Organic Chemistry Chapter 5 Stereoisomers H. D. Roth
advertisement

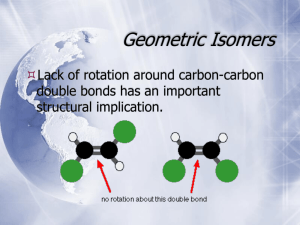
Organic Chemistry Chapter 5 Stereoisomers H. D. Roth LECTURE POSTING V Stereoisomerism A type of isomerism; two compounds are stereoisomers when they differ only in the spatial relationship of their components. In order to discuss isomerism we use the following terms (with which you are familiar): Composition: the type and number of atoms in a molecule; Constitution: the way in which these atoms are connected (we also call this connectivity); Configuration: the arrangement of the atoms in three-dimensional space; The term Conformation: also describes the arrangement of atoms in 3D space, when several arrangementsare possible; A. Conformational isomerism The conformation can be changed by free rotation about one or two single bonds; Br Br B. axial Br equatorial Br Configurational isomerism I: cis-trans or geometric isomerism The configuration cannot be changed; it is “fixed”, by restricting free rotation. 1) Rotation can be restricted if two carbons of an alkane chain are tied up forming a ring. You have seen examples in the cycloalkanes: substituents can be on the same side (cis) or on opposite sides (trans) of the ring plane; two examples are shown below. CH3 H H H CH3 H CH3 CH3 1 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth 2) Rotation can be restricted also when two adjacent carbon atoms are connected by a π bond in addition a σ bond (see chapter 1); the π bond holds the substituents in one plane; they can be on the same side (cis) or on opposite sides (trans) of the double bond (more in chapter 11). CH3 H3C H CH3 H H3C H cis-2-butene H trans-2-butene Geometric isomers could be interconverted by breaking and reforming a bond, either a σ bond of a cycloalkane or π bond of an alkene. These processes do occur, but they require high energies – therefore the geometric isomers of cycloalkanes and alkenes are stable at room temperature (unlike conformers, which are readily interconverted). When we compare structural and geometric isomers we note: Structural Isomers C. Geometric Isomers identical composition identical different connectivity identical different arrangement in 3D space different different heat of combustion different Configurational isomers, II: compounds with carbon stereocenters There is another way to arrange atoms in 3D space generating isomers. You have long been familiar with this type of relationship: look at your hands. Your two hands are identical in many ways, but they differ in their 3D arrangement: they are related as mirror images, they are different because they are not superimposable. 2 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth Objects that are different in their 3D arrangement, but related as mirror images, are called chiral (from Greek χειρ, hand). The difference lies in their “handedness” (analogy to your left and right hands). 1) We recognize chirality in a molecule by an absence of symmetry. We examine a molecule for elements of symmetry; objects that have a plane of symmetry are not chiral (achiral); most compounds that lack a plane of symmetry are chiral. One exception: compounds with a center of symmetry are achiral (example below). H 3C H 3C Br Br CH3 • Br Cl Achiral due to plane of symmetry CH3 Achiral due to center of symmetry CAUTION: It is not always easy to recognize symmetry, because molecules are three-dimensional and the representations we use are two-dimensional. 2) Do we have a positive way to show that a molecule is chiral? An unmistakable, structural feature identifying chirality: an asymmetrical carbon, or stereocenter. This is an sp3 hybridized tetrahedral carbon (angle ~ 109.5°), with four different substituents. Compounds containing an asymmetrical carbon exist in two isomeric forms that cannot be superimposed; they are "mirror images" of each other. We call such molecules enantiomers (the left molecule is the enantiomer of the right molecule and vice versa). 3 Organic Chemistry R1 Chapter 5 Stereoisomers R4 R4 C C R1 R3 R3 H. D. Roth R2 R2 (where R1, R2, R3, R4 are all different) Enantiomers are identical in just about all properties, except for their “handedness” (their asymmetry or chirality). If we compare some properties of geometric isomers with stereo- isomers, we note Geometric Isomers Stereo Isomers identical composition identical identical connectivity identical different arrangement in 3D space different different heat of combustion identical (enantiomers) If that is the case, why do we have to bother with it? As it turns out, the human body has many “handed” receptors. For example, the two stereoisomers of carvone taste and smell different, one like spearmint, the other like caraway seed. 4 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth More more important is the difference between the effect of the two stereoisomers of thalidomide: the (+)-isomer is a sedative without side effects whereas the (–)-isomer is a teratogen, causing serious birth defects in babies. O O O H N N O HN O H O N H O O (+)-R a sedative (–)-S a teratogen CAUTION: if a compound has two stereocenters that are mirror images of each other, they are symmetrical and, therefore, achiral. H C* C* CH3 H CH3 The starred carbons of cis-1,2-dimethylcyclopentane have four different substituents: these carbons are stereocenters. However, because the two carbons are related as mirror images, the molecule is symmetrical, that is, achiral. 3) Drawing chiral molecules requires that you represent this 3-D feature clearly and unmistakably in two dimensions. You are familiar with the wedge and dash (dottedline) method, which I have used above; this method is usually best for showing the 3-D relationships in chiral compounds. For drawing 3-D projections of substituted alkanes, it is convenient to place the main carbon chain as a horizontal zig zag in the plane of the paper (the book does NOT always do that). We will use this convention whenever possible. Also, if possible we 5 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth place a H on a stereocenter on a dotted line and the other substituent on the wedge. The projection of the two enantiomers (mirror images) of 2-bromopentane are shown below. H Br Br H Please, note that the dash and the wedge should always point away from the chain (this is a consequence of the carbon being tetrahedral). 4) Naming enantiomers - absolute configuration In order to properly name a stereocenter we have to take two steps: a) “rank” the four substituents; a (high) – b – c – d (low) b) determine their arrangement in 3-D space as either R (rectus) or S (sinister) 4a) We rank substituents according to a convention by Cahn, Ingold, Prelog, following these rules: Rule 1a atomic number of substituent High atomic number has preference over low F > O > N > C >>>> H I > Br > Cl > S > O Rule 1b isotopes (they really thought of everything ) heavier isotope has priority D 13 Rule 2 C > > H 12 C (not often encountered) For groups with the same atomic number (if there is a tie in the atomic numbers) break the tie by ranking its substituents to the first point of difference: the substituent with the first point of difference in priority wins. Since we are dealing with 6 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth compounds containing many carbon atoms, there is almost always a tie. For example, compare methyl (C with 3 H’s) to ethyl (the attached C has 2 H’s and one C), propyl (the attached C has 2 H’s and one C) and isopropyl (the attached C has one H and two C’s; the 2 C’s are the point of difference). CH2–CH3 > CH2–H CH(CH3)2 > CH2– CH3 C(CH3)2 > CH–CH2–CH3 Note: we don’t “weigh” the entire group, we look for the first difference; a higher ranking substituent further away does not matter. Rule 3 CH2-CH(CH3)2 > CH2-CH2-CH2-CH3 O–CH3 > O-H 2-methylpropyl > butyl CH2-Cl > CH2-CH2-Br CH2NH2 > C(t-Bu)3 multiple (double or triple) bonds H(C = C) treated as H(CC2) C≡C treated as CC3 HC=O treated as HCO2 HC=S treated as HCS2 4b) Once you have ranked the substituents by priority, determine the arrangement of the four substituents in 3-D space (R or S) as follows: i) Direct (point) the lowest priority group, R4 away from you: 7 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth ii) Draw an arrow connecting the substituents in order of decreasing priority: R2 R2 C R3 R3 C R1 R1 R Rectus S Sinister The arrow in the example on the left is clockwise – we call this 3D arrangement R (for Latin rectus); the mirror image (shown on the right) requires a counterclockwise arrow – we call this S (for Latin sinister). Let’s look at 2-bromopentane. Br highest: a a H lowest: d H Br H Br C3H7 second ranking: b c CH3 second lowest: c b clockwise – R The bromopentane shown (with a clockwise arrow) is the R-enantiomer, or R-2bromopentane. Its enantiomer, which requires a counterclockwise arrow to connect the substituents in order of decreasing priority, is the S-enantiomer, S-bromopentane. 8 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth a Br H counterclockwise – S c b 5. Detecting stereoisomers Is there a property, that helps us to distinguish between enantiomers? Enantiomers are identical in every respect except for their “handedness” (their asymmetry or chirality). If we compare some properties of geometric isomers with stereo isomers, we note Geometric Isomers Stereo Isomers identical composition identical identical connectivity identical different arrangement in 3D space different different heat of combustion identical (enantiomers) Differences between enantiomers are revealed only by asymmetric probes. The best-known probe is plane polarized light – therefore stereo isomerism also is called optical isomerism. How does this work? Glad you asked. 6. Optical Rotation Light is electromagnetic radiation with an electric and a magnetic component perpendicular to one another. Light has a dual nature, two different ways how light manifests itself: a) a quantized nature (light = photons, hν); you learned about this nature of light in the initiation step of free radical halogenation); b) light has wave character (we use this concept here). i. Ordinary light vectors oscillate randomly (in all directions); it is symmetrical. When ordinary light is passed through a Nicol prism (polarizer), only light whose 9 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth electric and magnetic vectors oscillate in one specific direction (plane) can pass: the light becomes plane polarized light (only one vector shown; electric and magnetic vectors are still ⊥). ii. When plane polarized light passes through a solution of chiral molecules the plane of polarization is rotated by a certain angle in a certain direction, either clockwise or counterclockwise (that’s why chiral compounds are said to be optically active). Compounds that rotates light clockwise are called dextrorotatory (dexter is Greek for right); they are designated by (+) or d in front of their names. Compounds that rotate light to the left are called levorotatory (Greek for left); they are designated by a (–) or letter l in front of their names. Please, note that there is no direct connection between the direction of rotation (d or l) and the absolute configuration (R and S). 10 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth iii. Measuring Optical Rotation We can measure optical rotation, the direction and the degree (angle) of rotation, using an instrument called a polarimeter. The measured angle of rotation, α, depends on several factors, including the type of molecule and the number of molecules in the light path; this quantity is given by concentration, c, of the chiral substance and the distance light travels through the solution, the cell length, d. Other factors include the temperature and wavelength of the polarized light. We combine these factors in defining the specific rotation, [α], which is measured in a solution of concentration, c = 1.0g/mL, and a path length, l = 1.0 dm. The specific rotation is calculated from the measured rotation, α, by observed rotation [α] T λ specific rotation α = l × c length concentration The temperature (T) and wavelength (λ) are indicated by superscripts and subscripts, respectively. iv. Absence of Optical Activity. When we detect optical activity, we can be sure that a chiral compound is present. But, failure to observe optical activity does not mean that no chiral compound is present. Each compound with an asymmetric C atom has two enantiomers, each rotating light with the same magnitude of specific rotation, but rotating light in opposite directions, one to the right (+) and one to the left (–). If we have a 50-50 mixture of the pair of enantiomers, we observe NO rotation at all, because the two rotations, equal in magnitude but opposite in direction, cancel each other. 11 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth We call a 50-50 mixture of two enantiomers a racemic mixture or a racemate, represented by (±). Racemic mixtures are found very often: reactions generating a chiral carbon in a symmetrical environment form a racemic mixture. Why? The energies are identical, so entropy prevails, resulting in random attack. For example, the catalytic hydrogenation of 2-butanone (Figure 5.16) can occur either from “top” or from “bottom”; because both sides are equivalent, attack is equally likely: a racemate is formed. The conversion of one enantiomer into a racemic mixture is called racemization. For example, the (+)-isomer of thalidomide (see p. 5) racemizes in the human body, converting a sedative into a teratogen. 7. Optical purity In addition to 50-50 (racemic) mixtures of two enantiomers we may have mixtures that are not racemic. This may happen if an environment is not completely asymmetric or because racemization was stopped before completion. In such cases the observed rotation is due to the excess of one enantiomer over the other. We can determine the composition of the mixture (if we know the specific rotation of the enantiomers) by comparing the rotation observed for the mixture to the rotation of the pure enantiomers. We define the optical purity of the mixture as: % optical purity = [α] observed × 100% ––––––––––––––––––– [α] for pure enantiomer 12 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth Assume that the optical purity is 60%; this means that 60% is one pure enantiomer and that the remaining 40% is a racemic (50:50) mixture. 40% racemate means that one half of this (= 20%) is the same as the dominant enantiomer; we have a total of 60% + 20% = 80% of the dominant isomer in the nonracemic mixture. 8. Compounds with more than one asymmetric carbon If there are no other factors, the maximum number of stereoisomers for a n compound with n asymmetric carbons is 2 . This means that for 2, 3, 4 asymmetric carbons there are 4, 9, 16 stereoisomers (this is getting out of hand fast!). Let’s begin with the case where n =2 and two adjacent asymmetric atoms in a chain, bearing two different substituents. We consider 2-bromo-3chlorobutane. First we draw one isomer and its mirror image: H Br Br H 3 2 Cl H H 2-R,3-R Cl 2-S,3-S By applying our “rank/assign3D” procedure we determine that these isomers are the 2-R,3-R (left) and 2-S,3-S (right) stereoisomers; they are enantiomers. If we exchange H and Br in these stereoisomers we change (invert) the absolute configuration at that carbon, but we do not change the other carbon. The structures we have drawn are the 2-S,3-R (left) and 2-S,3-R (right) stereoisomers. You can see that these structures are mirror images of each other. But what is their relationship to the two 13 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth stereoisomers above? One stereocenter has the same absolute configuration, but the second one is different. We call such compounds diastereomers. Br H H Br 3 2 H Cl H 2-S,3-R Cl 2-R,3-S We have learned above that pairs of enantiomers have identical energies. In contrast, diastereomers have different energies. So if we compare the different features of stereoisomers, as we did above, we have to make one significant amendment: Geometric Isomers Stereo Isomers identical composition identical identical connectivity identical different arrangement in 3D space different identical (for enantiomers) different heat of combustion different (for diastereomers) Diastereomers also have different physical and chemical properties so they can be separated by crystallization or chromatography through a column that is packed with some chiral material. Note that enantiomers have RR vs. SS or RS vs. SR conformations whereas diastereomers have RR vs. RS or SR or SS vs RS or SR conformations. We can summarize the relationship of the four stereoisomers as follows: 14 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth 9. Compounds with two identically substituted stereocenters A compound with two equally substituted carbons has three stereoisomers. We consider 2,3-dibromobutane, related to the 2-bromo-3-chlorobutane discussed above. H Br Br 3 enantiomers 2 Br H H Br 2-S,3-S 2-R,3-R Br H 2 2-S,3-R H H Br 3 Br identical H 2 H H rotate C-2 by 180° Br 2-S,3-R Br H Br 2 internal mirror plane The R,R- and S,S- stereoisomers clearly are enantiomers (mirror images); in contrast, the presumed diastereomers, S,R- and R,S- are actually identical. You can verify this by rotating one of the stereocenters by 180°, as shown. The resulting structure has an internal mirror plane: it is symmetrical. We call this stereoisomer a meso compound. 15 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth Thus, a compound with two equally substituted carbons has two enantiomers (forming a racemate) and their one diastereomer, a meso compound. The two geometric isomers of 1,2-dimethylcyclopentane are diastereomers; the cis-isomer is a meso compound. CH3 H H H CH3 H CH3 CH3 We also consider the two geometric isomers of 2-butene diastereomers, because they are non-superimposable. CH3 H3C H CH3 H H3C H H The two compounds shown below are achiral for different reasons. 1-Bromo-3chloro-2,4-dimethylcyclobutane isomer on the left has a plane of symmetry; that means the two carbons bearing the methyl groups are chiral and the compound is a meso compound. The 1,3-dibromo-3,4-dimethylcyclobutane on the right has identical pairs of bromine-bearing and methyl-bearing carbons. Accordingly, none of the carbons is chiral. H 3C H 3C Br Br CH3 • Br Cl Achiral due to plane of symmetry CH3 Achiral due to center of symmetry 16 Organic Chemistry Chapter 5 Stereoisomers H. D. Roth 10) Fischer projection The German chemist Emil Fischer was a pioneer of the chemistry of carbohydrates (sugars) that contain multiple –OH functions and stereocenters. In order to visualize these compounds Fischer used a dash and wedge projection without actually showing the dashes and wedges. The projection consists of intersecting perpendicular lines representing the four bonds of an asymmetric carbon located at their intersection. By convention, the two vertical lines are dotted, the two horizontal lines are wedges; the main carbon chain is oriented vertically with the lowest numbered carbon at the top. This means that all substituents are eclipsed. We can derive the Fischer projection of R,R-2,3dibromobutane in two steps as shown below. CH3 H Br 3 rotate 2 Br H CH3 Br C H Br H H C Br H Br CH3 CH3 We will not use the Fischer projection in this class, but note that an in plane rotation of a Fischer projection by 180° will leave the absolute configuration unchanged, whereas a rotation by 90° will convert S to R or R to S, because all substituents change (by definition) from “above the plane” to “below the plane” and by “below the plane” to “above the plane”. 17