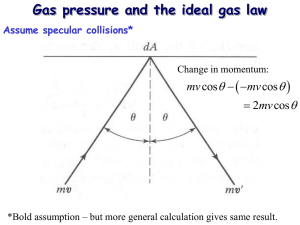
1. 2. Analysis of matter in terms of atoms in continuous motion is called Kinetic theory The theory is based on ideal gas but real gases follow the theory at low pressures & at temperature far away from liquefaction point The kinetic theory has postulates: There are a large number of molecules (N), each of mass m, moving in random directions with varying speeds Molecules are far apart from each other i.e. average separation is much greater than diameter of each molecule 3. 4. Molecules are assumed to observe classical laws of mechanics & only interact with each other when they collide. Although molecules exert weak attractive forces on each other during collisions, PE associated with these forces are small as compared to KE of movement i.e. it is negligible Collisions with another molecule or the wall of the vessel are assumed perfectly elastic. The collisions are of very short duration compared to time between collisions. This allows us to neglect PE during collisions Kinetic theory can be used to explain the gas laws. For Boyle’s law: pressure exerted on a wall of a container of a gas is due to constant bombardment of molecules. If V is halved, molecules are closer together & twice as many will be striking a given area of the wall per second i.e. pressure doubles Kinetic theory can also be used to calculate pressure exerted by gas: 𝑃= 1 𝑁𝑚𝑣ത 2 3 𝑉 (see Giancoli for derivation) where 𝑣ҧ is average speed of molecules After further manipulation: 1 𝑚𝑣ҧ 2 2 (see Giancoli for derivation) 𝐾𝐸 = Making speed subject of formula: 2 𝑣ҧ = 3𝑘𝑇 𝑚 = = 3 𝑘𝑇 2 3𝑅𝑇 𝑀 (after mathematical manipuilation) Average translational KE of molecules in random motion in an ideal gas is directly proportional to absolute T of the gas The higher the T, the faster the molecules (next slide) Speed distribution Maxwell distribution of speeds • Temperature is a measure of the average kinetic energy of gas molecules 1. A beam of electrons moving in the +ve x-direction impacts a target in a vacuum chamber. a) If 1.25x1014 electrons travelling at a speed of 3x107m/s strike the target surface during each brief pulse lasting 5x10-8s, what average force is exerted on the target during each pulse? b) What average pressure is exerted on the beam spot, which has a radius of 4mm? Mass of the electron = 9.11x10-31kg NOTES: Assume that all the electrons impacting on the surface are absorbed by the surface The bean spot is the target struck by the beam. By Newton’s 3rd Law: Average force exerted is the reaction force (its negative) a) F1e = -change in momentum /time = -∆p/∆t = -mvf – mvi/t Fav = -N ∆p/∆t = -N m(vf-vi)/∆t = -Nm (0-vi)/∆t Fav = - (1.25x1014)x(9.11x10-31)x (-3x107)/(5x10-8) N Therefore Fav = 0.0683N b) P = F/A = 0.0683N/πr2 = 0.0683/ π x (4x10-3)2 P = 1.36x103Pa What is the average translational KE of molecules in an ideal gas at 37 oC? Solution 𝐾𝐸 = 1 𝑚𝑣ҧ 2 2 3 = 𝑘𝑇 2 −21 = 3 2 1.38𝑥10−23 37 + 273 𝐾𝐸 = 6.42𝑥10 J NB: A mole of molecules would have a total KE of: 𝐾𝐸 = 6.42𝑥10−21 6.02𝑥1023 = 3900 J Root-mean-square velocity is the measure of the velocity of particles in a gas which is most convenient for problem solving within the kinetic theory of gases. It is defined as the square root of the average velocity-squared of the molecules in a gas. It is given by the formula. Remember from vector notation, velocity becomes speed once you average it out, because averaging takes care of both magnitude and direction So vrms is also written as urms where u is the average speed of the molecules Velocity Distributions - Distribution of Molecular Speeds u rms 3RT = u = M ump = 2 2RT M ump : uavg : urms = 1.000 : 1.128: 1.225 Thermodynamic temperature is the variable needed for subjects like heat transfer, because it is the translational kinetic energy which leads to energy transfer from a hot area (larger kinetic Energy, Higher temperature, higher molecular speeds) to a cold area (lower molecular speeds) in direct collisional transfer. At a certain speed, the root-mean-square-speed of the molecules of H2 gas in a sample of gas is 1055 m/s. Compute the root-mean square speed of molecules of O2 gas at the same temperature. Solution 1. Find T for the H2 gas with a vrms = 1055 m/s 2. 𝑇= 𝐻2 𝑣𝑟𝑚𝑠 2 𝑀𝐻2 3𝑅 = 1055 2 2 3 8.314 = 89249.1 K Find vrms of O2 gas at the same temperature 𝑂2 𝑣𝑟𝑚𝑠 = 3𝑅𝑇 𝑀𝑂2 = 3 8.314 89249.1 32 = 263.75 m/s Kinetic theory can also be used to explain the 4 states of matter & accompanying phase changes Homework: Explain states of matter using Kinetic theory 1. 2. 3. Melting – commonly used to indicate changing from solid to liquid Normal melting point – temperature at which vapour pressure of solid & vapour pressure of the liquid are equal Freezing – changing from a liquid to a solid. Melting and freezing occur at the same temperature Boiling/evaporation – used to indicate changing from a liquid to a gas/vapour Normal boiling point – temperature at which vapour pressure of liquid is equal to standard atmospheric pressure (101.325 kPa) Boiling point is a function of pressure, at lower pressures, boiling point is lower ✓ ✓ 4. 5. 6. 7. Evaporation takes place only at surface of a liquid or solid while boiling takes place throughout the body of a liquid What is the difference between a Gas & a Vapour? Gas – state of matter that exists at normal room temperature Vapour – produced by particles escaping from a state of matter that is normally liquid or solid at room temperature Condensation – used to indicate changing from a vapour to a liquid Liquefaction – turning a gas to a liquid Only happens at low temperature & high pressure situations Sublimation – used to indicate when a substance changes directly from a solid to a vapour e.g. "dry ice", solid CO2 Deposition – when a substance changes directly from a vapour to a solid (opposite of sublimation) e.g. formation of frost A cooling curve is a line graph that represents change of phase of matter, typically from a gas to a solid or a liquid to a solid The independent variable (X-axis) is time & the dependent variable (Y-axis) is temperature See an example of a cooling curve of castings The initial point of the graph is the starting temperature of the matter noted as pouring temperature When the phase change occurs there is a "thermal arrest” i.e. temperature stays constant Amount of energy required for a phase change is known as latent heat “Cooling rate" is the slope of the cooling curve at any point Next slide give a practical cooling curve for a well known substance Cooling curve of Naphthalene Watch Video: - Water the great mystery on Sakai. Answer Questions provided on the handout Explain the change of phase using the Kinetic Theory of matter. Indicate two areas where water does not seem to obey the tenets of the kinetic theory. Explain these exceptions Does the fact that water does not obey all the tenets of the kinetic theory invalidate the theory? Discuss