, an atom is visualized as having a planetary type of structure that consists of a central nucleus surrounded by orbiting electrons, as illustrated in Figure 1. The nucleus consists of positively charged particles called protons and uncharged particles called neutrons. The basic particles of negative charge are called electrons, which orbit the nucleus. Each type of atom has a certain number of protons that distinguishes it from the atoms of all other elements. As you have seen in the Bohr model, electrons orbit the nucleus of an atom at certain distances from the nucleus and are restricted to these specific orbits. Each orbit corresponds to a different energy level within the atom known as a shell. The shells are designated 1, 2, 3, and so on, with 1 being closest to the nucleus. Electrons further from the nucleus are at higher energy levels. Valence Electrons Electrons that are in orbits farther from the nucleus have higher energy and are less tightly bound to the atom than those closer to the nucleus. This is because the force of attraction between the positively charged nucleus and the negatively charged electron decreases with increasing distance from the nucleus. Electrons with the highest energy levels exist in the outermost shell of an atom and are relatively loosely bound to the atom. This outermost shell is known as the valence shell, and electrons in this shell are called valence electrons. Prepared by: Engr. Bedia Energy Levels and Ionization Energy Categories of Materials If an electron absorbs a photon with sufficient energy, it escapes from the atom and becomes a free electron. This is indicated by the ionization energy level in Figure 3. Any time an atom or group of atoms is left with a net charge, it is called an ion. When an electron escapes from the neutral hydrogen atom (designated H), the atom is left with a net positive charge and becomes a positive ion (designated H+). In other cases, an atom or group of atoms can acquire an electron, in which case it is called a negative ion. Conductors are materials that readily allow current. They have a large number of free electrons and are characterized by one to three valence electrons in their structure. Most metals are good conductors. The Copper Atom The copper atom has 29 electrons that orbit the nucleus in four shells. The number of electrons in each shell follows a predictable pattern according to the formula, 2N^2 where N is the number of the shell. The first shell of any atom can have up to two electrons, the second shell up to eight electrons, the third shell up to 18 electrons, and the fourth shell up to 32 electrons. Semiconductors are classed below the conductors in their ability to carry current because they have fewer free electrons than do conductors. Semiconductors have four valence electrons in their atomic structures. Insulators are nonmetallic materials that are poor conductors of electric current; they are used to prevent current where it is not wanted. Insulators have no free electrons in their structure. SECTION 1 CHECKUP 1. What is the basic particle of negative charge? 2. Define atom. 3. What does an atom consist of? 4. Define atomic number. 5. Do all elements have the same types of atoms? 6. What is a free electron? 7. What is a shell in the atomic structure? 8. Name two conductive materials. Prepared by: Engr. Bedia