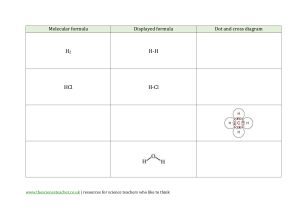
Acid Base Stations
advertisement

4 · Acids – Bases Solutions______________________________ STATION 6 – ACID STRENGTH, Ka H2SO4, Ka1 = very large; Ka2 = 1.2 x 10-2 HF, Ka = 7.2 x 10-4 NH3, Kb = 1.9 x 10-5 HCN, Ka = 6.2 x 10-10 Use this information to rank the following acids from weakest to strongest. Justify your choices using Ka’s. HCN, NH4Cl, HF, HSO4weakest Ka values pKa values strongest