AACR 2016 Abstract # 1217
advertisement

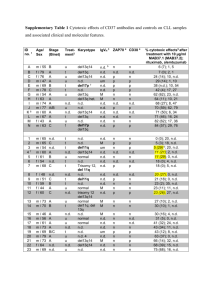
Preclinical evaluation of a next-generation, EGFR targeting ADC that promotes regression in KRAS or BRAF mutant tumors AACR 2016 Abstract # 1217 Lei Huang1, Bob Veneziale1, Mark Frigerio2, George Badescu2, Xiaoming Li1, Qiping Zhao1, Jesse Bahn1, Jennifer Souratha1, Ryan Osgood1, Chunmei Zhao1,Kim Phan1, Jessica Cowell1, Sanna Rosengren1, Anas Fathallah1, Jason Parise1, Martin Pabst2, Mathew Bird2, William McDowell2, Gina Wei1, Curtis Thompson1, Antony Godwin2, H. Michael Shepard1, and Christopher Thanos1 1Halozyme Therapeutics, San Diego, CA, USA; 2Abzena, Antitope Limited & PolyTherics Limited, Cambridge, United Kingdom ABSTRACT RESULTS RESULTS (cont.) RESULTS (cont.) • • ThioBridge™ ADC conjugation was performed as described in Ref 10. MMAE corresponds to monomethyl auristatin E. • Tumor xenograft growth studies were conducted in Ncr nu/nu mice. MDA-MB-231 cells were implanted in the mammary fat pad, and HT-29 (colorectal adenocarcinoma) cells were injected subcutaneously, both at 3.3 x 106 cells/site. • • • • Mutation promotes cell growth and is resistant to mAb therapy ADC overcomes mutation resistance and selectively kills tumor cell Naked mAb EGF EGFR 20 30 40 50 60 70 80 90 3 Summary 500 100 0 10 20 30 40 50 60 70 80 90 100 110 T im e ( D a y s ) Dosing stopped at Day 38 Dosing stopped at Day 39 Fig. 6 Targeting EGFR+, mutant tumors with an ADC. Halo Variant 1-MMAE conjugates (maleimide based conjugation, vc-PAB-MMAE) were used to dose EGFR+, mutant tumors (bi-weekly) at either 1, 3, 10, or 30 mg/kg. Cetuximab was dosed bi-weekly at 30 mg/kg. A dose response for Halo Variant 1-MMAE was observed in both models. In both models, no activity was observed for cetuximab. N=6 mice group. 1 2 3 4 5 6 7 0 1 2 Random Lysine Conjugation Limited dermal scoring findings comparable with vehicle control group • No unexpected findings observed at either dose (2.5 mg/kg and 8 mg/kg) • Safety profile met criteria for candidate nomination HTI-1511 In Vivo Stability in Primates HALO mAb Maleimide MMAE HTI-1511 ER: 91% 3 4 5 25.0 6 7 8 9 0 1 2 3 4 5 6 7 20.0 5.0 12.4 min (1.8 %) -2.0 7.5 Exposure Ratio (ER) = Group Mean AUC of ADC/Group Mean AUC of Total x 100 HTI-1511 Potency in EGFR+, KRASMUT PDx Models NSCLC (EGFR+, KRASpG12C) PDx Fig. 8 Generation of HTI-1511. The HALO anti-EGFR mAb (see Methods section) generated an ADC through use of ThioBridge conjugation technology, and linked with a branched PEG spacer to a val-cit PAB-MMAE warhead, resulting in isolation of 98.2% DAR 4 species preparation, as assessed by analytical HIC. MMAE PEG side chain 10.0 5.0 Fig. 10 PK and in vivo ADC stability assessment. HTI-1511 was more stable than a maleimide-based ADC control. Dose=8 mg/kg for each ADC. n=3 for HTI-1511, n=2 for HALO mAb Maleimide MMAE. ThioBridge Chemistry ThioBridge HTI-1511 DAR 4 Peak 2.5 Toxin on ADC Detection 8 9 13.6 min (98.2 %) 15.0 10.0 12.5 15.0 17.5 20.0 22.5 KRASMut KRASMut BRAF BRAF BRAF BRAF MEK MEK MEK MEK ERK/MAPK ERK/MAPK ERK/MAPK ERK/MAPK Internalized ADC BRAF Migration Angiogenesis Survival 1000 last dose 500 0 20 40 60 10 mg/kg 2500 V e h ic le H T I-1 5 1 1 2000 1500 1000 500 last dose 0 0 80 20 40 60 S tu d y D a y S tu d y D a y F c g R IIb F c g R IIa F c g R IIIa 1 5 8 V 10000 50000 H A LO m A b N aked 40000 H A L O m A b P T 2 -M M A E 20000 H A LO m A b N aked H A LO m A b N aked H A L O m A b P T 2 -M M A E 30000 6000 20000 4000 10000 2000 0 2 3 4 1 L O G ( u g /m L ) 40000 10000 0 0 2 3 -1 4 0 L O G ( u g /m L ) F c g R IIIa 1 5 8 F 15 mg/kg H A L O m A b P T 2 -M M A E 8000 H A LO m A b N aked H A L O m A b P T 2 -M M A E HA L O 30000 m Ab N aked 3 4 H A LO m A b N aked H A L O m A b P T 2 -M M A E 40000 2 FcgR I FcRn 50000 1 L O G ( u g /m L ) H A L O m A b P T 2 -M M A E 20000 Vehicle V e h ic le 2500 Human TNBC KRASG13D MDA-MB-231M HALO mAb H ALO m A b v cvcPAB-MMAE P A B -M M A E 2000 V e h ic le Vehicle 2500 HTI-1511 H A L O m A b P T 2 -M M A E 2500 Vehicle V e h ic le 2000 H A L OmAb m A b vcPAB-MMAE v c P A B -M M A E HALO HTI-1511 H A L O m A b P T 2 -M M A E H ALO m A b v cvcPAB-MMAE P A B -M M A E HALO mAb 2000 HALO m Ab HTI-1511 P T 2 -M M A E 1500 1500 1000 1000 1000 500 500 500 0 0 0 20 40 60 80 0 20 40 S tu d y D a y 60 20000 80 100 30000 20000 10000 10000 10000 0 0 1500 Fig. 11. KRAS-mutated PDx tumor model studies. HTI-1511 was tested in EGFR+, KRAS mutated, NSCLC or cholangiocarcinoma models (Champions Oncology). Dosing was 2.5 mg/kg weekly, N=8 mice per group. Tumor regressions were observed for all mice. No impact on body weight was observed. Attenuated Binding of HTI-1511 to FcRg Subtypes 30000 0 1 2 3 4 0 -2 0 2 4 L O G ( u g /m L ) L O G ( u g /m L ) -3 -2 -1 0 1 L O G ( u g /m L ) 2 Fig. 12. HTI-1511 exhibited attenuated binding to several FcRg subtypes when compared to the parental mAb. Binding was assessed by FACS. HTI-1511 demonstrated significantly attenuated binding to FcγRIIa, FcγIIb, FcγIIIa 158V, and FcγIIIa 158F receptors, but not attenuated binding to FcγR1, suggesting that HTI-1511 might have improved tolerability due to lack of binding by FcγRII-III receptors, possibly due steric hindrance from the PEG side chain. HTI-1511 PROGRAM SUMMARY • A mAb was engineered with attenuated binding to human skin, compared to cetuximab 0 20 40 S tu d y D a y 60 80 100 • An ADC mechanism of action that delivers a cytotoxic payload can treat KRAS- or BRAF-mutated tumors in mice S tu d y D a y • HTI-1511 utilizes next generation, ThioBridge chemistry V e h ic le Vehicle Human CRC BRAFV600E HT29 Vehicle V e h ic le 3000 H ALO m A b v cvcPAB-MMAE P A B -M M A E HALO mAb H A L O m A b P T 2 -M M A E HTI-1511 2000 2000 1000 1000 Vehicle V e h ic le 3000 H ALO m A b v cvcPAB-MMAE P A B -M M A E HALO mAb HALO H A L O mAb m A b vvcPAB-MMAE c P A B -M M A E HALO m Ab HTI-1511 HALO m Ab HTI-1511 P T 2 -M M A E 2000 P T 2 -M M A E • More homogeneous, active and stable (in primates) than maleimide based conjugation, in preclinical studies • Safety profile in a pilot primate toxicology study met criteria for candidate nomination • Tumor regressions observed in PDx tumor models that resulted in no remaining measurable tumors in mice 1000 • IND enabling studies underway 0 0 10 20 30 S tu d y D a y Fig. 5 Can the cytotoxic mechanism of action of an anti-EGFR ADC overcome downstream, activating mutations that are resistant to naked antibody therapy? 1500 0 REFERENCES 0 Released Cytotoxins Tumor Cell Death H T I-1 5 1 1 2000 1 0 Proliferation V e h ic le -1 0 0 0 0 5 mg/kg Cholangiocarcinoma (EGFR+,KRASpG12A) PDx 2500 0 ADC KRAS ADC Detection mAb Detection Fig. 7 Testing alternative conjugation chemistry. ThioBridge conjugation Technology10 leads to a consistent drug:antibody ratio (DAR) of 4, whereas first generation chemistries that are based on random lysine or cysteine conjugation lead to more heterogeneous mixtures.9 # drugs / Ab Analytical Hydrophobic Interaction Chromatography (HIC) mAU ER: 40% ThioBridge Cysteine Conjugation9 Cysteine Conjugation 3000 No signaling • More Homogeneous # drugs / Ab 8 9 4 29 Dose Ligand Proliferation 3 22 1000 0 T im e ( D a y s ) 0 KRAS Migration Angiogenesis Survival T u m o r V o lu m e (m m ) ± S E M 3 10 2 15 3 T u m o r V o lu m e (m m ) S E M 0 ADC = antibody-drug conjugate Pilot Primate Toxicology. Experiments were performed at a CRO under veterinary supervision and appropriate ACUC guidelines. Female animals were intravenously administered vehicle (phosphate buffered saline) or test article on Days 1 and 22. Animals were administered doses of 2.5 and 8.0 mg/kg (3 per group). Fc Receptor binding studies. CHO cells were engineered to express various Fc receptors. The unconjugated antibody or HTI1511 was incubated with each of these cell lines, then washed with PBS, labeled with R-PE-conjugated F(ab’)2 fragment of goat anti-human IgG (Jackson ImmunoResearch, Cat# 109-116-097, washed, then assessed by FACS. Naked anti-EGFR mAb inhibits signaling pathway 1500 1 8 0 1 M FI Day 3 Day 2 Fig. 4 In vivo imaging of tumor vs. skin binding. A human donor foreskin xenograft and human derived A431 tumor xenograft were paired and imaged over 7 days following DyLight755 labeled cetuximab and HALO mAb V1 administration. For HALO mAb V1, the ratio of binding between tumors and skin was higher than that of cetuximab (P<0.05). Rationale for Treatment of EGFR+ Mutation Resistant Tumors PDX Studies. Female Harlan nu/nu athymic nudes mice were implanted bilaterally with approximately 5x5x5 mm tumor fragments subcutaneously in the left and right flanks with Champions TumorGraft™ models CTG-0828 and CTG-0941. When tumors reached 1 to 1.5 cm3, they were harvested and viable tumor fragments approximately 5x5x5 mm were implanted subcutaneously in the left flank of female study mice. Primate Pharmacokinetics. An ELISA-based assay was used to measure antibody or ADC concentrations in cyno plasma samples drawn under study protocol from the toxicology study a CRO (antibodies used were goat-anti-human IgG polyclonal antibody from Bethyl Laboratories, Cat. No. A80-319A and mouse-anti-MMAE monoclonal antibody (Epitope Diagnostics, No. MAB30699). 0 # drugs / Ab HALO Anti-EGFR mAb EGFR-mediated signaling promotes cell growth 500 (S E M ) • 1000 1st Gen Chemistries: Heterogeneous METHODS Recombinant antibodies were expressed as full-length IgG1 in CHO cells and purified with Protein A affinity resin. HALO antiEGFR mAb Variant 1 (V1) corresponds to an early antibody lead. The anti-EGFR mAb used in HTI-1511 conjugate is a further engineered, humanized, candidate mAb. In vitro TME-specificity ELISA. An EGFR-coated plate was subjected to increasing concentrations of either HALO mAb or cetuximab in 25% human serum and 16.7 mM lactic acid for 60 minutes at either pH 6.0, pH 6.5, or pH 7.4 and washed with PBS. A secondary antibody-HRP conjugate was then added, washed and detected at OD450. For in vivo antibody binding studies, human foreskin skin grafts were implanted into Ncr nu/nu mice. Antibodies were labeled with the near-IR fluoroprobe DyLight755NHS ester dye, and administered IV at 10 mg/mouse at ~4 weeks post skin graft, to A431 tumor-bearing mice 3 weeks post tumor inoculation. Binding intensity was monitored by a Caliper IVIS system. 1500 Heterogeneous Fig. 1 Altered physicochemical properties of the solid tumor microenvironment. • Week Day H A L O V a r ia n t 1 - M M A E ( 3 0 m g /k g ) Assessing a Next Generation ADC Technology Cetuximab Control 1mm H A L O V a r a n t 1 - M M A E ( 3 0 m g /k g ) H A L O V a r ia n t 1 - M M A E ( 1 0 m g /k g ) Clinical observations and food consumption Body weight Dermal scoring Clinical pathology ECG and blood pressure Veterinary physical examinations and ophthalmology Histology Pharmacokinetics M FI H+ H A L O V a r ia n t 1 - M M A E ( 1 0 m g /k g ) H A L O V a r ia n t 1 - M M A E ( 3 m g /k g ) 6/6 Regressions, no evidence of tumors 0 Day 1 H A L O V a r ia n t 1 - M M A E ( 3 m g /k g ) H A L O V a r ia n t 1 - M M A E ( 1 m g /k g ) 2000 M FI O2 Opportunities for TME Specific Protein Design ↓ pH vs. healthy tissue (acidic pH) ↑ Lactic acid ↑ Albumin pH 7.4 2B. Fig. 3 Human skin graft model. Immunohistochemical analysis demonstrated EGFR expression in a normal foreskin xenograft. Human EGFR is visible in keratinocytes of both the superficial and basal layer in the xenograft (right arrow). Mouse skin is negative for human EGFR (left arrow). No human EGFR staining was observed in adjacent mouse tissue. The grafts were stable in mice for >3 months. 40X magnification is shown and 20X magnification is shown in the inlet. Staining was performed using a commercially available anti-human EGFR antibody. Proliferation (iododeoxyuridine) Hypoxia (pimonidazole) pH 6.5 H A L O V a r ia n t 1 - M M A E ( 1 m g /k g ) 2000 M FI pH 6.0 C e t u x im a b ( 3 0 m g /k g ) • • • • • • • • Endpoint T u m o r V o lu m e ( m m ) S E M 0 V e h ic l e C e t u x im a b ( 3 0 m g /k g ) 3 Blood vessel (immunofluorescent) 10 Cycle 1 2500 V e h ic l e T u m o r V o lu m e ( m m ) S E M Solid Tumor Architechtecture8 20 Parameters M FI KRAS mutations present in over 50% of mCRC, predictive of lack of benefit from anti-EGFR4 BRAF mutation in ~10% of mCRC5, debate whether anti-EGFR benefit EGFR mutation present in about 3% to 19% of NSCLC in Western world, predictive of PFS benefit from anti-EGFR TKI6-7 30 1 Cycle, 2 Dose Study Design, N=3 animals per group (3 each of Vehicle, 2.5 mg/kg, 8 mg/kg groups) M FI • • • HALO mAb Cetuximab Pilot Toxicology in Non-human Primates Human CRC Tumor Xenografts HT29 (BRAFV600E) 2500 (S E M ) • Downstream, Activating Mutations Skin pH Acidic pH, TME conditions 40 Fig. 2 Differential in vitro EGFR binding of Halozyme lead anti-EGFR antibody, HALO mAb. High affinity was observed for both HALO mAb and cetuximab at pH 6.0 in high lactate (16.7 mM) and 25% human serum buffer, a condition that we approximated as mimicking the tumor microenvironment in vitro. The difference in EGFR binding between the 2 antibodies was negligible. However, significantly attenuated EGFR binding was observed for HALO mAb at pH 7.4 in low lactate (1.0 mM) and 25% human serum, when compared to cetuximab. 3 • limits dosing and discourages patients • ~90% cutaneous side effects1-3 • 8% to 20% severe1-3 50 T u m o r V o lu m e , m m • EGFR expression leads to serious skin rash Human TNBC Tumor Xenografts MDA-MB-231M (KRASG13D) 3 Two Limitations with Anti-EGFR Therapeutics ADC Targeting of KRAS or BRAF Mutant EGFR+ Tumors T u m o r V o lu m e , m m INTRODUCTION Engineering a TME-Specific Anti-EGFR mAb EGFR Binding (EC50 ng/mL) Cancers with downstream activating KRAS or BRAF mutations in the EGFR pathway are resistant to EGFR targeting agents such as cetuximab and correspond to a significant unmet need. We hypothesized that an anti-EGFR ADC could be effective against KRAS or BRAF mutated tumors due to the cytotoxic mechanism of the ADC warhead. In an effort to eliminate the known dermal toxicity associated with anti-EGFR therapy, and to mitigate potential toxicities associated with treatment by an anti-EGFR ADC, a mAb was engineered with increased tumor microenvironment (TME) specificity for EGFR. The lead mAb demonstrated undetectable in vivo binding to human donor foreskins grafted onto nude mice, while binding to human A431 tumor xenografts with similar intensity to cetuximab (P < 0.005, detected using DyLight-755 conjugated versions of each mAb, measured with a Caliper IVIS system). The lead mAb was further optimized and conjugated to the potent cytotoxic drug MMAE using a novel bis-alkylating conjugation linker, which covalently re-bridged the inter-chain disulfide bonds, creating a stable and defined ADC. The resulting ADC, HTI1511, incorporated a vc-PAB cleavable moiety and a short linear PEG (24 ethylene glycol units) in a side-chain configuration. Analytical HIC revealed that HTI-1511 possessed a nearly homogenous drug:antibody ratio (DAR) of 4 (>99.7%). Approximately 70% of this compound was rapidly internalized by human tumor cells grown in vitro over 4 hours, overlapping the internalization kinetics of the unconjugated mAb. HTI-1511 was evaluated for efficacy against two human EGFR overexpressing tumor models, MDA-MB-231M (triple-negative breast cancer, KRAS-G13D) and HT-29 (colorectal cancer, BRAF-V600E), and dosed at 5, 10, and 15 mg/kg, (qw, IV). A clear dose dependent anti-tumor response was observed with complete tumor regressions observed at the 15 mg/kg dose in both models, which were resistant to treatment by cetuximab. In addition, HTI-1511 was well-tolerated at 2 and 8 mg/kg in a cynomolgus monkey toxicity study (n=3 per group), with limited dermal findings that were comparable with the vehicle control group. No adverse findings were observed at either dose. HTI-1511 showed a high degree of circulating stability in cynomolgus monkeys, and lacked in vivo degradation and instability that was observed in a control ADC conjugated using maleimide chemistry. HTI-1511 demonstrated significantly attenuated binding to FcγRIIa, FcγIIb, FcγIIIa 158V, and FcγIIIa 158F receptors, but not attenuated binding to FcγR1, in a FACS based assay format specific for each receptor, suggesting that HTI-1511 might have improved tolerability due to lack of binding by FcγRII-III receptors, possibly due steric hindrance from the PEG side chain. Thus, HTI-1511 holds promise as a potentially safe and effective treatment of EGFR overexpressing tumors with KRAS or BRAF mutations. 40 50 0 20 40 S tu d y D a y 60 80 0 10 20 30 40 50 60 70 80 90 100 110 S tu d y D a y Fig. 9 Comparison of potency between HTI-1511 and HALO anti-EGFR mAb conjugated with 1st generation chemistry (maleimide vc-PAB-MMAE). Each ADC was dosed weekly at 5,10, and 15 mg/kg, weekly. HTI-1511 showed superior potency across all groups. n=6 mice per group. Arrows indicate doses. 1) N Engl J Med. 2004 Jul 22;351(4):337-45. 2) J Clin Oncol. 2012 Oct 1;30(28):3499-506. 3) J Clin Oncol. 2008 Apr 1;26(10):1626-34. 4) Cancer Discov. 2014 Nov;4(11):1269-80. 5) Biomark Cancer. 2015 Sep 6;7(Suppl 1):9-126. 6) Ann Oncol. 2013 Sep;24(9):2371-6 7) J Natl Cancer Inst. 2013 May 1;105(9):595-605. 8) Lancet Oncology, 2010; 11: 661–69. 9) Thanos CD, Springer, 2016, in press. 10) Bioconjugate Chemistry 2014 Jun 18;25(6):1124-36.