MasteringPhysics: Print View with Answers
1 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
Signed in as Weida Wu , Instructor
Rutgers Analytical Physics 750:228, Spring 2016
My Courses
Help
Sign Out
( RUPHY228S16 )
Course Settings
University Physics with Modern Physics, 14e
Young/Freedman
Instructor Resources
Course Home
eText
Assignments
Study Area
Roster
Gradebook
Item Library
10. Schrodinger Equation, 1D Quantum Mechanics, 41.1-3,Many-Electron Atoms: 41.3-7
Overview
Summary View
Diagnostics View
[ Edit ]
Print View with Answers
10. Schrodinger Equation, 1D Quantum Mechanics, 41.1-3,Many-Electron Atoms: 41.3-7
Due: 11:59pm on Sunday, April 10, 2016
To understand how points are awarded, read the Grading Policy for this assignment.
Selection Rules
Description: Multiple-choice questions about the selection rules for electron transitions.
Because of conservation of angular momentum, there are certain selection rules for determining whether a particular transition between atomic states is allowed. There are
two rules:
1.
,
2.
.
Any transition that does not meet these requirements is called a forbidden transition and will not occur.
Part A
Which of the following is an allowed transition?
ANSWER:
to
to
to
to
Part B
Which of the following is a forbidden transition?
ANSWER:
to
to
to
to
Part C
In the Zeeman effect, the energy levels of hydrogen are split by a magnetic field. Each state with a different value of
has a different energy. The differences in energy
between adjacent values of
are all equal. The Zeeman effect is observed as a splitting in the spectral lines of hydrogen in the presence of a magnetic field. What is the
greatest number of lines that a single spectral line can be split into by a magnetic field?
Hint 1. What determines the line energies?
In the absence of magnetic fields, the energy of a spectral line is determined by the initial and final values of . Since the energies for different values of
are
equally spaced in the Zeeman effect, the change in
will determine the energies of the new lines. How many values for the change in
are possible? Think
about the selection rules.
ANSWER:
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
2 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
2
3
4
5
6
Part D
Suppose that a hydrogen atom is in an excited state such that its electron is found in the state
this electron to decay into?
1.
2.
3.
. Which of the following is an allowable state for
,
,
.
ANSWER:
1
2
3
1 and 2
1 and 3
2 and 3
Rules for Orbital Angular Momentum
Description: The basic rules for allowable orbital angular momentum and magnetic quantum numbers are given and then applied to finding the available states in various
situations.
Learning Goal:
To understand and be able to use the rules for determining allowable orbital angular momentum states.
Several numbers are necessary to describe the states available to an electron in the hydrogen atom. The principal quantum number determines the energy of the electron.
The orbital quantum number determines the total angular momentum of the electron, and the magnetic quantum number
determines the component of the angular
momentum parallel to a specific axis, usually the z axis.
For a given principal quantum number , the orbital quantum number can take integer values ranging from zero to
. For a given orbital quantum number , the magnetic
quantum number can take integer values from
to . A fourth number, the spin
, is important for interactions with magnetic fields and counting states. The spin can be
either
or
, independent of the values of the other quantum numbers.
The energy of an electron in hydrogen is related to the principal quantum number by
. The orbital angular momentum is related to the orbital quantum
number by
, and the orbital angular momentum in the z direction is related to the magnetic quantum number by
.
Part A
How many different values of are possible for an electron with principal quantum number
= 3?
Express your answer as an integer.
ANSWER:
= 3.00
Part B
How many values of
are possible for an electron with orbital quantum number = 3?
Express your answer as an integer.
ANSWER:
= 7.00
Part C
The quantum state of a particle can be specified by giving a complete set of quantum numbers
principal quantum number is = 5?
. How many different quantum states are possible if the
To find the total number of allowed states, first write down the allowed orbital quantum numbers , and then write down the number of allowed values of
for each orbital
quantummath
number. Sum these quantities, and then multiply by 2 to account for the two possible orientations of spin.
Typesetting
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
3 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
Express your answer as an integer.
Hint 1. Find the number of states for
Keeping in mind that
can assume only integer values in the range from
to , how many states are possible for
?
to , how many states are possible for
?
Express your answer as an integer.
ANSWER:
2
Hint 2. Find the number of states for
Keeping in mind that
can assume only integer values in the range from
Express your answer as an integer.
ANSWER:
6
ANSWER:
2*n_2^2 = 50.0
Part D
Is the state
,
,
,
an allowable state? If not, why not?
ANSWER:
Yes it is an allowable state.
No: The magnetic quantum number must equal the principal quantum number.
No: The orbital quantum number cannot equal the principal quantum number.
No: The magnetic quantum number cannot be negative.
No: The magnetic quantum number must equal the orbital quantum number.
Part E
What is the maximum angular momentum
that an electron with principal quantum number
= 3 can have?
Express your answer in units of . (You don't need to enter the , it is in the units field for you.)
Hint 1. Find the maximum allowed value for
Recall from the introduction that the orbital angular momentum is related to the orbital quantum number via the equation
. The maximum angular
momentum will be associated with the largest allowed value for . For = 3, what is the maximum allowed value
for the orbital quantum number?
Express your answer as an integer.
ANSWER:
=
= 2.00
ANSWER:
=
= 2.45
Exercise 41.14
Description: (a) Make a chart showing all the possible of quantum numbers l and m_l for the states of the electron in the hydrogen atom when the principal quantum
number is n. How many combinations are there? (b) What are the energies of these states?
Part A
Make a chart
Typesetting
mathshowing all the possible of quantum numbers and
for the states of the electron in the hydrogen atom when the principal quantum number is
= 3. How
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
4 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
many combinations are there?
ANSWER:
=
=9
Part B
What are the energies of these states?
Express your answer using four significant figures.
ANSWER:
=
= -1.511
Exercise 41.19
Description: A hydrogen atom in a 3p state is placed in a uniform external magnetic field B_vec. Consider the interaction of the magnetic field with the atom's orbital
magnetic dipole moment. (a) What field magnitude B is required to split the 3p state into...
A hydrogen atom in a
state is placed in a uniform external magnetic field
. Consider the interaction of the magnetic field with the atom's orbital magnetic dipole moment.
Part A
What field magnitude
is required to split the
state into multiple levels with an energy difference of
between adjacent levels?
ANSWER:
=
= 0.465
Also accepted:
= 0.465,
= 0.465,
= 0.465
Part B
How many levels will there be?
ANSWER:
3
Exercise 41.21
Description: A hydrogen atom in the 5g state is placed in a magnetic field of B that is in the z-direction. (a) Into how many levels is this state split by the interaction of the
atom's orbital magnetic dipole moment with the magnetic field? (b) What is...
A hydrogen atom in the
state is placed in a magnetic field of 0.250
that is in the -direction.
Part A
Into how many levels is this state split by the interaction of the atom's orbital magnetic dipole moment with the magnetic field?
ANSWER:
= 9
Part B
What is the energy separation between adjacent levels?
ANSWER:
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
5 of 14
=
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
= 2.32×10−24
Also accepted: 1.602*10^-19*h*B/(4*pi*m_e) = 2.32×10−24,
= 2.32×10−24,
= 2.32×10−24
Part C
What is the energy separation between the level of lowest energy and the level of highest energy?
ANSWER:
= 2*e*h*B/(pi*m_e) = 1.85×10−23
Also accepted: 2*1.602*10^-19*h*B/(pi*m_e) = 1.85×10−23,
= 1.85×10−23, 2*e*h*B/(pi*m_e) = 1.85×10−23
The Zeeman Effect
Description: The Zeeman effect is described and the main results are derived.
Learning Goal:
To understand and be able to perform simple calculations relating to the Zeeman effect.
The Zeeman effect is a change in the energy levels of electrons in an atom in the presence of a magnetic field. It can be observed as the spliting of spectral lines when the
atom is in a strong magnetic field. To understand this effect, you need to recall a few facts about magnetic moments.
When a loop of wire with a current running through it is placed in a magnetic field, it acquires a potential energy
, where is the magnetic field and is the
magnetic moment of the loop. The magnetic moment is a vector perpendicular to the plane of the loop of wire, with its direction determined by applying the right-hand rule to
the direction of current flow. The magnitude of is given by
, where is the current in the loop and is the area enclosed by the loop.
In the Bohr model of the atom, electrons follow circular orbits around the nucleus. Such an orbit would constitute a loop of current, since the electron has charge and moves
repeatedly through the same circular path. While the Bohr model is fundamentally incorrect, its conclusions about electrons interacting with magnetic fields can be used as a
basis for correctly understanding the Zeeman effect.
Consider an electron moving in a circular orbit of radius
at a speed . The orbit will serve as the loop of current as you calculate the magnetic moment.
Part A
What is the area
enclosed by the electron's orbit?
Express your answer in terms of .
ANSWER:
= pi*r^2
Part B
What is the current
an electron.
due to the orbit of the electron? Recall that current is defined as the amount of charge passing a point in one unit of time. Use
for the charge on
Express your answer in terms of , , and .
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
6 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
Hint 1. How to approach the problem
A particle with charge passing a point times per second would give the same current as different charges each with charge passing every second.
Therefore, the current would be
. The number of times that the particle passed could also be written
, where is the time it takes for the particle to
pass the point again after it passes it once. If the particle were following a specific path, then would be the time required for the particle to traverse that path
once.
Since the only charged particle is the electron, the current due to the orbit is just the charge on the electron multiplied by the number of times that the electron
passes through a certain point in one unit of time. As described above, this is the same as the charge on the electron multiplied by
, where is the time
required for one orbit. Simply divide the charge on the electron by to obtain the current.
Hint 2. Find the time for the electron to complete one orbit
Find the time \texttip{T}{T} for the electron to complete one orbit. Recall the formula for the circumference of a circle: C=2\pi r.
Express your answer in terms of \texttip{v}{v} and \texttip{r}{r}.
ANSWER:
\texttip{T}{T} = (2*pi*r)/v
ANSWER:
\texttip{I}{I} = -e*v/(2*pi*r)
Part C
What is the magnitude \mu = |\vec{\mu}| of the magnetic moment for the orbiting electron?
Express your answer in terms of \texttip{e}{e}, \texttip{v}{v}, and \texttip{r}{r}.
ANSWER:
\texttip{\mu }{mu} = e*v*r/2
Part D
In the Schrödinger picture of the hydrogen atom, the electrons are waves with associated probability distributions. Thus, \texttip{v}{v} and \texttip{r}{r} are not well-defined
quantities. However, the angular momentum is a well-defined quantity. Use the classical definition of angular momentum L=mvr, where \texttip{m}{m} is the mass of the
electron, to find an expression for the magnitude \texttip{\mu }{mu} of the magnetic moment of the electron.
Express your answer in terms of \texttip{L}{L}, \texttip{e}{e}, and \texttip{m}{m}.
ANSWER:
\texttip{\mu }{mu} = e*L/(2*m)
Part E
Consider a magnetic field of magnitude \texttip{B}{B} oriented along the z axis. The energy associated with the interaction of this field with an orbiting electron with
magnetic moment \texttip{\mu }{mu} is U=-\vec{\mu}\cdot \vec{B}. Since you know that the magnetic field is oriented along the z axis, this inner product will be equal to
-\mu_z B, where \texttip{\mu _{\mit z}}{mu_z} is the z component of the magnetic moment. Since the only part of the expression for the electron's magnetic momentum
that has direction is the angular momentum \texttip{L}{L}, we conclude that \mu_z=-eL_z/(2m).
You know that in the Schrödinger picture of the hydrogen atom, the angular momentum component in a specific direction (here we are concerned with the z direction) is
equal to \hbar m_l, where \texttip{m_{\rm l}}{m_l} is the magnetic quantum number of the electron's state. What is the magnitude of the magnetic potential energy
\texttip{U}{U} for an electron with magnetic quantum number \texttip{m_{\rm l}}{m_l} in a magnetic field of strength \texttip{B}{B}?
Express your answer in terms of \texttip{m_{\rm l}}{m_l}, \texttip{e}{e}, \texttip{B}{B}, \texttip{\hbar }{hbar}, and \texttip{m}{m}.
ANSWER:
\texttip{U}{U} = e*m_l*hbar*B/(2*m)
Part F
An electron in a hydrogen atom is in a state with magnetic quantum number m_l=2. How much does the energy of this state change when a magnetic field of magnitude
1.5 tesla is turned on? Use e=1.602\times10^{-19}\; {\rm C}, \hbar=1.055\times10^{-34}\;{\rm J\cdot s}, m=9.11\times10^{-31}\; {\rm kg}, and the conversion 1\; {\rm
eV}=1.602\times10^{-19}\; {\rm J}.
Express your answer in electron volts to three significant figures.
ANSWER:
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
7 of 14
\texttip{U}{U} = 1.74×10−4
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
\rm eV
The Zeeman effect serves as a strong confirmation of the Schrödinger model of the atom. It also gives a way to measure the magnetic field of sunspots, by
measuring how strongly the spectral lines of hydrogen near a sunspot are split.
The Electron Spin
Description: Calculate the interaction energy for an electron in a magnetic field, followed by a multiple choice question about allowable values of the total angular
momentum J.
You know that an electron experiences the effect of an electric field because it has electric charge. An electron also experiences magnetic fields, because it has intrinsic
angular momentum known as spin. The strength of the electron's interaction with a magnetic field is determined by its magnetic moment \texttip{\mu_z}{mu_z}, given by
\large{\mu_{z} = 2.00232 \frac{e}{2m}S_{z}},
where in the case of the electron,
\large{S_{z} = \pm \frac{1}{2}\hbar}.
The factor 2.00232 comes from quantum electrodynamics.
Part A
Find the interaction energy \texttip{U}{U} of an electron in an atom with orbital quantum number l=0 with a uniform 2.00-tesla magnetic field.
Express your answer in electron volts to three significant figures. Give only the absolute value. This means that you should not include the \pm sign. Use
\mu_{\rm B}=5.788 \times 10^{-5} eV/T for the Bohr magneton.
Hint 1. Potential energy of an electron in a magnetic field
The potential energy of an electron in a magnetic field is given by U = -\mu_z B_z. Assume that all of the magnetic field is directed along the z component of the
electron's magnetic moment.
Hint 2. Potential energy of an atom with l=0 in a magnetic field
Since l=0, there is no potential energy due to the orbital angular momentum of an atom. Thus, all potential energy in the magnetic field is due to the electron's spin.
ANSWER:
\texttip{U}{U} = 1.159×10−4 \rm eV
Part B
What is the energy difference \texttip{\Delta E}{Delta E} between the two split states?
Express your answer in electron volts to three significant figures.
ANSWER:
\texttip{\Delta E}{Delta E} = 2.318×10−4 \rm eV
Part C
Due to the spin of an electron \texttip{\vec{S}}{S_vec}, orbital angular momemtum \texttip{\vec{L}}{L_vec} is not sufficient to explain the behavior of an atom. A better
quantum number is the total angular momentum. The total angular momentum \texttip{\vec{J}}{J_vec} of an atom is given by \vec{J} = \vec{L} + \vec{S}. Just as
\texttip{\vec{L}}{L_vec} has an associated quantum number (the orbital quantum number \texttip{l}{l}), \texttip{\vec{J}}{J_vec} has the associated total angular quantum
number \texttip{j}{j}. If the orbital quantum number is l=1, what are the possible value(s) of the total angular quantum number \texttip{j}{j}?
Hint 1. Adding \texttip{l}{l} and \texttip{m_{\rm s}}{m_s}
The total angular quantum number \texttip{j}{j} for a system with orbital quantum number \texttip{l}{l} and spin quantum number \texttip{m_{\rm s}}{m_s} is given by
j=|l+m_s|.
Hint 2. Values of \texttip{m_{\rm s}}{m_s}
Recall that an electron can have spin quantum number of \large{+\frac 1 2} or \large{- \frac 1 2}.
ANSWER:
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
8 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
\large{j = \frac{1}{2}}
\large{j = \frac{3}{2}}
\large{j = \pm \frac{1}{2}}
\large{j = \frac{1}{2}} or \large{\frac{3}{2}}
Many-Electron Atoms
Description: Conceptual questions concerning the approximations needed to deal with many-electron atoms and their results.
Learning Goal:
To understand how electrons are placed into subshells as atomic number increases and how this leads to the trends of the periodic table.
The Schrödinger equation was highly successful at describing the hydrogen atom. Unfortunately, for any other atom the equation becomes too complex to solve, because of
the interactions of the electrons with one another. Luckily, approximations can be made to get some useful results, including an explanation of the periodic table of elements!
The most useful approximation is called the central field approximation. In the central field approximation, the potential energy is described by a spherically symmetric function
(i.e., a function dependent only on distance from the center of the atom). In this approximation, it is assumed that the electrons closest to the nucleus (n=1) experience a force
from the full charge on the nucleus, but electrons further out experience force from only a fraction of that charge, because the inner electrons "screen" the nucleus.
To understand this, think of the probability distribution of the electrons as a spherically symmetric charge distribution. From Gauss's law, you know that the electric field at a
distance \texttip{r}{r} from the nucleus will be proportional to the net charge contained within a sphere of radius \texttip{r}{r}. Since such a sphere will always contain the
positive charge on the nucleus, increasing \texttip{r}{r} leads to a decreased net charge, as more of the charge from the electrons is included to cancel the nucleus's charge.
This approximation tells us several important things. First, since the potential is spherically symmetric, the electrons must have the same angular wave functions \texttip{\Theta
\left(\theta \right)}{Theta(theta)} and \texttip{\Phi \left(\phi \right)}{Phi(phi)} as the hydrogen atom. This means that the same quantum numbers \texttip{l}{l} and m_l will be used
to describe electrons in many-electron atoms, and that the same rules that apply to these numbers in hydrogen will apply in many-electron atoms.
Part A
Which of the following gives the correct permitted values of m_l for l=2?
ANSWER:
-1\; 0\; 1
0\; 1\; 2
0\; -1\; -2
0\; -1
0\; 1
-2\; -1\; 0\; 1\; 2\;
Part B
A second important result is that electrons will fill the lowest energy states available. This would seem to indicate that every electron in an atom should be in the n=1 state.
This is not the case, because of Pauli's exclusion principle. The exclusion principle says that no two electrons can occupy the same state. A state is completely
characterized by the four numbers \texttip{n}{n}, \texttip{l}{l}, m_l, and m_s, where m_s is the spin of the electron.
An important question is, How many states are possible for a given set of quantum numbers? For instance, n=1 means that l=0 with m_l=0 are the only possible values for
those variables. Thus, there are two possible states: (1,\ 0,\ 0,\ 1/2) and (1,\ 0,\ 0,\ -1/2). How many states are possible for n=2?
Express your answer as an integer.
ANSWER:
8 states
Part C
Because the amount of screening of the nucleus is related to distance from the nucleus, the s and p subshells of a many-electron atom with the same value of
\texttip{n}{n} are not of equal energy, as they are in hydrogen. An electron in the p subshell has a lower probability of being inside the electrons of the n=1 shell than does
an electron in the s subshell. Thus, the nucleus is screened somewhat more completely for electrons in the p subshell than for electrons in the s subshell. This makes
states in the p subshell of higher energy than states in the s subshell.
The electron structure of an atom shows how many electrons are in each subshell. For instance, helium's electron structure is written 1s^2, signifying the two electrons in
the 1s subshell. Neon, with ten electrons, has electron structure 1s^22s^22p^6, signifying two electrons in the 1s subshell, two in the 2s subshell, and six in the 2p
subshell. Observe that the exponents sum to the number of electrons in the atom.
Which of the following is not a valid electron structure for an atom in its ground state? Use the fact that electrons fill the lowest energy states first and keep in mind the
number of allowed states for each subshell.
ANSWER:
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
9 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
1s^22s^22p^63s^3
1s^22s^22p^63s^23p^4
1s^22s^22p^63s^2
1s^22s^22p^63s^23p^5
1s^22s^22p^5
Part D
For states with larger values of \texttip{n}{n} that admit a d subshell, the d subshell is at even higher energy than the p subshell. In fact, its energy is higher than that of the
s subshell for the n+1 state. With this in mind, which of the following are valid ground-state electron structures?
1. 1s^22s^22p^63s^23p^63d^2
2. 1s^22s^22p^63s^23p^63d^{11}
3. 1s^22s^22p^63s^23p^64s^23d^5
ANSWER:
1
2
3
1 and 2
1 and 3
2 and 3
Part E
The structure of the periodic table is based on the order in which electron subshells are filled with increasing
atomic number \texttip{Z}{Z}. You know that the first two electrons go into the 1s subshell and correspond to
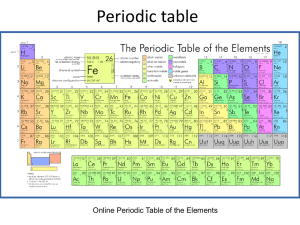
hydrogen and helium (red in the top row). The next two electrons go into the 2s subshell. The elements with
Z=3 and Z=4 are lithium and beryllium, respectively (red, second row). The periodic table shown in the figure
has each element colored based on the subshell in which the highest-energy electron is found. Which of the
following correctly pairs the colors with the subshells that they indicate?
ANSWER:
red=p yellow=s blue=d
red=s yellow=p blue=d
red=d yellow=p blue=s
red=s yellow=d blue=p
red=p yellow=d blue=s
Part F
Why do the elements with d subshell electrons not appear until the fourth row, even though there is a d subshell for n=3?
ANSWER:
Electrons in the d subshell do not have noticeable chemical activity for n=3.
The d subshell is at higher energy than the s subshell with the next-higher value of \texttip{n}{n}.
Pauli's exclusion principle does not allow electrons into the d subshell for n=3.
Since the first row actually corresponds to n=0 it follows that the fourth row is the correct place for the d subshell with n=3.
Electron Shell Conceptual Questions
Description: Conceptual questions on electron shell structure.
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
10 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
Consider an electron in the state n = 4, l = 3, m = 2, s = 1/2.
Part A
In what shell is this electron located?
Hint 1. Electron shells
The terminology for electron shells is based exclusively on the principal quantum number, \texttip{n}{n}. For n = 1, 2, 3, 4, 5,\ldots the electron is said to be in shell
K, L, M, N, O,\ldots respectively.
ANSWER:
Part B
In what subshell is this electron located?
Hint 1. Electron subshells
The terminology for electron subshells is based exclusively on the angular momentum quantum number,\texttip{l}{l}. For l = 0, 1, 2, 3, 4,\ldots the electron is said to
be in subshell s, p, d, f, g, \ldots respectively.
ANSWER:
Part C
How many other electrons could occupy the same subshell as this electron?
Hint 1. Maximum electron occupancy
Each subshell has a maximum possible electron occupancy. This occurs because of the Pauli exclusion principle. The exclusion principle states that no two
electrons can share exactly the same state, where an electron's state is defined as the value of its four quantum numbers.
For example, consider the \texttip{3d}{3d} subshell. Since l = 2 (corresponding to the \texttip{d}{d} designation), the magnetic quantum number can take on the
values m = 2, 1, 0, -1,and -2. The spin quantum number can always take on the values +1 and -1. Thus, for each of the five possible \texttip{m}{m} values, there
are two possible \texttip{s}{s} values, resulting in 10 (the product of the possibilities for \texttip{m}{m} and the possibilities for \texttip{s}{s}) possible electron states
inside the \texttip{3d}{3d} subshell. Thus, the maximum number of \texttip{3d}{3d} electrons is 10.
Hint 2. A formula for electron occupancy
Since the magnetic quantum number \texttip{m}{m} can always take on values ranging from \texttip{l}{l} to -l, this corresponds to (2l + 1) possible values for
\texttip{m}{m}. The spin quantum number always has only two possible values (\pm1), so the total number of possible states inside any particular subshell is given
by the relationship
\rm maximum\; occupancy = 2(2l + 1).
ANSWER:
Part D
What is the orbital angular momentum \texttip{L}{L} of this electron?
Express your answer in units of \hbar.
Hint 1. Orbital angular momentum
The orbital angular momentum, \texttip{L}{L}, of an electron is determined exclusively by its angular momentum quantum number,\texttip{l}{l}, via the relationship
L = \sqrt{l(l + 1)}\, \hbar.
ANSWER:
\texttip{L}{L} = 3.46
\hbar
Part E
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
11 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
What is the z component of the orbital angular momentum of this electron, \texttip{L_{\mit z}}{L_z}?
Express your answer in units of \hbar.
Hint 1. The z component of orbital angular momentum
The z component of orbital angular momentum, \texttip{L_{\mit z}}{L_z}, of an electron is determined exclusively by its magnetic quantum number, \texttip{m}{m},
via the relationship
L_z = m \,\hbar.
ANSWER:
\texttip{L_{\mit z}}{L_z} = 2
\hbar
Part F
What is the z component of the spin angular momentum of this electron, \texttip{S_{\mit z}}{S_z}?
Express your answer in units of \hbar.
Hint 1. The z component of spin angular momentum
The z component of spin angular momentum, \texttip{S_{\mit z}}{S_z}, of an electron is determined exclusively by its spin quantum number, \texttip{s}{s}, via the
relationship
S_z = s \,\hbar.
ANSWER:
\texttip{S_{\mit z}}{S_z} = 0.500
\hbar
Electron States Conceptual Questions
Description: Conceptual questions on whether hypothetical electron states can exist.
Consider the six hypothetical electron states listed in the table.
\texttip{n}{n}
\texttip{l}{l}
\texttip{m_{\rm l}}{m_l}
\texttip{m_{\rm s}}{m_s}
A
3
1
-1
0
B
3
1
0
\large{-\frac{1}{2}}
C
3
0
+1
\large{-\frac{1}{2}}
D
2
2
0
\large{+\frac{1}{2}}
E
2
-1
0
\large{-\frac{1}{2}}
F
2
0
0
\large{+\frac{1}{2}}
Part A
Which, if any, of these states are not possible?
Select the letters of the states that are not possible.
Hint 1. Angular momentum quantum number
The angular momentum quantum number, \texttip{l}{l}, for a valid electron state ranges from zero to one less than the principal quantum number, i.e.,l = 0, 1, 2,
\ldots, n-1.
Hint 2. Magnetic quantum number
The magnetic quantum number, \texttip{m}{m}, for a valid electron state must be an integer in the range between \texttip{l}{l} and -l, i.e., m = 0, \pm 1, \pm 2,\ldots,
\pm l.
Hint 3. Spin quantum number
The spin quantum number, \texttip{s}{s}, for a valid electron state must be either positive or negative \large{\frac{1}{2}}, i.e., \large{s = \pm \frac{1}{2}}.
ANSWER:
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
12 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
A
B
C
D
E
F
Part B
List the spectroscopic notation for state B.
Hint 1. Spectroscopic notation
The spectroscopic notation for an electron state is the numerical value of the principal quantum number followed by a letter representing the angular momentum
quantum number. For example, the state n = 4 and l = 3 would be referred to as \texttip{4f}{4f}.
Hint 2. Electron subshells
The terminology for electron subshells is based exclusively on the angular momentum quantum number, \texttip{l}{l}. For l = 0, 1, 2, 3, 4,\ldots the electron is said to
be in subshell s, p, d, f, g, \ldots, respectively.
ANSWER:
Part C
List the spectroscopic notation for state F.
Hint 1. Spectroscopic notation
The spectroscopic notation for an electron state is the numerical value of the principal quantum number followed by a letter representing the angular momentum
quantum number. For example, the state n = 4 and l = 3 would be referred to as \texttip{4f}{4f}.
Hint 2. Electron subshells
The terminology for electron subshells is based exclusively on the angular momentum quantum number, \texttip{l}{l}. For l = 0, 1, 2, 3, 4,\ldots the electron is said to
be in subshell s, p, d, f, g,\ldots respectively.
ANSWER:
Screening the Nucleus
Description: Qualitative questions about screening followed by a pair of calculations of Z_eff.
One facet of the central field approximation for many-electron atoms is that inner-shell electrons screen the nuclear charge. To understand how this works quantitatively, first
note that the probability distributions for electrons in different shells generally do not overlap much. For instance, the electrons in the M shell (n=3) are almost always farther
from the nucleus than the electrons of the K (n=1) and L (n=2) shells. Thus, it is a good approximation to assume that the inner shells completely screen the nucleus from the
outer shells. For example, if there are ten electrons altogether in the K and L shells of an atom, then the electrons in the M shell experience force from a charge of roughly
Z-10, where \texttip{Z}{Z} is the charge on the nucleus as an integer multiple of \texttip{e}{e}, the magnitude of the charge on an electron. This is called the effective nuclear
charge \texttip{Z_{\rm eff}}{Z_eff}.
Part A
In a beryllium atom (Z=4), how many electrons are in the K shell?
Express your answer as an integer.
ANSWER:
2 electrons
Part B
In xenon (Z=54), what is the effective charge \texttip{Z_{\rm eff}}{Z_eff} experienced by an electron in the M (n=3) shell?
Express your answer as an integer.
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
13 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
Hint 1. Find the number of electrons in the K and L shells
How many electrons are there altogether in the K and L shells of xenon? Recall that for n=2, the orbital quantum number \texttip{l}{l} must be zero or one and that
\texttip{m_{\rm l}}{m_l} can take any value between postive and negative \texttip{l}{l}.
Express your answer as an integer.
ANSWER:
10 electrons
Now just subtract this number of electrons from the atomic number, because these electrons will be closer to the nucleus than the M-shell electrons. Thus, by
Gauss's law, you will obtain the effective charge that M-shell electrons experience.
ANSWER:
\texttip{Z_{\rm eff}}{Z_eff} = 44
e
Part C
How many electrons are there altogether in the K, L, and M shells of xenon? Recall that for n=3, the orbital quantum number \texttip{l}{l} must be zero, one, or two and
that \texttip{m_{\rm l}}{m_l} can take any value between postive and negative \texttip{l}{l}.
Express your answer as an integer.
ANSWER:
28 electrons
The idea of simply subtracting the number of inner-shell electrons works well only for atoms with low values of \texttip{Z}{Z}. For atoms with larger nuclear charge, the different
shapes of the probability distributions for different subshells becomes an important factor. For instance, in the N shell, electrons in the s subshell have a much higher
probability than those in the p or d subshells of being found closer to the nucleus than some of the K, L, or M electrons. It is said that the s electrons penetrate the inner shells
more readily than do the p or d electrons. Therefore, it is to be expected that our simple subtraction model may not work well for outer-shell s electrons in large atoms.
Keeping this in mind, we see that the effective nuclear charge \texttip{Z_{\rm eff}}{Z_eff} for the outermost electron in an atom may be found experimentally by measuring the
ionization energy of an atom and then calculating the effective charge using the equation
\large{E_n=- \frac{Z_{\rm eff}^2}{n^2}(13.6\;\rm eV)}.
Part D
The energy for the 5p valence electron in indium (Z=49) is -5.79 electron volts. What is the effective nuclear charge \texttip{Z_{\rm eff}}{Z_eff} experienced by this
electron?
Express your answer to three significant figures.
ANSWER:
\texttip{Z_{\rm eff}}{Z_eff} = 3.26
e
The simple subtraction model in the introduction predicts 3 as the effective nuclear charge. This is in reasonable agreement with your calculation for the 5p electron
in this part.
Part E
The energy for the 5s valence electron in rubidium (Z=37) has energy -4.18 electron volts. What is the effective nuclear charge \texttip{Z_{\rm eff}}{Z_eff} experienced by
this electron?
Express your answer to three significant figures.
ANSWER:
\texttip{Z_{\rm eff}}{Z_eff} = 2.77
e
The simple subtraction model in the introduction predicts 1 as the effective nuclear charge. Though it was still reasonably accurate for the p subshell electron in Part
D, you can see that the model breaks down badly for s-subshell eletrons, which penetrate the inner shells rather readily.
Exercise 41.25
Description: (a) Calculate the energy difference between the (normalsize m_s= )(1/2) ("spin up") and (normalsize m_s= - )(1/2) ("spin down") levels of a hydrogen atom
in the 1s state when it is placed in a B magnetic field in the negative z-direction...
Typesetting math
4/22/2016 12:19 AM
MasteringPhysics: Print View with Answers
14 of 14
https://session.masteringphysics.com/myct/assignmentPrintView?assign...
Part A
Calculate the energy difference between the \large{{\normalsize m_s= \,}{\frac{1}{2}}} ("spin up") and \large{{\normalsize m_s= - \,}{\frac{1}{2}}} ("spin down") levels of a
hydrogen atom in the 1s state when it is placed in a 1.20 {\rm {\rm T}} magnetic field in the negative z-direction.
ANSWER:
\Delta E = (2.00232)*(5.788*10^-5)*B = 1.39×10−4
{\rm eV}
Also accepted: (2.00232)*(5.79*10^-5)*B = 1.39×10−4, (2.00232)*(5.788*10^-5)*B = 1.39×10−4
Part B
Which level, \large{{\normalsize m_s= \,}{\frac{1}{2}}} or \large{{\normalsize m_s= - \,}{\frac{1}{2}}}, has the lower energy?
ANSWER:
\large{{\normalsize m_s= \,}{\frac{1}{2}}}
\large{{\normalsize m_s= - \,}{\frac{1}{2}}}
Exercise 41.31
Description: The 5s electron in rubidium Rb sees an effective charge of 2.771e. (a) Calculate the ionization energy of this electron.
The 5s electron in rubidium ({\rm Rb}) sees an effective charge of 2.771e.
Part A
Calculate the ionization energy of this electron.
Express your answer using three significant figures.
ANSWER:
E_5 = 4.18
{\rm eV}
Copyright © 2016 Pearson. All rights reserved.
Legal Notice
Privacy Policy
Permissions
Support
Typesetting math
4/22/2016 12:19 AM