Honors Chemistry Questions: Acids, Bases, and pH
advertisement

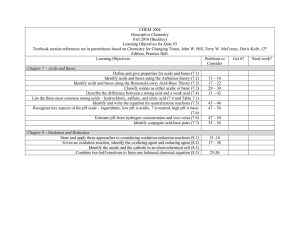
Honors Chemistry: 1. How are pH, H+ concentration and OH- concentration conceptually related? 2. How are indicators used in acid and base chemistry? 3. What mathematical information about the reactants and products can be obtained by using a balanced neutralization reaction through a titration process? 4. What is the net ionic equation for an acid-base reaction? 5. How does the concentration and the pH change when an acid or base is diluted? 6. How is the behavior of strong acids different than that of weak acids in aqueous solutions? 7. How does the Ka value relate to the strength of a weak acid? 8. What are the significant components of the Bronsted-Lowry acid base theory? Honors Chemistry: 1. You will conceptually relate pH, [H+] and [OH-] 2. You will determine how indicators are used in acid base chemistry. 3. You will use the titration process to obtain mathematical information about reactants and products using a balanced neutralization reaction. 4. You will determine the net ionic equation for an acid-base reaction. 5. You will determine the concentration and pH change when an acid or base is diluted. 6. You will determine how strong acids differ from weak acids in aqueous solutions. 7. You will relate the strength of a weak acid to its Ka value. 8. You will understand the Bronsted-Lowry acid-base theory.