ARTICLE IN PRESS
Progress in Retinal and Eye Research 26 (2007) 263–301
www.elsevier.com/locate/prer
Ion channels in the RPE
Sönke Wimmers, Mike O. Karl, Olaf Strauss
Experimentelle Ophthalmologie, Klinik und Poliklinik für Augenheilkunde, Universitätsklinikum Hamburg-Eppendorf,
MartinistraX e 52, 20246, Hamburg, Germany
Abstract
In close interaction with photoreceptors, the retinal pigment epithelium (RPE) plays an essential role for visual function. The analysis
of RPE functions, specifically ion channel functions, provides a basis to understand many degenerative diseases of the retina. The
invention of the patch-clamp technique significantly improved the knowledge of ion channel structure and function, which enabled a
new understanding of cell physiology and patho-physiology of many diseases. In this review, ion channels identified in the RPE will
be described in terms of their specific functional role in RPE physiology. The RPE expresses voltage- and ligand-gated K+, Cl, and
Ca2+-conducting channels. K+ and Cl channels are involved in transepithelial ion transport and volume regulation. Voltage-dependent
Ca2+ channels act as regulators of secretory activity, and ligand-gated cation channels contribute to RPE function by providing driving
forces for ion transport or by influencing intracellular Ca2+ homoeostasis. Collectively, activity of these ion channels determines the
physiology of the RPE and its interaction with photoreceptors. Furthermore, changes in ion channel function, such as mutations in ion
channel genes or a changed regulation of ion channel activity, have been shown to lead to degenerative diseases of the retina. Increasing
knowledge about the properties of RPE ion channels has not only provided a new understanding of RPE function but has also provided
greater understanding of RPE function in health and disease.
r 2007 Elsevier Ltd. All rights reserved.
Contents
1.
2.
3.
4.
Introduction . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
The function of the RPE . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
K+ . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
3.1. K+ channels and cell function . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
3.2. K+ ions in RPE function . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
3.3. K+ channels of the RPE . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
3.3.1. Inward rectifier K+ channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
3.3.2. Voltage-gated K+ channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
3.3.3. Ca2+-activated K+ channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
3.3.4. Two-pore K+ channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
3.4. Summary of K+ channel function . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Chloride . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.1. The role of Cl and Cl channels in cell function . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.2. Cl ions in RPE function . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.2.1. Fluid transport . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.2.2. Volume control . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.2.3. pH regulation. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.2.4. Intracellular organelles . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Corresponding author. Tel.: +49 40 42803 9469; fax: +49 40 42803 5017.
E-mail address: o.strauss@uke.uni-hamburg.de (O. Strauss).
1350-9462/$ - see front matter r 2007 Elsevier Ltd. All rights reserved.
doi:10.1016/j.preteyeres.2006.12.002
264
264
266
266
266
267
267
269
270
271
272
273
273
273
273
274
274
275
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
264
Identification and properties of Cl channels in the RPE . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.3.1. ClC Cl channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.3.2. CFTR . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.3.3. Ca2+-activated Cl channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.3.4. Volume regulated anion channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.3.5. Other Cl channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
2+
Ca
....................................................................................
5.1. Ca2+ and cell function in general . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
5.2. Ca2+ homeostasis in the RPE . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
5.3. Ca2+ and RPE function . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
5.4. Ca2+ channels of the RPE. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
5.4.1. Voltage-dependent Ca2+ channels . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
5.4.2. TRP channels. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
5.4.3. ATP receptors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
5.4.4. Glutamate receptors . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Na+ . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
6.1. Na+ ions in RPE cell function . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
6.2. Na+ channels of the RPE . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Ion channels and RPE disease . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
7.1. Change or loss of ion channel function leading to RPE diseases . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
7.2. Involvement of ion channels in signalling cascades leading to degenerative processes in the retina . . . . . . . . . . . . . .
Summary . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
Acknowledgements . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
References . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . .
4.3.
5.
6.
7.
8.
275
275
277
277
278
279
279
279
280
281
281
282
284
285
286
286
286
287
287
287
288
289
289
289
1. Introduction
2. The function of the RPE
With the invention of the patch-clamp technique,
understanding of the structure and function of ion channels
substantially improved and changed (Ackerman and
Clapham, 1997; Colquhoun, 1991; Hamill et al., 1981;
Jurkat-Rott and Lehmann-Horn, 2004; Neher and
Sakmann, 1976, 1992; Sakmann and Neher, 1984; Sigworth, 1986). General ion channel characteristics were
described in detail and the understanding of their cellspecific behaviour in different tissues improved. This
knowledge shed new light on many different cell functions
and also opened new doors toward an understanding of
patho-physiologic changes in cell behaviour. The latter
studies coined the term ‘‘channelopathies’’ for ion channelrelated diseases (Ackerman and Clapham, 1997; LehmannHorn and Jurkat-Rott, 1999).
The retinal pigment epithelium (RPE) fulfills many
tasks, which are essential for visual function (Bok, 1993;
Steinberg, 1985; Strauss, 2005). The characterization
of ion channels in the RPE is, therefore, a necessity to
understand these important functions in retinal health
and disease. The first patch-clamp study using cells of
RPE was published in 1988 by Fox et al. (1988). Since
then, the investigation of RPE ion channels has been
undertaken by several different research groups who
have now described a large number of different ion
channels in the RPE. In this review we have endeavoured
to summarize existing knowledge about ion channels
of the RPE in relation to RPE function and its pathophysiology.
The RPE is a monolayer of pigmented cells covering the
inner wall of the eye bulb (Bok, 1993). The apical
membrane of the RPE faces the light-sensitive outersegments of photoreceptors and the basolateral membrane
of the RPE is enface with the fenestrated capillaries of the
choroid (Bok, 1993; Boulton and Dayhaw-Barker, 2001;
Marmorstein, 2001; Marmorstein et al., 1998). The
interaction with the adjacent tissues relies on extracellular
matrices on both sides of the RPE. For a close interaction
with photoreceptors, the RPE has long apical microvilli
that surround the outer-segments of photoreceptors
(Boulton, 1991). The space in-between the RPE and
photoreceptors is filled with the interphotoreceptor matrix
(IPM). The IPM is essential for the interaction between
RPE and photoreceptors (Acharya et al., 1998; GonzalesFernandez, 2003; Hageman and Johnson, 1991; Hollyfield,
1999; Pepperberg et al., 1993; Uehara et al., 1990). In the
differentiated eye, the IPM facilitates the interaction
between RPE and photoreceptors enabling the exchange
of nutrients, signalling molecules, and metabolic end
products (Acharya et al., 1998; Gonzales-Fernandez,
2003; Hageman and Johnson, 1991; Hollyfield, 1999;
Pepperberg et al., 1993; Uehara et al., 1990). At the
basolateral side, the RPE is separated from the choriocapillaris by Bruch’s membrane, a multilayered extracellular
matrix structure (Garron, 1963; Guymer et al., 1999;
Lerche, 1963; Marshall et al., 1998; Sumita, 1961). Bruch’s
membrane represents an interface for exchange of nutrients
and signalling molecules, in this case between RPE and the
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
265
Fig. 1. Summary of RPE function to support vision. The figure is from Strauss (2005). MV ¼ apical microvilli; OS ¼ photoreceptor outer segments;
PEDF ¼ pigment epithelium-derived factor; VEGF ¼ vascular endothelial growth factor.
blood stream and between the RPE and the choroidal
endothelial cells (Fig. 1).
Electrophysiological studies have characterized the RPE
as a tight epithelium with a paracellular resistance 10 times
higher than the transcellular resistance (Miller and Steinberg, 1977a, b). This is achieved by a specific subset of
tight-junction proteins and defines the RPE as a part of the
blood/retina barrier (Ban and Rizzolo, 2000a, b; Kniesel
and Wolburg, 1993; Kojima et al., 2002; Konari et al.,
1995; Nishiyama et al., 2002; Williams and Rizzolo, 1997).
With its location and characteristics, the RPE is able to
fulfil a multitude of functions that are essential for visual
function (Bok, 1993; Steinberg, 1985; Strauss, 2005).
With its pigmentation, the RPE absorbs light energy,
which is focussed onto the macula by the lens (Boulton,
1991, 1998; Boulton and Dayhaw-Barker, 2001). As part of
the blood/retina barrier, it transports nutrients such as
glucose or vitamin A from the blood to the photoreceptors
(Baehr et al., 2003; Ban and Rizzolo, 2000b; Marmorstein,
2001; Sugasawa et al., 1994). On the other hand, the RPE
eliminates water from the subretinal space. Water accumulates in the subretinal space from metabolic activity of
photoreceptors and from the vitreous humour driven by
intraocular pressure (Hamann, 2002; Marmor, 1990;
Moseley et al., 1984). The transport of water is mainly
driven by a transepithelial transport of Cl from the
subretinal space to the blood (Bialek et al., 1996; Blaug
et al., 2003; Edelman et al., 1994a, b; Edelman and Miller,
1991; Hamann, 2002; Hughes et al., 1984; Maminishkis
et al., 2002; Miller et al., 1982; Peterson et al., 1997; Rymer
et al., 2001; Stamer et al., 2003; Steinberg, 1985).
Furthermore, the RPE is essential for establishing and
maintaining the immune privilege of the eye. This is not
only established by its barrier function, but also by
interfering with the signalling pathways which coordinate
the immune system and has immune suppressive function
in the healthy eye (Ishida et al., 2003; Streilein et al., 2002;
Wenkel and Streilein, 2000). The RPE not only controls the
amount of fluid in the subretinal space, it helps to establish
a constant ion composition in the subretinal space by
compensating for light-induced changes (Dornonville de la
Cour, 1993; Gallemore et al., 1997; Hughes et al., 1998;
Steinberg, 1985; Steinberg et al., 1983; Steinberg and
Miller, 1973). For example, illumination of photoreceptors
results in a decrease of the K+ concentration in the
subretinal space from 5 to 2 mM (Dornonville de la Cour,
1993; Gallemore et al., 1997; Miller and Steinberg, 1977b;
Steinberg et al., 1983). To maintain constant excitability
of photoreceptors this change in the K+ concentration
is compensated for by the RPE, which releases K+ into the
subretinal space (Dornonville de la Cour, 1993; Gallemore
et al., 1997; Miller and Steinberg, 1977b; Steinberg et al.,
1983).
The transport and metabolism of vitamin A is coupled to
another function essential for visual function, a metabolic
pathway named the visual cycle of retinal (Baehr et al.,
2003). The process of vision starts with absorption of light
energy by 11-cis retinal in the pigment of rhodopsin (Baehr
et al., 2003; Baylor, 1996; Bok, 1993). The light absorption
leads to the isomeric change of retinal from the 11-cis to the
all-trans isomer. Photoreceptors are unable to re-isomerise
all-trans retinal into 11-cis retinal. For re-isomerisation,
retinal is transported to the RPE where it is re-isomerised
into the 11-cis isomer (Arshavsky, 2002; Mata et al., 2002;
Thompson and Gal, 2003). After re-delivery to the
photoreceptors it serves to regenerate rhodopsin.
Light exposure of photoreceptors is accompanied by
photo-oxidative damage of proteins and phospholipids of
the outer segments (Beatty et al., 2000). To maintain
the excitability of photoreceptors, the outer segments have
to be regenerated (Bok, 1993; Bok and Hall, 1971;
Finnemann, 2003; LaVail, 1976; Steinberg, 1985). In this
regeneration process, new photoreceptor outer-segments
are constantly assembled from the connecting cilium. The
ARTICLE IN PRESS
266
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
tips of the outer segments are constantly shed from the
photoreceptors and phagocytized by the RPE. Proper
function of photoreceptors depends on the maintenance of
the right size of outer segments. This is achieved by the
coordinated shedding and phagocytosis of the tips and
requires a tight regulation in communication between RPE
and photoreceptors. The regulation takes place in a
circadian manner with a peak after onset of light (LaVail,
1976). In humans, the photoreceptor outer segments are
completely renewed every 11 days.
Transport, phagocytosis, and immune suppression require communication with adjacent tissues. For this
purpose, the RPE expresses a large variety of receptors
and is also able to secrete a variety of factors (Campochiaro, 1993; Strauss, 2005). The secretory function of the
RPE tissue enables not only a functional coordination with
photoreceptors and endothelium, but also immune suppression and stabilization of the functional integrity of
photoreceptors and endothelial cells. For the latter
purpose, the RPE secretes growth factors, such as the
pigment epithelium derived factor (PEDF) to the apical or
the photoreceptor side (Becerra et al., 2004; King and
Suzuma, 2000; Steele et al., 1992) and the vascular
endothelial growth factor (VEGF) to the basolateral or
endothelial side (Blaauwgeers et al., 1999; Slomiany and
Rosenzweig, 2004a; Witmer et al., 2003).
These collective functions of the RPE are tightly linked
to alterations in ion channel activity and underscore the
importance of the study of ion channels in the RPE in
order to shed new light on the cellular mechanisms by
which the RPE supports visual function.
3. K+
3.1. K+ channels and cell function
The largest group of ion channels in the human genome
represents the family of K+ channels consisting of at least
78 pore-forming a subunits (Yu et al., 2005). These include
the 15 members of the inwardly rectifying K+ channel
subfamily with two transmembrane domains, 40 mainly
outwardly rectifying voltage-gated K+ channels with six
transmembrane domains, 9 Ca2+-activated K+ channels
with six or seven transmembrane domains, and 15 twopore or leak K+ channels with four transmembrane
domains and two pores.
The term rectification refers to the characteristic
conductance changes of ion channels upon membrane
potential changes. The inwardly rectifying ion channels
have larger ion conductivity in response to hyperpolarizing
membrane potential changes than in response to depolarization. Nevertheless, the current mediated by inwardly
rectifying K+ channels is predominantly a K+ efflux since
the direction of K+ flux is controlled by the reversal
potential or equilibrium potential for this ion, which is
typically around 80 mV. Accordingly, at potentials
positive to 80 mV, K+ ions flow out of the cell and at
potentials negative to 80 mV, K+ flows into the cell. The
outwardly rectifying channels have a larger ion conductance in response to depolarization of the membrane
potential. The subsequent outflow of K+ ions repolarizes
the membrane potential back towards the K+ equilibrium
potential. All K+ channels are highly selective for K+ ions.
Why this diversity in K+ channels? K+ as a charge
carrier and an osmotically active molecule is involved in
many cell functions. One task of K+ channels in excitable
cells is to control the resting membrane potential of the
cells shifting it towards the reversal potential of K+.
Additionally, K+ channels are involved in the shaping/
duration of action potentials and in adaptive mechanisms
by repetitive excitation of neurons (Hille, 2001).
In non-excitable cells, K+ channels are responsible for
the maintenance of the hyperpolarized resting membrane
potential. In addition to this, these channels play essential
roles in (1) transepithelial transport processes, (2) the
control of cell volume and intracellular pH, (3) the
recycling and secretion of K+, (4) the cell cycle and
apoptosis, and (5) in the regulation of hormone and growth
factor secretion (Ashcroft and Gribble, 1999; Bauer et al.,
1999; MacDonald and Wheeler, 2003; Masi et al., 2005;
Warth, 2003).
3.2. K+ ions in RPE function
As stated above, (Section 2) one function of the RPE is
to control the ion composition in the subretinal space. The
K+ concentration of the subretinal space in the absence of
light is approximately 5 mM. Under these conditions K+
enters the RPE cells through the apically located Na+/K+
ATPase and leaves the cells via the basolateral membrane,
leading to a net absorption of K+ (la Cour et al., 1986;
Miller and Steinberg, 1982). When exposed to light,
photoreceptors hyperpolarize due to the closure of cyclic
nucleotide-gated Na+-conducting cation channels, which
are located in the membrane of the light sensitive outer
segments (Liebman et al., 1987). In the darkness, the K+
ions entering the photoreceptors and the RPE cells through
their Na+/K+-ATPases and the K+ ions leaving the
photoreceptors as counterion to Na+ are in equilibrium
(Baylor, 1996; Strauss, 2005). Due to the hyperpolarization
induced by light exposure, less K+ ions leave the
photoreceptors. Consequently, the K+ concentration in
the subretinal space decreases in the light from 5 to 2 mM
(Oakley and Steinberg, 1982; Steinberg et al., 1980).
Because of the large K+ conductivity of the apical
membrane, the decrease of the extracellular K+ concentration leads to a hyperpolarization of the apical membrane of
the RPE (Griff and Steinberg, 1984; Miller and Steinberg,
1977b; Steinberg et al., 1970). Additionally, the low
extracellular K+ concentration inhibits the Na+/K+/
2Cl co-transporter in the apical membrane of the RPE
cells (Bialek et al., 1995; Joseph and Miller, 1991) resulting
in a decrease of the intracellular Cl concentration (Joseph
and Miller, 1991), and as a consequence to an extrusion of
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
cytosolic water and shrinkage of the RPE cells and
acidification of the cytosol (Adorante, 1995; Adorante
and Miller, 1990; Bialek and Miller, 1994; Huang and
Karwoski, 1992; Kennedy, 1994; Li et al., 1994a; Lin and
Miller, 1991). The extrusion of water together with cell
shrinkage leads to an increase of the subretinal space and
to a further decrease of the subretinal K+ concentration.
The large apical K+ conductance helps to restore the
normal subretinal K+ concentration.
Another function of K+ channels is to counteract Ca2+
influx. As Ca2+ influx leads to an increase of intracellular
free Ca2+ and to depolarization of the cell, and Ca2+ ions
are involved in many RPE functions (see Section 5) there
must be a counterbalance for Ca2+ flux in RPE cells. K+
channels are good candidates because they are activated by
depolarization (voltage-gated K+ channels) and by Ca2+
itself (Ca2+-activated K+ channels).
Additionally, they may support functions as described in
other epithelial cell types such as wound healing, regulation
of nutrient transport, and cell proliferation/differentiation
(Warth, 2003).
3.3. K+ channels of the RPE
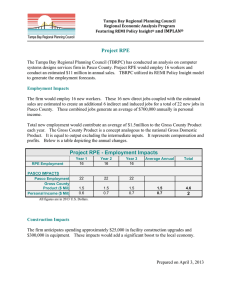
In Fig. 2, patch-clamp data from two representative RPE
cells are shown. Both cells have an inwardly rectifying current
but with very different kinetics. Cell 2 has an additional
outwardly rectifying current showing the heterogeneity of
K+ conductances in these cells. In the next chapters, the
different K+ conductances identified in RPE cells will be
introduced in relation to their particular K+ channel classes
and discussed in their possible roles for RPE function.
3.3.1. Inward rectifier K+ channels
3.3.1.1. Inward rectifier K+ (Kir) channels in general. Kir
channels are activated upon hyperpolarization. Though
K+
RPE
Cell 1
30 mV
K+
Cell 2
-60 mV
-150 mV
100 pA
10 ms
100 pA
10 ms
Fig. 2. Patch-clamp measurements of two mouse RPE cells exemplifying
the heterogeneity of membrane K+ currents in these cells. The cells were
held at 60 mV and stimulated with voltage-steps of 10 mV, first 10
depolarizing steps then 10 hyperpolarizing steps.
267
their name suggests that they maintain K+ inward current,
their main function in most cells is to bring the membrane
potential towards the K+ equilibrium potential. As in most
cells, in RPE cells the equilibrium potential is negative to
the resting membrane potential; thus Kir channels mainly
conduct outward K+ currents. Kir channels are composed
of four pore-forming subunits with two transmembrane
domains, a pore loop and cytoplasmic N- and C-termini
(Bichet et al., 2003). Their inward rectification is caused by
a block of the channel pore by internal Mg2+ and by
cytoplasmic polyamines such as spermine or spermidine at
positive voltages (Lu, 2004; Nichols and Lopatin, 1997).
Based on their biophysical properties and their sensitivity
to intracellular signals the Kir channels are subdivided into
seven subfamilies (Kubo et al., 2005). The Kir1 subfamily
consists of only one member with weak inwardly rectifying
properties (Ho et al., 1993). The four Kir2 subfamily
members are strong inward rectifiers (Kubo et al., 1993a;
Morishige et al., 1994; Takahashi et al., 1994; Topert et al.,
1998). The members of the Kir3 or GIRK subfamily are
G-protein-coupled inward rectifiers being activated by the
Gbg subunits of G proteins (Dissmann et al., 1996; Kofuji
et al., 1995; Kubo et al., 1993b; Spauschus et al., 1996).
Members of the Kir4 and 5 subfamilies are able to form
heteromultimers with each other (Pearson et al., 1999;
Pessia et al., 1996). While Kir4 members are able to form
functional channels alone (Pessia et al., 1996), the Kir5
subunit must be co-expressed with Kir4 subunits (Bond et
al., 1994). Both homomeric Kir4 channels and heteromeric
Kir4/5 channels are weakly inwardly rectifying (Pessia
et al., 1996). The Kir6 subfamily members co-assemble
with sulfonylurea receptor SUR1 and 2 to form K+
channels that are sensitive to the intracellular ATP/ADP
ratio (Aguilar-Bryan et al., 1995; Inagaki et al., 1995a, b,
1996; Sakura et al., 1995). Finally, the Kir7.1 subunit forms
a weak inward rectifier that has higher conductance with
decreasing extracellular K+ concentrations (Doring et al.,
1998; Krapivinsky et al., 1998). Though, most Kir channels
are blocked by extracellular Ba2+, the subfamilies can be
further characterized by their different sensitivity to Ba2+.
Kir7.1 has, for example, a very low Ba2+ sensitivity with
an IC50 of 1 mM (Doring et al., 1998; Krapivinsky et al.,
1998) while Kir4.1 is much more sensitive with an IC50 in
the micromolar range (Bond et al., 1994).
3.3.1.2. Inward rectifier K+ (Kir) channels in the RPE. Different subtypes of inwardly rectifying K+ channel subunits
have been identified in the RPE (Ettaiche et al., 2001;
Ishii et al., 1997; Kusaka et al., 1999, 2001; Shimura et al.,
2001; Yang et al., 2003). In the libraries of the National
Eye Institute only KCNJ13 (or Kir7.1) is listed in both
the human and the mouse library of expressed sequence
tags (EST; http://neibank.nei.nih.gov/). RT-PCR and
immunohistological investigations have identified the
additional expression of Kir4.1 (Kusaka et al., 1999)
and Kir6.2 together with its auxiliary subunit SUR1 in
the RPE of rats (Ettaiche et al., 2001). Further, several
ARTICLE IN PRESS
268
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
electrophysiological investigations using Ussing chambers
as well as the patch-clamp technique, have shown that RPE
cells contain K+ currents with inwardly rectifying properties (Gallemore et al., 1993; Hernandez et al., 1995; Hughes
et al., 1995a; Hughes and Steinberg, 1990; Hughes and
Takahira, 1996, 1998; Immel and Steinberg, 1986; Kusaka
et al., 1999, 2001; Poyer et al., 1996; Segawa and Hughes,
1994; Shimura et al., 2001; Strauss et al., 1993, 1994;
Takahira and Hughes, 1997; Tao et al., 1994; Wen et al.,
1993; Wollmann et al., 2006; Yuan et al., 2003). The Ussing
chamber investigations allowed only their indirect identification by the application of the unspecific blockers Ba2+
and Cs+, which also block other ion channels including
voltage-gated, Ca2+-activated, and two-pore K+ channels
(Goldstein et al., 2005; Grissmer et al., 1993; Ho et al.,
1999; Hurst et al., 1996). Nevertheless, it has been shown
by these studies that there is a large Ba2+-sensitive K+
conductance in both the apical and the basolateral
membrane (Gallemore et al., 1993; Hughes et al., 1995a).
Using the patch-clamp technique, single freshly isolated as
well as cultivated RPE cells of different species have been
studied. In most of these studies, inwardly rectifying
K+ currents were identified. The inwardly rectifying K+
currents appeared in all species investigated: frog, monkey,
human, rat, toad, rabbit, bovine, and mouse. In rat and
bovine RPE cells, combined patch-clamp and molecular
biological studies revealed that these inwardly rectifying
currents are carried either by Kir7.1 channels alone or by
Kir7.1 and Kir4.1 channels (Kusaka et al., 1999, 2001;
Shimura et al., 2001; Yang et al., 2003). Both of these
channels were shown to be located in the apical membrane
of the RPE cells by immunohistochemistry (Kusaka et al.,
1999, 2001; Yang et al., 2003). The currents measured in
these studies showed some features typical for Kir7.1
channels: (i) weak inward rectification, (ii) an increase in
the conductance with reduced K+ concentration in the
bath (all other inward rectifiers show a reverse K+
dependence) and (iii) a relative low Ba2+ sensitivity. While
some of the observations seem to fit very well with the
presence of Kir7.1 channels, some data suggest that there
might be different types of inward rectifiers expressed in the
RPE, even in the same species (Strauss et al., 1993; Wen et
al., 1993). In bovine RPE cells an inward rectifier has been
described that exhibits a pronounced rundown of current
when measured without intracellular ATP (Hughes and
Takahira, 1998). Although it has not been investigated in
detail, the Kir7.1 channels seem to be ATP-independent
while Kir4.2 is known to be enhanced by ATP (Yang et al.,
2000). This finding supports the study of Kusaka et al.
(1999) who showed by single channel recordings from the
apical side of RPE cells and by immunohistochemistry that
Kir4.1 channels are also localized in the apical membrane.
Additionally, in cultured RPE cells, currents have been
measured with a much more pronounced inward rectification than that of Kir7.1 and Kir4.1 channels which both
are weak inward rectifiers (Hughes and Takahira, 1996).
Thus, there might also be channels of the Kir2 or the Kir3
subfamilies expressed. Since comparison of inwardly
rectifying currents between freshly isolated and cultured
human or monkey RPE cells revealed some noticeable
differences in respect to inactivation, Ba2+-sensitivity, and
dependence on extracellular K+, the expression of inward
rectifiers with strong inward rectification may be an
expression of the potential of RPE cells to de- and transdifferentiate in culture (Zhao et al., 1997). Accordingly, for
the investigation of the in vivo situation, the use of freshly
isolated RPE cells should be favoured.
3.3.1.3. Role of inwardly rectifying K+ (Kir) channels for
RPE function. The RPE is responsible for the homoeostasis of K+ in the subretinal space (Dornonville de la
Cour, 1993; Strauss, 2005). Since K+ continuously enters
the RPE cells through the apical membrane via the
electrogenic Na+/K+-ATPase and a Na+-K+-2Cl cotransporter and since very little K+ is transcellularly
transported at least with low subretinal K+ concentrations
of 2 mM as occurs in the light (Dornonville de la Cour,
1993), the RPE needs a way to recycle the K+ at the apical
membrane, e.g. for the activity of the apical Na+/K+
ATPase (la Cour et al., 1986). Kir7.1 channels, perhaps
together with Kir4.1 channels, are expressed in the apical
membrane. Due to their weak inwardly rectifying properties, these channels are well suited for K+ recycling
through the apical membrane. In the dark, this K+
recycling through the inward rectifier and the Na+
recycling through the Na+/K+-ATPase on the apical
membrane supports Cl transport through the cell (Bialek
and Miller, 1994; La Cour, 1992). This Cl transport
across the cell supports fluid transport through the RPE in
the retina to choroid direction (DiMattio et al., 1983;
Frambach and Misfeldt, 1983; Miller and Edelman, 1990;
Tsuboi et al., 1986). Thus, the inward rectifier in the apical
membrane supports the absorption of water across the
RPE. The consequent reduction of the subretinal space
holds the retina in the proximity of the RPE, which is
necessary for the maintenance of retinal function and helps
to prevent retinal detachment. Upon illumination, the K+
concentration in the subretinal space decreases from 5 to
2 mM (Huang and Karwoski, 1992; Li et al., 1994a, b;
Oakley and Steinberg, 1982; Steinberg et al., 1980) and the
volume of the subretinal space increases. This volume
increase seems to be caused by an increased water efflux
from the RPE (Huang and Karwoski, 1992). The driving
force for this water efflux is believed to be a decreased
intracellular K+ concentration in RPE cells. Part of this
K+ decrease is likely driven by K+ efflux through inwardly
rectifying K+ channels. The atypical behaviour of Kir7.1
channels in their dependence to the extracellular K+
concentration, whereby at lower K+ concentrations they
possess a higher conductivity (Doring et al., 1998;
Krapivinsky et al., 1998), makes them good candidates to
generate the underlying increased K+ efflux.
Besides this volume regulation of the subretinal space,
there are some reports that a Ba2+-sensitive K+ channel is
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
involved in the regulation of the RPE cell volume itself
(Adorante, 1995; Kennedy, 1994). Since inwardly rectifying
K+ channels are blocked by the Ba2+ concentrations used
in these studies, the participation of inward rectifiers in the
regulatory volume decrease (RVD) induced by hypoosmotic stress seems very likely. Nevertheless, Ba2+ is a nonspecific blocker of voltage-gated and Ca2+-activated
K+ channels (Bello and Magleby, 1998; Grissmer et al.,
1993; Park et al., 2003). As the direct activation of
Ca2+-activated K+ channels has been demonstrated (Sheu
et al., 2004), the participation of inward rectifiers in volume
regulation remains to be proven.
In other cell types a participation of inwardly rectifying
channels on hormone secretion has been reported (e.g. in
pancreatic b cells the intracellular ATP/ADP ratio is
coupled to the activity of inwardly rectifying K+ channels).
The increase of this ratio leads to the closure of ATPsensitive inward rectifiers composed of the same subunits
found in the RPE, Kir6.2 and SUR1 (Ashcroft and
Gribble, 1999; Ettaiche et al., 2001). This closure leads to
depolarization of the cells, to Ca2+ entry through voltagegated Ca2+ channels, and to increased insulin secretion. As
the secretion of a variety of cytokines is controlled by
intracellular Ca2+, which enters the cell via voltage-gated
Ca2+ channels (see Section 5), a participation of these
ATP-sensitive inward rectifiers on the regulation of
secretion in RPE cells may not be excluded. Nevertheless,
functional data showing the physiological relevance of
these channels in the RPE are still missing.
3.3.2. Voltage-gated K+ channels
3.3.2.1. Voltage-gated K+ channels in general. Like inwardly rectifying K+ channels, voltage-gated K+ channels
are composed of four pore-forming a subunits (Yellen,
2002). The channels can be formed of four of the same
subunits forming homomeric channels or of different
subunits forming heteromeric channels (Jan and Jan,
1997). As the channels open upon membrane depolarization, most channels are outwardly rectifying (Gutman
et al., 2005). Homology sequence analysis was used to
subdivide the voltage-gated K+ channels into 11 subgroups with up to eight subfamily members. Though, the
members of the Kv5, 6, 8 and 9 subfamilies do not form
functional K+ channels when expressed alone in heterologous expression systems, they form part of the channel
pore when expressed together with members of different
subfamilies (Gutman et al., 2005). The different a subunits
are further characterized by different biophysical and
pharmacological properties. While some channels inactivate very fast at depolarizing voltages (so-called A
currents), others show only slow or no inactivation at all.
Additionally, the channels differ in their voltage-sensitivity,
and for most channels highly selective blockers have been
described.
In non-excitable cells the role of voltage-gated K+
channels is difficult to define because most of these cells
show only small membrane potential changes and some of
269
them stay in hyperpolarized states in which the expressed
channels should not be active. Nevertheless, voltage-gated
K+ channels in non-excitable cells have been shown to be
involved in the following functions: (i) transport of
nutrients and electrolytes, (ii) recycling and secretion
of K+, (iii) regulation of cell volume and pH, (iv) control
of cell cycle progression, and (v) regulation of growth
factor secretion (O’Grady and Lee, 2005; Warth, 2003).
3.3.2.2. Voltage-gated K+ channels in the RPE. According to the databases of EST of the National Eye Institute,
six different pore-forming subunits of voltage-gated K+
channels are expressed in the RPE/choroid (Kv1.2, 2.1, 7.1-3
and 8.2). Additionally, two ESTs of auxiliary subunits have
been identified in these preparations (Kvb2, KCNE4 ¼
MiRP3). By RT-PCR the expression of Kv1.2 and 2.1 has
been confirmed (Pinto and Klumpp, 1998). Further,
transcripts of Kv1.3, 1.4 and 4.2 have been identified by
RT-PCR. Western blot and immunohistochemcical data
show that Kv1.3 and 1.4 subunits are located in the apical
membrane of RPE cells (Pinto and Klumpp, 1998; Strauss
et al., 2002; Wollmann et al., 2006). As voltage-gated K+
channels are composed of four pore-forming subunits and
the different members of one subfamily are able to form
functional channels together, the Kv1.2, 1.3 and 1.4
subunits may form heteromeric channels. The Kv4.2
subunits were localized to the basolateral membrane (Pinto
and Klumpp, 1998). The expression of Kv7 subunits has
recently been confirmed in human and monkey RPE cells
(Hughes et al., 2006) in a combined molecular biological
and electrophysiological study. The data confirmed the
expression of Kv7.1 but instead of Kv7.2 and 7.3 they
found transcripts of Kv7.4 and 7.5. Patch-clamp experiments with different blockers suggest that these currents
are mainly mediated by Kv7.5 channels. Additionally, in
preparations from bovine RPE, K+ currents have been
measured that resemble the M current, which is known to
be carried by Kv7 channels (Takahira and Hughes, 1997).
In all species investigated, in addition to these M currents,
other voltage-gated outwardly rectifying K+ currents
have been characterized electrophysiologically (Fox and
Steinberg, 1992; Hughes and Steinberg, 1990; Hughes and
Takahira, 1996; Hughes et al., 1995b; Poyer et al., 1996;
Strauss et al., 1993, 1994, 2002; Takahira and Hughes,
1997; Tao et al., 1994; Wen et al., 1993; Wollmann et al.,
2006). But the currents display very heterogeneous
biophysical properties. Even in the same species outwardly
rectifying K+ currents with different properties were
described (Hughes and Takahira, 1996; Hughes et al.,
1995b; Strauss et al., 1993; Wen et al., 1993). For example,
in freshly isolated human RPE cells a non-inactivating
current with an activation threshold of 30 mV was
observed (Wen et al., 1993) while in another preparation
non-inactivating currents with a threshold of 60 mV were
found (Hughes et al., 1995b). Despite these differences all
identified currents have a relatively low sensitivity to
tetraethylammonium (TEA) in common—an unspecific
ARTICLE IN PRESS
270
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
blocker of voltage-gated K+ channels. Additionally, with
one exception, all currents exhibit slow or no inactivation.
These findings suggest that members of the Kv4.x
subfamily are not functionally expressed—though Pinto
and Klumpp found Kv4.2 in the basolateral membrane of
rat RPE cells (Pinto and Klumpp, 1998)—since these
channels display a very fast inactivation (Gutman et al.,
2005). Such a fast inactivation was only found in one
preparation of foetal human RPE cells (Wen et al., 1993)
and not in the rat cells which were studied by Pinto and
Klumpp. So far, only one attempt has been made to
identify the molecular basis of outwardly rectifying K+
currents in RPE cells (Strauss et al., 2002). The current
investigated in that study was inhibited by the K+ channel
blocker agitoxin-2, which is highly specific for Kv1.1, 1.3
and 1.6 channels (Garcia et al., 1994) while blockers
specific for other K+ channels had no effect on the current.
Furthermore, the expression of Kv1.3 was confirmed by
western blots. However, it cannot be excluded that these
Kv1.3 subunits form heteromeric channels with other
subunits of the same subfamily.
3.3.2.3. Role of voltage-gated K+ channels for RPE
function. Most voltage-gated K+ currents described in
RPE cells activate at potentials positive to 40 to 30 mV
(Fox and Steinberg, 1992; Hughes and Steinberg, 1990;
Strauss et al., 1993, 1994, 2002; Takahira and Hughes,
1997; Tao et al., 1994; Wen et al., 1993), some of them at
even more positive potentials (Hughes and Takahira, 1996;
Wollmann et al., 2006). As RPE cells have resting
membrane potentials between 50 and 40 mV with only
small changes to more positive potentials (Dornonville de
la Cour, 1993; Fujii et al., 1992; Gallemore et al., 1997;
Hughes and Steinberg, 1990; Joseph and Miller, 1991;
Miller and Steinberg, 1977b; Quinn and Miller, 1992;
Steinberg et al., 1978; Wen et al., 1993), it remains unclear
how these channels might contribute to RPE cell function.
One possible mechanism that may lead to their opening has
been shown by Strauss et al. (2002). This study demonstrated that the outwardly rectifying K+ channel investigated in rat RPE cells is completely abolished by the
inhibition of a tyrosine kinase and increased by the
addition of an activated pp60csrc tyrosine kinase. As this
activation might be attributed to a shift in the activation
threshold, this provides a way to activate voltage-gated
channels without changing the membrane potential. These
channels are probably composed of Kv1.3 subunits. Kv1.3
channels are widely expressed in cells of the immune system
(Cahalan et al., 2001; George Chandy et al., 2004). In
microglia Kv1.3 channels are necessary for their immune
reaction (Fordyce et al., 2005). As RPE cells are part of the
immune system in the posterior part of the eye (Streilein,
2003; Streilein et al., 2002), the delayed rectifier in the RPE
cells may be part of their immune response.
The M current identified in freshly isolated bovine RPE
cells activated at potentials positive to 80 mV. Accordingly, these channels may contribute to the maintenance of
the resting membrane potential. M currents, carried by
Kv7 channels, are inhibited by Gq/11 coupled membrane
receptors (Delmas and Brown, 2005). Gq proteins activate
phospholipase Cb which hydrolyze phosphatidylinositol4,5-bisphosphate (PIP2) into inositol-1,4,5-trisphosphate
(InsP3) and diacylglycerol (DAG). The breakdown of PIP2
leads to the closure of M channels. RPE cells have been
shown to express different Gq/11 protein coupled receptors:
epinephrine receptors a1B and a1D (Moroi-Fetters et al.,
1995), serotonin receptor 5HT2 (Nash et al., 1999; Osborne
et al., 1993) and purinergic receptors P2Y1 and P2Y2
(Maminishkis et al., 2002; Reigada et al., 2005; Sullivan
et al., 1997). Stimulation of purinergic and epinephrine
receptors leads to elevated intracellular free Ca2+ in RPE
cells (Maminishkis et al., 2002; Quinn et al., 2001). Further,
these stimulations lead to the depolarization of apical and
basolateral membranes. While depolarization of the
basolateral membrane may be attributed to the activation
of a Ca2+-activated Cl conductance, the apical depolarization may be due to the decrease of a K+ conductance.
As M currents are inhibited by the activation of these
receptors, they are good candidates for this purpose
presuming that they are expressed in the apical membrane
of RPE cells. Furthermore, stimulation of both purinergic
and epinephrine receptors is involved in reattachment of
the retina to the RPE by elevated fluid transport from the
subretinal space to the choroid (Maminishkis et al., 2002;
Rymer et al., 2001). When these M channels are expressed
in the apical membrane and Ca2+-activated K+ channels
in the basolateral membrane this would provide a
possibility for the regulation of directed K+ transport
through the RPE, as both channels are reciprocally
regulated by Gq/11-coupled receptors. One member of this
subfamily (Kv7.1) is involved in ion transport in various
tissues. This includes Cl secretion in airway (Leroy et al.,
2004) and gastrointestinal epithelia (Kunzelmann et al.,
2001a, b), K+ secretion in the stria vascularis (Marcus
et al., 1997, 1998; Shen and Marcus, 1998; Sunose et al.,
1997) and K+ recycling in gastric parietal cells (Warth and
Barhanin, 2003). Thus, in RPE, M channels may participate in the ion homoeostasis of the subretinal space.
3.3.3. Ca2+-activated K+ channels
3.3.3.1. Ca2+-activated K+ channels in general. Many
processes in the RPE are coupled to changes in the
intracellular free Ca2+ concentration (see Section 5). As
Ca2+-activated K+ channels are gated by changes in
intracellular free Ca2+, they are capable of fulfilling a
variety of cellular functions (Vergara et al., 1998; Weiger
et al., 2002). Based on single channel conductance,
sequence homologies and pharmacology, Ca2+-activated
K+ channels are subdivided into three subgroups (Wei
et al., 2005): (i) SK channels with a small K+ conductance
(4–14 pS), (ii) IK channels with an intermediate K+
conductance (20–80 pS), and (iii) BK or maxi K channels
with a large conductance (200–300 pS). While SK and IK
channels are gated solely by intracellular Ca2+, BK
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
channels are gated by both intracellular Ca2+ and voltage.
Like voltage-gated K+ channels they are formed of four
subunits with six transmembrane domains. The BK
channel has an additional N-terminal transmembrane
domain (S0) (Bond et al., 1999; Ghatta et al., 2006).
Ca2+-activated K+ channels are found in close vicinity
to voltage-gated Ca2+ channels in neurons (Gola and
Crest, 1993; Grunnet and Kaufmann, 2004; Marrion
and Tavalin, 1998) and in cochlear hair cells (Issa and
Hudspeth, 1994; Roberts et al., 1990) proposing a role as
feedback modulators for voltage-gated Ca2+ channels
that provide the Ca2+ needed for the activation of
Ca2+-activated K+ channels. In non-neuronal tissues
Ca2+-activated K+ channels were found to be involved
in volume regulation, and in transepithelial ion secretion or
absorption, and cell cycle regulation (Begenisich et al.,
2004; Feranchak et al., 2004; Huang et al., 2002; Joiner
et al., 2003; Liu et al., 2002; Turnheim et al., 2002).
3.3.3.2. Ca2+-activated K+ channels in the RPE. So far,
Ca2+-activated K+ channel subunits have not been
identified in the RPE, either by molecular biological or
by protein biochemical methods. However, patch-clamp
measurements with RPE cells from different species have
provided evidence that an outwardly rectifying K+ channel
is activated by elevated intracellular Ca2+ concentrations
(Ryan et al., 1999; Sheu and Wu, 2003; Sheu et al., 2004,
2005; Tao and Kelly, 1996). These currents were completely
blocked by the specific BK channel inhibitor iberiotoxin
and had a unitary conductance of 150 pS (Sheu and Wu,
2003; Tao and Kelly, 1996). As charybdotoxin, a blocker of
BK and IK channels, and apamin, a blocker of SK
channels, had no additional blocking effect on these
currents, it seems likely that the K+ channels activated by
raising intracellular Ca2+ are of the BK subtype (Sheu and
Wu, 2003). Since BK channels have not yet been studied by
either immunohistochemistry or electrophysiological measurements using intact RPE tissue, it is not known whether
these channels are in the apical or basolateral membrane.
3.3.3.3. Role of Ca2+-activated K+ channels for RPE
function. Different physiological stimuli have been used
to activate Ca2+-activated K+ channels in the RPE.
Application of ATP has been shown to increase intracellular Ca2+ concentrations in RPE cells through the
activation of different purinergic receptors (see Section 5).
This increase in intracellular free Ca2+ in turn stimulates
the apical to basolateral fluid transport through the RPE
(Peterson et al., 1997). The fluid transport is driven by
Ca2+-dependent Cl currents in the basolateral membrane
(see Section 4) and may be supported by Ca2+-activated
K+ channels. Ca2+-activated K+ channels were shown to
be involved in volume regulation in RPE cells in response
to hypotonic stress (Sheu et al., 2004). In that study, BK
currents were increased by hypoosmotic stress. The
induced K+ efflux together with a yet unidentified anion
efflux is thought to be responsible for the RVD (Kennedy,
271
1994). Furthermore, intracellular Ca2+ signalling regulates
a variety of RPE cell functions (e.g. photoreceptor
phagocytosis, growth factor secretion, immune responses,
differentiation). To control these processes the cells need
mechanisms to terminate the Ca2+ signals. One possibility
is simply to extrude the Ca2+ from the cytosol via different
membrane transporters and pumps (Kennedy and Mangini, 1996; Loeffler and Mangini, 1998; Mangini et al., 1997).
The second possibility is to terminate the signal by
stopping the Ca2+ influx. As in other cell types, the
participation of Ca2+-activated K+ channels in the latter
process has been proven (Ghatta et al., 2006) and it is
tempting to speculate that these channels may serve the
same purpose in the RPE. Furthermore, BK channels in
the RPE are inhibited by oxidizing agents (Sheu and Wu,
2003). Mutational analysis has revealed a protective
mechanism against oxidative stress that involves the
participation of heterologously expressed BK channels
(Santarelli et al., 2006). As RPE cells are exposed to high
oxidative stress (Mainster, 1987; Tanito et al., 2002; van
Best et al., 1997), the BK channels may provide part of the
oxidant protective mechanisms in these cells.
3.3.4. Two-pore K+ channels
The last group of K+ channels to mention is the family
of two-pore K+ channels. In contrast to other K+
channels, which are all formed by four subunits with one
pore domain each, two-pore K+ channels are formed by
two subunits with two pore domains each (Goldstein et al.,
2005; Lesage and Lazdunski, 2000). In vertebrates, 15
members of this K+ channel subclass have been identified
which are further divided into six subfamilies: the weakly
inwardly rectifying TWIK channels, the arachidonic acid
and mechanosensitive TREK channels, the acid-sensitive
TASK channels, the alkaline-sensitive TALK channels, the
halothane-inhibited THIK channels, and the TRESK
channel (Goldstein et al., 2001, 2005). Two-pore K+
channels have a widespread tissue distribution being
expressed in neuronal and non-neuronal tissues (Lesage
and Lazdunski, 2000). They are also called leak channels
because they are constitutively open. The activity of these
channels is influenced by a plethora of chemical and
physical stimuli—oxygen tension, pH, lipids, mechanical
stretch, neurotransmitters, and G-protein-coupled receptors (Plant et al., 2005)—and thus, may be involved in
many physiological functions relevant for RPE cells
including maintenance of the resting membrane potential,
pH regulation, and K+ homoeostasis. As some of these
channels are blocked by low concentrations of Ba2+
(Decher et al., 2001; Meadows et al., 2000; Patel et al.,
2000) it cannot be excluded that some of the Ba2+-sensitive
effects measured in Ussing chamber experiments must be
attributed to two-pore channels and not exclusively to
inwardly rectifying K+ channels as concluded in most
reports. However, as there is no direct evidence for the
expression of two-pore channels in the RPE, their possible
role in the RPE will not be further discussed.
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
272
3.4. Summary of K+ channel function
extracellular K+ concentrations in that they have an
increased K+ conductance when extracellular K+ is
reduced. Thus, they are activated when light falls on the
retina as illumination is accompanied by a decrease in
subretinal K+ concentration. By this mechanism, Kir7.1
channels are involved in the K+ homoeostasis in the
light. The Kv7 channels on the other hand are regulated by
Gq/11-protein-coupled receptors. These receptors activate
phospholipase C, which hydrolyzes PIP2 into InsP3 and
DAG, leading to an inhibition of Kv7 channels and an
increase in intracellular free Ca2+, which in turn activates
Ca2+-activated K+ channels. If these two K+ channels are
expressed in opposing membranes of the RPE cells, e.g. the
M channels in the apical membrane and the BK channels in
the basolateral membrane, this would lead to a regulated
K+ transport through the RPE. Under normal conditions,
K+ recycling across the apical membrane would be favored
whereas upon activation of Gq/11 proteins, the absorption
of K+ ions through the basolateral membrane would be
favored. This regulation of K+ transport direction could
support water transport through the RPE, which is
elevated upon ATP stimulation, although this water flux
is mainly thought to be coupled to Cl absorption. This
Cl absorption is also increased by the activation of
purinergic receptors, mediated by Ca2+-activated Cl
channels. An additional role of the BK channels is
probably to counteract the voltage-gated Ca2+ channels
In Fig. 3, a scheme is presented that summarizes the
possible roles of K+ channels in RPE cells. Although it has
not been resolved which K+ channels are expressed in the
RPE and what their specific functions are, some conclusions can be drawn from the electrophysiological data.
It is well accepted that the Na+/K+-ATPase, the
Na+-K+-2Cl cotransporter, and an inwardly rectifying
K+ channel are expressed in the apical membrane of RPE
cells. They are functionally coupled via Na+ ions, which
leave the cells through the Na+/K+-ATPase and are used
by the Na+-K+-2Cl cotransporter to uptake K+ and Cl
into the cell. K+ is recycled into the subretinal space
through an inwardly rectifying K+ channel and Cl ions
leave the cells across the basolateral membrane through a
not yet identified Cl channel. This Cl absorption drives
apical to basolateral water transport. Kv7 channels and
Ca+-activated K+ channels may also play a role in K+
homoeostasis of the subretinal space. Due to the negative
activation threshold of Kv7 channels they may also be
involved in the K+ recycling at the apical membrane. As
they do not inactivate at the negative membrane potentials
in RPE cells, they are capable of conducting a sustained
outward current. The participation of two different K+
conductances allows the cells to fine-tune the K+ efflux.
The inwardly rectifying Kir7.1 channels are regulated by
K+
Epinephrine ATP
M channel
(Kv7)
apical
K+
Kv1.3
AQ1
?
PLC
H 2O
3 Na+
Kir7.1
Kir4.1
Gαq/11
ati
sm
p la m
o
d
E n icu l u
t
Re
K+
K+
Na+
2 K+
2 Cl-
IP3
Ca2+
c
H2O
IP3R
Ca2+
?
L-type channel
basolateral
BK
channel
K+
AQ1
Cl channel
CaCl
channel
Kv1.3
Cl-
K+
Cl-
H 2O
Fig. 3. Schematic diagram summarizing how different K+ currents may be involved in RPE cell function. All ion channels as well as the water channels
are drawn in blue, second-messenger pathways are in yellow with red arrows, ion transporters are drawn in light blue. The Na+/
K+-ATPase and the Na+-K+-2Cl cotransporter are drawn in grey, G-protein-coupled receptors and their ligand in pale blue. Black arrows show
the ion through the cell and through the cell membrane, green arrows mean activating influence, red arrows inhibiting influence. Abbreviations are:
AQ1 ¼ aquaporine 1; ATP ¼ adenosine triphosphate; BK channel ¼ large conductance Ca2+-activated K+ channel; CaCl channel ¼ Ca2+-activated Cl
channel; Gaq/11 ¼ G protein a-subunit q/11; IP3 ¼ inositol-1,4,5-trisphosphate; PLC ¼ phospholipase C.
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
in the RPE as the entering Ca2+ activates BK channels.
Their activation in turn leads to the hyperpolarization of
the cell and consequently to the closure of the voltagegated Ca2+ channels. The role of the other voltage-gated
K+ channels including Kv1.3 in RPE cells remains obscure
as these channels should be inactive at the RPE cell’s
resting membrane potentials. Possibly, they are involved in
cell proliferation/differentiation and wound-healing processes.
4. Chloride
4.1. The role of Cl and Cl channels in cell function
Chloride is the most abundant permeable anion under
physiologic conditions; therefore, anion channels (hereafter
only referred to as chloride channels) mostly conduct
chloride. In contrast to most other ions, the Cl resting
concentration significantly differs among various mammalian cell types and during cell development (Cherubini
et al., 1991; Chipperfield and Harper, 2000). Like other
ions, Cl transport is regulated across plasma membranes
as well as membranes of intracellular organelles. Chloride
can be actively released out of cells, accumulated within
cells, or passively distributed or co-transported to maintain
electroneutrality. Depending on Cl ion channel localization, in the plasma membrane or intracellular organelles,
Cl channels are involved in modulating the excitability of
cells, transepithelial salt transport, cell volume regulation
and homoeostasis or regulation of organelle volume, the
acidification of intra- and extracellular compartments,
calcium or proton gradients, the cell cycle, and apoptosis
(Jentsch et al., 2005c; Nilius and Droogmans, 2003). In
many epithelia Cl channels are involved in the vectorial,
transepithelial transport of salt and water (Begenisich and
Melvin, 1998; Spring, 1998). Cl channels also help protect
epithelial cells against excessive osmotic cell swelling
(Sarkadi and Parker, 1991; Spring and Ericson, 1982;
Strange, 2004; Strange et al., 1996). The existence of Cl
conductance is important for endosomal, lysosomal, and
phagolysosomal vesicles and turnover along the endocytic
apparatus (Faundez and Hartzell, 2004; Jentsch et al.,
2005a).
Three major Cl ion channel families (A–C) have been
well characterized using molecular means. Other Cl
conductances/channels (D) are described either by expression studies or by functional characterization, but their
molecular counterpart and /or their role in health and
disease remains unknown.
(A) Voltage-gated Cl channels (CLC1–7, CLC-K1,2,
CLC-0), (B) Cystic fibrosis transmembrane conductance
regulator (CFTR) channel, and (C) extracellular ligandgated ion channels such as g-aminobutyric acid (GABA) or
glycine receptors have been distinguished. Some of these
Cl channels are located primarily, or exclusively, in
membranes of intracellular organelles, but their localisation is still controversial (ClC3–7). (D) Two different
273
classes of Cl channels primarily defined from their
mechanism of activation and biophysical properties are:
(I) calcium-activated Cl channels (CaCC) (Evans and
Marty, 1986) and (II) swelling-activated/volume-regulated
anion channels (VRAC). However, this classification is an
oversimplification as many Cl channels are regulated by
more than one mechanism and might consist of molecularly diverse classes. It should be mentioned that most Cl
channel blockers are unselective, have a low potency, and
have various side effects; therefore, pharmacological
studies have to be interpreted with caution (d’Anglemont
de Tassigny et al., 2003; Jentsch et al., 1999; Kidd and
Thorn, 2000). Finally, the discrepancy of functional Cl
channels and actual cloned proteins may stem from the fact
that there are no conserved gene sequences characterizing
Cl channels among the known channel families. Consequently, entire families of Cl channels may not have been
determined.
4.2. Cl ions in RPE function
4.2.1. Fluid transport
The RPE mediates water transport from the subretinal
space to the choroid. Together with Müller glial cells,
which transport water from the subretinal space to the
vitreous, the RPE controls the volume of the subretinal
space (Bringmann et al., 2005; Hughes et al., 1998;
Marmor, 1983; Newman and Reichenbach, 1996; Steinberg
et al., 1983; Strauss, 2005). In fact, the primary function of
Cl channels in secretory cells is transepithelial ion
transport (Begenisich and Melvin, 1998; Do and Civan,
2004). The retina produces a large amount of water due to
its enormous metabolic turnover and intraocular pressure
drives water movement from the vitreous body through the
retina. This water is eliminated from the subretinal space
into the choroid plexus by the RPE and into the vitreous by
Müller cells. Therefore, fluid transport from choroid to
retina (secretion) and vice versa (absorption) by the RPE
are essential pathways for fluid regulation in the eye. Fluid
absorption must occur without interruption for retinal
adherence and interaction with the RPE to be maintained
(Hamann, 2002; Marmor, 1990, 1993; Verkman, 2003). In
pathologic states like retinal detachment or macular
oedema, increased fluid absorption by the RPE may lead
to a recovery (Bringmann et al., 2004; Marmor, 1999).
RPE transport of Cl and K+ is thought to drive
transepithelial water transport. Under control conditions it
was estimated that the Cl conductance is up to 70% of the
total basolateral conductance. The transport rate of water
was estimated between 1.4 and 11 ml cm2 h1 (Gallemore
et al., 1997; Hamann, 2002; Strauss, 2005). Fluid absorption involves different mechanisms operating in the apical
and basolateral membranes of the RPE cells (Fig. 5).
Several Cl transporters use the energy stored in transmembrane gradients of other ions to move Cl across the
RPE cell membranes against an electrochemical gradient.
The electrogenic Na+/K+-ATPase generates a Na+
ARTICLE IN PRESS
274
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
gradient from the extracellular (subretinal) to the intracellular space, which also can be measured as the steady-state
retina-positive potential of about 10 mV (Gallemore et al.,
1997). This so-called standing potential depends on the
separation of apical and basolateral properties. In the dark,
photoreceptors maintain K+ recycling leading to a higher
subretinal K+ concentration than during illumination of
the retina (Bialek and Miller, 1994). The increase in [K+]o
stimulates the apical Na+-K+-2Cl cotransporter, thereby
increasing the cellular uptake of K+ and Cl (Adorante
and Miller, 1990; Hamann, 2002; Hughes et al., 1989;
Joseph and Miller, 1991; Keller et al., 1988; Kennedy,
1990, 1994; la Cour, 1991a, b; Lin and Miller, 1991). Thus,
a high intracellular Cl activity of up to 60 mM is
established providing a driving force for Cl out of the
cell (Bialek and Miller, 1994; Fujii et al., 1992; Quinn et al.,
2001; Rymer et al., 2001). Retinal adhesion as an indicator
of functional fluid transport is lost by inhibition of the
apical Na+/K+-ATPase by ouabain (Endo et al., 1988;
Marmor, 1983; Marmor and Yao, 1989). Thus, Cl
absorption in tight epithelia like the RPE is accompanied
by the transport of water to balance osmotic pressure
through aquaporins (Bialek and Miller, 1994; Tsuboi and
Pederson, 1988a, b; Verkman, 2003). Evidence strongly
suggests that Cl exits across Ca2+-activated, volumeactivated, and/or cAMP-activated ion channels in the
basolateral membrane of the RPE, but its differential
contributions are not understood and its molecular
counterparts are still controversial (Gallemore et al.,
1997; Hartzell et al., 2005b; Hughes et al., 1998; Strauss,
2005).
4.2.2. Volume control
Many transporting epithelia regulate their volume after
osmotic perturbations that shrink or swell the cells
(Sarkadi and Parker, 1991; Spring and Ericson, 1982;
Strange et al., 1996). The acute component after cell
swelling is frequently mediated by conductive losses of Cl
and K+, and related water efflux to control cell volume
(RVD). Contrary, cell shrinkage results in regulatory
volume increase (RVI) by mechanisms that decrease K+
and Cl conductance. The specific features of cell-volume
regulation vary considerably among cell types and the
physiological significance is not always proven (la Cour,
1985; Lang et al., 1993, 1998a, b), and we don’t know the
exact extent of physiological osmotic changes on subretinal
and choroidal sites of the RPE cell layer.
RPE cells have effective volume regulating mechanisms,
as shown by volume manipulation. Cultured RPE cells
possess hypertonically-activated Na+-K+-2Cl cotransporter, hypotonically-activated K+-Cl cotransporter,
and a Ba2+—inhibitable hypotonically activated K+ efflux
pathway (Civan et al., 1994; Tsuboi and Pederson,
1988a, b). Remarkably, Cl channel inhibition with the
broad Cl channel blocker NPPB reduced cell shrinkage.
Thus, the RVD of RPE cells likely reflects efflux of K+ and
Cl through parallel channels.
The transport situation seems to differ in the light- and
dark-adapted eye. In the dark, lactate increases subretinally whereas it decreases in the RPE. Therefore, a gradient
from retina to choroid is established which imposes an
osmotic load for the RPE (Adler and Southwick, 1992;
Hamann, 2002; Hsu and Molday, 1994; Winkler, 1995).
During illumination, subretinal K+ decreases, subretinal
space volume increases and RPE cell volume decreases
(Gallemore et al., 1997). Some studies suggest that changes
in subretinal hydration and ion composition, RPE cell
volume regulation and fluid transport mechanisms are
interrelated (Adorante and Miller, 1990; Bialek and Miller,
1994; Botchkin and Matthews, 1993; Civan et al., 1994;
Hartzell and Qu, 2003; Kennedy, 1994). Moreover,
phagocytosis is regulated by illumination and hypoosmotic
cell swelling or application of the Cl channel blocker
tamoxifen decreases RPE phagocytosis (Irschick et al.,
2006; Mannerstrom et al., 2001). Therefore, it is not yet
clear whether cell volume alterations due to photoreceptor
phagocytic uptake, nutrient and lactate transport, or
pathophysiologic disturbances in subretinal osmolarity
(hydration and ion composition) require RPE cell volume
control capability and fluid transport across the RPE.
4.2.3. pH regulation
Controlling the ionic composition of the cytoplasm is
inevitable. Most cells are more alkaline intracellularly than
calculated from electrochemical equilibrium (Chipperfield
and Harper, 2000). Light-induced changes in ion transport
maintain ion homoeostasis in the subretinal space and
evoke changes in pH (Gallemore et al., 1997; Strauss,
2005). The mechanism by which pH homeostasis is
maintained in RPE cells is not completely understood.
RPE cells support photoreceptor function by eliminating
lactic acid thereby regulating the extracellular pH (Hamann, 2002). Transcellular fluid transport requires an
efficient regulation of intracellular pH as well, since some
Cl channels are modulated by pH changes. The subretinal
pH and intracellular pH are regulated by transepithelial
transport of HCO
3 , which needs pathways for Cl
recycling (Edelman et al., 1994a; Lin and Miller, 1991).
Apically, RPE cells have a Na+/H+ exchanger and a Na+/
2HCO3-cotransporter, and basolaterally a HCO
3 /Cl
exchanger, and they are acid-loaded due to transepithelial
lactate transport (Hamann, 2002; Keller et al., 1986, 1988;
Strauss, 2005). Increased cytosolic HCO
3 alkalinizes the
cytosol which stimulates the basolateral Cl–HCO
3
exchanger (Edelman et al., 1994a; Kenyon et al., 1997;
Lin and Miller, 1991). The increased Cl–HCO
3 exchanger
activity results in acidification of the cytosol by HCO
3
extrusion and to Cl uptake. This CI recycled to the
choroidal site by basolateral CI channels and facilitates
acidification by HCO
3 extrusion. Furthermore, the enhanced CI uptake by the Cl–HCO
3 exchanger results in
reduced Cl and water secretion. A favoured hypothesis is
that this recycling determines the balance of fluid absorption and secretion so that the net Cl movement is toward
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
275
the subretinal space. Therefore the RPE behaves as a net
secretory epithelium.
4.2.4. Intracellular organelles
Intracellular Cl channels have received much less
attention, but genetic evidence has revealed some of their
physiological significance. Cl channels in vesicles might be
involved in endosomal, lysosomal, phagosomal, and
synaptic vesicle pathways (Jentsch et al., 2005c).
b
4.3. Identification and properties of Cl channels in the RPE
Molecular counterparts of most Cl channels in the RPE
are still not known. CFTR, ClC-2, -3, -5, and -7, Ca2+
activated, and volume activated Cl channels have been
claimed to be expressed in RPE cells to date (Blaug et al.,
2003; Bosl et al., 2001; Botchkin and Matthews, 1993;
Hartzell and Qu, 2003; Hughes and Segawa, 1993; Loewen
et al., 2003; Reigada and Mitchell, 2005; Strauss et al.,
1996, 1998a, 1999; Ueda and Steinberg, 1994; Weng
et al., 2002; Wills et al., 2000; Wollmann et al., 2006; Wu
et al., 2006). Fig. 4 shows an example of CI currents in
RPE cells.
4.3.1. ClC Cl channels
4.3.1.1. ClC Cl channels in general. Mammalian genome contain nine different genes that encode for the ClC
channels. Some comprise plasma membrane channels
(ClC-1,-2, -ka, kb) and others are predominately in
intracellular membranes (ClC-3, -4, -5, -6, -7). All ClC
channels examined to date are dimers. Most of these
channels are characterized by voltage-dependent gating, all
display a specific permeability sequence (Cl4Br4I)
and are mostly regulated by extracellular anions and pH
(Jentsch et al., 2005a–c, 2002; Nilius and Droogmans,
2003). ClC-1 has been described as a double-barrel model
represented by two identical parallel pores (Middleton
et al., 1996; Pusch and Jentsch, 2005). Each pore
independently opens a fast gate (10 ms range) upon
depolarization which displays inward rectification, and a
common slow inactivating gate (10–100 s) closes both pores
at the same time. The slow gate opens upon hyperpolarization. Gating mechanisms and selectivity of ClC channels
are still unknown.
The functional role of ClC-1 is stabilizing the membrane
potential; ClC-2 channels are inwardly rectifying after
activation by hyperpolarization, cell swelling, or extracellular acidosis; and ClC-3, -4 and -5 are constitutively active
and outwardly rectifying as well as are ClC-Ka and -Kb in
the presence of the b-subunit Barttin. In a recent study,
vesicular ClC-4, -5 and -7 (and possibly ClC-3 and -6) have
been demonstrated to function as electrogenic chloridehydrogen exchangers (also called antiporters) which extrude
protons against their electrochemical gradient, demonstrating secondary active transport (Accardi and Miller, 2004;
Kasper et al., 2005; Kornak et al., 2001; Picollo and Pusch,
2005; Pusch et al., 2006; Scheel et al., 2005).
c
d
Fig. 4. Example of voltage-dependent Cl channel currents in freshly
isolated mouse RPE cells. (a) Schematic diagram showing the recording
configuration. Membrane currents were measured in K+-free solutions in
the whole-cell configuration. (b) Pattern of electrical stimulation: from a
holding potential of 40 mV the cells were stepwise depolarized up to
a potential +50 mV and then stepwise hyperpolarized to a potential
130 mV. The step duration is 50 ms and the increment 10 mV. (c)
Example of currents activated by electrical stimulation. (d) Effect of the
Cl channel blocker DIDS on the voltage-dependent currents. Application
of 1 mM DIDS (right panel) reduced the outwardly directed currents
observed under control conditions (left panel).
4.3.1.2. ClC Cl of the RPE. So far ClC channels -2, -3, -5,
and -7 have been reported to be expressed in human RPE cell
(Fig. 5) by RT-PCR, Western blotting and immunohistochemistry, but biophysical characterization has not been
completed (Hartzell and Qu, 2003; Weng et al., 2002; Wills
et al., 2000). ClC-2 has been implicated as a potential Cl
exit pathway in epithelia. Strikingly, disruption of ClC-2
leads to degeneration of the retina (Bosl et al., 2001). Retinal
degeneration might be secondary due to RPE dysfunction,
which likely would result in abnormal composition of subretinal fluid and secondarily impairs photoreceptor viability.
RPE short-circuit currents are reduced in ClC-2 / mice
ARTICLE IN PRESS
276
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Fig. 5. Schematic diagram summarizing the Cl channel function in RPE cells. All ion channels as well as the water channels are drawn in blue, secondmessenger pathways are in yellow with red arrows, ion transporters are drawn in light blue, intracellular ion channels are drawn in light yellow. The
abbreviations are: AQ1 ¼ aquaporine 1; ATP ¼ adenosine triphosphate, cAMP ¼ cyclic adenosine monophosphate; CaCl ¼ Ca2+-dependent Cl
channel; ClC 2,3,5,7 ¼ ClC chloride channels 2,3,5,7; CFTR ¼ cycstic fibrosis transmembrane regulator; ER ¼ endoplasmic reticulum; Gaq/11 ¼ G
protein a-subunit q/11; IP3 ¼ inositol-1,4,5 trisphosphate; IP3R ¼ inositol-1,4,5 trisphosphate receptor; PLC ¼ phospholipase C; VMD2 ¼ product of
the vitelliform macular dystrophy 2 gene.
supporting the role in transepithelial fluid transport. But the
physiological role of ClC-2 and the pathomechanism of the
RPE phenotype in ClC-2 knockout mice remain unknown.
Currents resembling ClC-2 (hyperpolarization-activated,
slowly activating, inwardly rectifying, acid-activated, Zn2+
-inhibited) have been described for the RPE, but definite
characterization including a description of the anion permeability sequence (Cl4I) is missing (Hartzell and Qu,
2003). In order to understand the contribution of various Cl
ion channels to transepithelial transport the identification of
the apical and basolateral channels is required and is, to date,
unknown.
Cellular pH might be regulated by basolateral Cl/
HCO
3 exchangers and linked Cl cycling through ClC-2.
In support of this, ClC-2 is activated by an increased
intracellular Cl concentration (Catalan et al., 2004;
Niemeyer et al., 2003; Pusch et al., 1999) as well as by
extracellular acidification (Edelman et al., 1994a; Jordt and
Jentsch, 1997).
ClC-3, -5 and -7 transcripts have been found in RPE
cells, but neither has been investigated functionally (Kasper
et al., 2005; Kornak et al., 2001; Stobrawa et al., 2001). In
general, these channels are located on intracellular vesicles,
endosomes, and lysosomes. Their disruption might impair
the acidification of intracellular compartments. Whether or
not ClC-3 channels may get to the cell membrane is
controversial (Coca-Prados et al., 1996; Do et al., 2005;
Duan et al., 1999; Hara-Chikuma et al., 2005; Jentsch
et al., 2005b, c). ClC-3, -5 and -7 channels may provide an
electric shunt for the H+-ATPase in acidic intracellular
compartments like endosomes, and ClC-7 to a large degree
to lysosomes, and therefore are likely involved in processes
like endocytosis (Hartzell et al., 2005b; Kasper et al., 2005;
Kornak et al., 2001; Piwon et al., 2000; Stobrawa
et al., 2001). An important RPE cell function, phagocytosis, depends on vesicular membrane trafficking, lysosomal fusion, phagolysosomal maturation, and digestion
(Besharse and Defoe, 1998; Hartzell et al., 2005b).
Lipofuscin granules which originate from phagocytosis
accumulate with age in the RPE, the largest increase
occurring after the first decade of life in humans (Delori
et al., 2001). Intracellular Cl ion channels are involved in
regulating pH and ionic composition of functionally
different organellar compartments. Perturbation of the
ionic composition of cell organelles by inhibition of the V
type ATPase or by addition of a Na+ ionophor impairs
RPE functions like secretion of interphotoreceptor retinoid-binding protein, leading to lysosomal dysfunction and
inhibition of phagocytosis of photoreceptor outer segments
(Deguchi et al., 1994; Edwards et al., 1987; Mahon et al.,
2004; Peters et al., 2006; Sundelin et al., 1998; Sundelin and
Terman, 2002; Toimela et al., 1998). Therefore, regulation
of vesicular [Cl] is important in RPE cells in daily
processes. Some interference might not be obvious
immediately, but might still bear some age-related pathophysiological implications.
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
4.3.2. CFTR
4.3.2.1. CFTR in general. CFTR resides at the membrane
surfaces and in endosomes in various epithelia, is involved
in transepithelial fluid transport. In some epithelia (e.g.
colon) CFTR represents the major apical Cl conductance
(Begenisich and Melvin, 1998; Greger, 2000; Jentsch et al.,
2002). A genetic defect in CFTR causes cystic fibrosis, a life
threatening disease characterized by the clogged ducts or
tubes of various organs by mucus or other secretions
(Fuller and Benos, 1992; Thiagarajah and Verkman, 2003).
CFTR belongs to the traffic ATPases or so called ABC
transporters, but represents the only protein in that family
which was identified as ion channel (Anderson et al., 1991).
Intracellularely, CFTR contains two nucleotide binding
domains (NBD1 and NBD2) separated by a large polar
regulatory domain with nine sites for phosphorylation by
PKA and seven sites for phosphorylation by PKC
(Riordan et al., 1989). ATP-driven conformational changes
by binding of ATP, which in other ABC proteins fuels
uphill substrate transport across cellular membranes, in
CFTR, opens and closes a gate to allow transmembrane
flow of anions down their electrochemical gradient
(Gadsby et al., 2006; Vergani et al., 2005). Furthermore,
cAMP-dependent activation of PKA activates CFTR
channels, which are voltage independent and display a
permeability sequence of Br 4Cl4I4F (Dawson
et al., 1999; Li et al., 1988; McCarty, 2000).
Besides its Cl channel function, CFTR might be
involved in the regulation of other ion channels, transporters, or cellular functions like: electrolyte and osmolyte
transport, cell volume regulation, efflux of ATP through
CFTR channels or CFTR regulated ATP release by
vesicles, inhibition of epithelial sodium channel (ENaC),
CaCC and VRAC.
4.3.2.2. CFTR of the RPE. Human RPE cells display
CFTR mRNA expression and CFTR protein has been
immunolocalized to the apical and basolateral membranes.
A cAMP-dependent increase in transepithelial Cl transport or conductance indicates a functional role of CFTR in
the RPE (Fig. 5) (Blaug et al., 2003; Hartzell et al., 2005b;
Hughes and Segawa, 1993; Loewen et al., 2003; Quinn
et al., 2001; Reigada and Mitchell, 2005; Weng et al., 2002;
Wills et al., 2000).
A main lead for CFTR in RPE function stems from the
fact that part of the transepithelial Cl transport and net
fluid absorption is dependent on cAMP-activated Cl
conductance (Gallemore et al., 1997). An increase of
[cAMP]i in foetal human RPE induces a decrease in
basolateral membrane resistance and the activation of a
Cl conductance (Weng et al., 2002). Thereby intracellular
Cl decreases and generates a depolarization. Moreover,
cAMP increases short circuit current, increases transepithelial potential, and decreases the ratio of basolateral
to apical resistance (Hughes et al., 1987, 1988; Miller and
Farber, 1984). Although these data suggest an increase in
basolateral conductance, the effects are complex, possibly
277
reflecting different transport pathways. ClC-2 is another
possible cAMP-activated candidate. ClC-2 is also a cAMP
activated Cl channel with a NO
3 ¼ I 4Br 4Cl b
HCO3 permeability sequence (Hughes and Segawa,
1993). From studies of CFTR /ClC-2 double knockout
mice it was concluded that ClC-2 does not compensate for
CFTR (Zdebik et al., 2004), suggesting that CFTR and
ClC-2 have differential roles for Cl ion efflux regulation.
Another line of observations supports a different or
additional role for CFTR in RPE cells. RPE cells release
ATP and its release mechanism has been linked to CFTR
Cl channels in various epithelial cells, including RPE.
cAMP-activating RPE cell manipulation led to increased
apical ATP release whereas glibenclamide and the more
specific inhibitor CFTR-172 (Ma et al., 2002) prevented
hypotonically triggered ATP release. A precise pathway for
the release of Cl and ATP in RPE cells and the
relationship to CFTR remains controversial (Braunstein
et al., 2001; Prat et al., 1996; Reigada and Mitchell, 2005).
4.3.3. Ca2+-activated Cl channels
4.3.3.1. Ca2+-activated Cl channels in general. Chloride
channels activated by intracellular calcium (CaCC) are
widely expressed. Voltage-dependence and Cl concentration gradients allow Cl influx or efflux that produces
hyperpolarization or depolarization, respectively. CaCC
conductances are implicated in diverse functions like
transepithelial fluid transport, excitability modulation,
and regulation of smooth muscle cell tonus. Endogenous
calcium activated Cl currents are voltage-dependent
(outwardly rectifying), and display a SCN4NO
3 4I 4
Br 4Cl 4F permeability sequence. The voltage-dependence disappears with high [Ca2+]i (Begenisich and Melvin,
1998; Eggermont, 2004; Hartzell et al., 2005a; Jentsch
et al., 2002; Kidd and Thorn, 2000; Melvin, 1999; Morris,
1999; Nilius and Droogmans, 2003; Scott et al., 1995;
Suzuki et al., 2006).
So far at least three potential classes of CaCC have been
suggested: (i) activated by increased [Ca2+]i, (ii) regulated
by calmodulin-dependent protein kinase II, and (iii)
cGMP-dependent channel types (Jentsch et al., 2002;
Matchkov et al., 2004). Putative candidate proteins (e.g.
CLCA, bestrophin) have been cloned from various species
(Bakall et al., 1999; Gandhi et al., 1998; Marquardt et al.,
1998; Stohr et al., 2005; White et al., 2000), but the
correlation of biophysical properties of heterologously
expressed channels to native calcium dependent Cl
currents are still unsolved or controversial (Fuller et al.,
2001; Jentsch et al., 2002; Loewen et al., 2002a; Nilius and
Droogmans, 2003; Pauli et al., 2000). Bestrophins have
been shown to be anion-selective channels and are
activated by physiological [Ca2+]i in heterologous expression systems (Fischmeister and Hartzell, 2005; Hartzell et
al., 2005b; Hartzell and Qu, 2003; Qu et al., 2003, 2004,
2006b; Qu and Hartzell, 2004; Sun et al., 2002; Tsunenari
et al., 2006). Four bestrophin isoforms have been
discovered, all of which generate membrane currents in
ARTICLE IN PRESS
278
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
transfected HEK293 cells and two of them with a I4
Br4Cl permeability sequence matching endogenous
CaCC (Qu et al., 2004, 2006b; Qu and Hartzell, 2004;
Sun et al., 2002; Tsunenari et al., 2006). But heterologously
expressed CLCAs and bestrophins showed instantaneous
but not time dependent, outwardly rectifying currents as
characteristic for endogenous CaCC. Other studies suggest
that CLCAs and bestrophins might not only constitute a
Cl channel itself, but also modulate other ion channels
(Loewen et al., 2002a, b, 2003; Marmorstein et al., 2004,
2006; Pusch, 2004; Rosenthal et al., 2006).
4.3.3.2. Ca2+-activated Cl channels in the RPE. Indirect
evidence from choroid-RPE preparations suggests that
CaCC might be the light-induced basolateral Cl conductance important for fluid, and possibly volume regulation, in the RPE (Fig. 5). Extracellular epinephrine and
ATP activate calcium signalling, basolateral Cl conductance, and fluid absorption in RPE cells (Edelman and
Miller, 1991; Joseph and Miller, 1991; Leipziger, 2003; Lin
and Miller, 1991; Peterson et al., 1997). ATP is currently
still discussed as a factor activating fluid transport across
RPE cells due to illumination (light-peak substance) and
triggering this fluid absorption by stimulating apical P2Y2
receptors with a specific agonist can reduce the size of fluid
blebs in the subretinal space in vivo (Maminishkis et al.,
2002; Meyer et al., 2002). External application of DIDS (an
unspecific Cl channel blocker) reduced basolateral Cl
conductance and fluid absorption and therefore CaCC are
suggested to be important for fluid and ion transport
regulation across the RPE (Bultmann and Starke, 1994;
Edelman et al., 1994a; Gallemore et al., 1997; Peterson
et al., 1997; Quinn et al., 2001; Soltoff et al., 1993;
Ziganshin et al., 1996). CaCC have been shown to be
present in Xenopus, rat, and mice RPE cells by patchclamp measurements (Botchkin and Matthews, 1993;
Hartzell and Qu, 2003; Marmorstein et al., 2006; Strauss
et al., 1996, 1998a, 1999; Ueda and Steinberg, 1994;
Wollmann et al., 2006). InsP3 induced calcium influx into
the cell activates a Cl conductance, which was inhibitable
by DIDS, but not flufenamic acid or Zn+. The currents
showed outward rectification, a fast voltage-dependent
activation, and transient activation depending on [Ca2+]i.
Direct application of a high intracellular Ca2+ concentration activates these currents as well.
VMD2 (hBest1), was originally positionally cloned from
families with Best’s vitelliform macular dystrophy (Marquardt et al., 2000; Petrukhin et al., 1998; Stanton et al.,
2006; Stohr et al., 2002; White et al., 2000) and its gene
product was recently suggested as a novel prospective Cl
channel candidate expressed by the RPE (Fischmeister and
Hartzell, 2005; Hagen et al., 2005; Qu et al., 2004, 2006b;
Qu and Hartzell, 2004; Stanton et al., 2006; Sun et al.,
2002; Tsunenari et al., 2003, 2006). hBest1 and hBest2, are
highly expressed in the RPE (Bakall et al., 2003; Kramer
et al., 2004; Marmorstein et al., 2000; Stohr et al., 2002).
Best patients have a reduced light-peak propably caused by
a reduced basolateral Cl conductance. Heterologeous
expression of the VMD2 gene product bestrophin-1 in the
human kidney cell line HEK293 results in an increase in the
membrane conductance for Cl, which was dependent on
increased intracellular free Ca2+ (Sun et al., 2002). Sidedirected mutagenesis of the putative pore region of the
channel protein resulted in a change of the permeability for
SCN ions of the channel (Qu et al., 2004, 2006a; Qu and
Hartzell, 2004).
4.3.4. Volume regulated anion channels
4.3.4.1. Volume regulated anion channels in general. Volume regulated anion channels (VRAC) are ubiquitously
expressed in mammalian cells and play an important role in
cell volume homoeostasis (Clapham, 1998; d’Anglemont de
Tassigny et al., 2003; Hoffman et al., 1998; Hoffmann,
1992; Lang et al., 1998a, b, 2005; Nilius and Droogmans,
2003; Nilius et al., 1997; Okada, 2004; Okada and Maeno,
2001; Okada et al., 2001; Sardini et al., 2003). Small cell
volume changes might even be linked to vectorial transport
of solutes and water across epithelia (Foskett, 1990). The
activation of VRAC is believed to provide one of the initial
triggers linking cell swelling to the subsequent loss of KCl
through Cl and K+ channels—osmolyte and water efflux
resulting in a RVD. The associated current is referred to as
ICl,swell. In experimental setups, exposure to hypotonic
solutions is the common technique to swell cells. In most
cells this manoeuvre activates outwardly rectifying anion
channels (anion influx), which are voltage-dependent
(inactivate at positive membrane potentials in many, but
not all cell types), depend on intracellular ATP, and exhibit
an anion selectivity of SCN 4I 4NO
3 4Br 4Cl 4
F 4glutamate . This conductance is also regulated by cell
shrinkage (exposure to hypertonic solution is the common
experimental manoeuvre) and in some cells a similar
conductance initiated by the hypotonic solution appears
to develop spontaneously under isotonic conditions, which
can be suppressed by exposure to hypertonic solutions.
Moreover, ICl,swell may be activated by a reduction in
intracellular ion strength, shear stress, and application of
GTPgS. Some lines of evidence suggest that VRAC is also
permeable to lactate and bicarbonate pointing to a role in pH
regulation. This putative channel was named volumestimulated osmolyte and anion channel (VSOAC) and
depends like VRAC on intracellular ATP (Jackson et al.,
1994). Besides ICl,swell, other ion channels (e.g. ClC-2,
bestrophin, maxi-Cl channel) appeared to be volumeactivated as well, but display discernable biophysical
characteristics. Besides these mentioned candidates others
have been proposed, but none of these has yet received
acceptance (Clapham, 1998; Jentsch et al., 2002; Pusch, 2004).
ICl,swell may also be important for other processes including regulation of membrane excitability, transcellular Cl
transport, cell proliferation, release of nucleotides (e.g.
ATP), and apoptosis. VRAC may not be a single entity,
but may instead stand for several different ion channels
(Lang et al., 1993, 1998b; Nilius et al., 1996).
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
4.3.4.2. Volume regulated anion channels in the
RPE. Epithelial cells involved in transepithelial transport need to balance their ion transport rates to
maintain their volume within certain limits. Furthermore, RPE cells phagocytose photoreceptor outer segments
at a high rate likely challenging cell volume and maybe
leading to successive osmolarity changes, which must be
compensated (Hartzell et al., 2005b; Nguyen-Legros and
Hicks, 2000; Strauss, 2005; Wettstein et al., 2000). Hypoosmotic swelling inhibits RPE phagocytosis and suggests that
cell volume regulation and phagocytic functions are linked
(Irschick et al., 2006). The RPE is likely exposed to lightdependent osmotic challenge, as photoreceptor activity alters
the solute composition in the restrictive volume of the
subretinal space (Bialek and Miller, 1994; Borgula et al.,
1989; Dmitriev et al., 1999; Huang and Karwoski, 1992; Li et
al., 1994b; Oakley and Steinberg, 1982; Shirao et al., 1987).
And it has been demonstrated that iso-osmotic changes in
RPE cell volume affect the level of RPE-retina adhesivity
(Marmor, 2005) and also alter the volume of the subretinal
space (Adorante, 1995; Adorante and Edelman, 1997;
Adorante and Miller, 1990; Bialek and Miller, 1994).
Moreover, transepithelial salt and water transport could
place an additional osmotic load on the RPE cell.
Hypotonic swelling of RPE cells in vitro leads to a RVD
(Fig. 5) due to parallel efflux of Cl and K+ through
respective ion channels (Adorante, 1995; Civan et al.,
1994). Inhibition of RVD by both basal Ba2+ (blocking
K+ channels) and elevated basal K+ strongly suggests the
RVD is mediated by K+ efflux across the basolateral
membrane (Adorante, 1995; Hughes et al., 1988; Immel
and Steinberg, 1986; Kennedy, 1994). The K+-induced
depolarization also reduced the driving force for conductive Cl efflux across the basolateral membrane. Therefore,
cell swelling is most likely due to a decrease in the driving
force for K+ and Cl efflux across the basolateral
membrane during continued solute uptake by the electroneutral Na+-K+-2Cl and electrogenic Na+/HCO
3 cotransporters located at the apical membrane of the RPE
(Hughes et al., 1988, 1989; la Cour, 1991a, b).
RPE cell shrinkage in vitro produces RVI likely reflecting
the work of ion antiporter under baseline conditions. Na+
-K+-2Cl cotransporter activation likely increases [Cl]i as
has been discussed for fluid transport and, therefore, might
again be coupled to a Cl efflux through ion channels.
In RPE cells, application of intracellular pressure or
decrease in extracellular osmolarity induced cell swelling
and activation of outwardly rectifying currents. These
currents showed time-dependent inactivation and were
inhibited by DIDS, SIDS. In addition, the currents were
sensitive to the potential VRAC blockers niflumic acid and
anthracene-9-carboxylic acid (Botchkin and Matthews,
1993; Fischmeister and Hartzell, 2005; Ueda and Steinberg,
1994). Cell swelling increased Cl conductance by as much
as 5- to 13-fold. The mechanism of activation of the
swelling activated Cl conductance is not identified yet,
and could consist of, or be modulated by, Ca2+-activated
279
channels. The latter hypothesis stems from the observation
that hyperosmotic shrinkage of RPE cells suppressed part
of the Cl current and that heterologous expression of
hBest1 induced Cl currents are likewise profoundly
inhibited by that manoeuvre (Fischmeister and Hartzell,
2005). Among other candidate volume sensitive channels
ClC-3 (CLCN3), ICln (CLNS1A) and bestrophin (VMD2)
is located in RPE (NEI EST search; http://neibank.nei.
nih.gov/).
4.3.5. Other Cl channels
There are further well-characterized putative anion
channels not mentioned in this review. Searching NCBI
Geo database profiles (http://www.ncbi.nlm.nih.gov/geo/)
(Barrett et al., 2005) of the genes expressed in the human
RPE determined through serial analysis of gene expression
(SAGE) and both in the human and in the mouse RPE/
choroid library of EST (http://neibank.nei.nih.gov/)
yielded nonconfirmed data on expression of CLICs,
GABAA receptors, MCLC and Tweety.
GABA (g-aminobutyric acid) A and C receptors are
ligand-gated Cl channels, whereas GABA B receptors are
metabotropic receptors (Chebib and Johnston, 1999;
Hevers and Luddens, 1998; Jentsch et al., 2002). GABAA
and GABAC receptors are biochemically, pharmacologically, and physiologically different. Currently, there are 16
human GABAA receptor subunits (a1–6, b1–4, g1–4, d, e)
and two human GABAC receptor subunits (r1 and r2) that
have been cloned. GABAA receptor subunits GABRa2,
GABRa6, GABRb1, GABRg2, GABRd, GABRe and
GABRp appeared in NCBI Geo database for native or
cultured RPE cells. Using antibodies recognizing GABAA
receptor subunit b2 and b3 showed positive immunoreactivity for human and rat RPE in cultured cells, and
distinctive bands at two different molecular masses in
Western blot data (Wood and Osborne, 1996). And,
benzodiazepine binding sites on rat, monkey, and human
RPE cells support the notion of functional GABAA
receptors on RPE cells (Robbins and Ikeda, 1989; Zarbin
and Anholt, 1991), but the details still need to be
elucidated.
The Cl intracellular channel (CLIC1, 4, 5, 6), MID-1related Cl channel (MCLC) and TTYH1–3, human
homologues of the Drosophila melanogaster Tweety (tty)
genes suggested to represent novel maxi-Cl channel
proteins (Suzuki et al., 2006) have not been investigated
in RPE cells.
5. Ca2+
5.1. Ca2+ and cell function in general
Ca2+ ions have a high affinity to proteins (Carafoli,
2005a, b; Williams, 1974, 1994). The binding of Ca2+ to
proteins results in changes of their conformation and
subsequent alteration of the protein function (Carafoli,
2005a, b; Williams, 1974, 1994). With these characteristics,
ARTICLE IN PRESS
280
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
the functional role of Ca2+ differs from that of many other
ions. It can directly act as a modifier of protein function
and, thus, regulate cell function. In many different tissues,
Ca2+ was found to act in the initiation or modulation of
contraction, secretion, gene expression, migration, apoptosis, necrosis, cell division, endocytosis, coagulation,
excitation, energy metabolism, and signal transduction
(Berridge, 2005; Carafoli, 2005a; Shuttleworth, 1997;
Williams, 1974). Ca2+ controls these functions either by
directly triggering protein interaction such as in excitation–contraction coupling or by acting as a secondmessenger, activated in response to extracellular stimuli
(Berridge et al., 2003; Berridge et al., 2000). Small changes
in Ca2+ concentration can have profound changes in cell
function. Normally, cells keep the concentration of
intracellular free Ca2+ very low (at a proximal 100 nM)
and maintain a concentration gradient of 1:10,000 between
intracellular space and extracellular environment, although
their total intracellular Ca2+ content can be quite high
(Berridge et al., 2000, 2003; Carafoli, 2005a; Saris and
Carafoli, 2005). To keep the concentration of intracellular
free Ca2+ low, cells use energy to store Ca2+ in intracellular compartments such as mitochondria or intracellular Ca2+ stores of the endoplasmic or sarcoplasmic
reticulum (Berridge et al., 2000, 2003; Carafoli, 2005a;
Saris and Carafoli, 2005).
Ca2+-induced changes in cell function are triggered by
an increase in intracellular free Ca2+. The specificity of this
signal is encoded by the complex pattern of each Ca2+
signal which follows a specific stimulus (Berridge, 2005;
Berridge et al., 2000, 2003). These patterns differ in the
amplitude, and the spatial- and time-dependent distribution inside the cell. The need for a tight control of a low
intracellular Ca2+ concentration at rest and the generation
of complex patterns of Ca2+ increases during stimulation
requires the presence of many different Ca2+ transporting
proteins, such as Ca2+ conducting ion channels, Ca2+
cotransporters, and Ca2+pumps—as well as the presence
of Ca2+ binding proteins (Berridge, 2005; Berridge et al.,
2000, 2003). Thus the specificity of Ca2+ signals arises
from recruitment of specific sets of Ca2+ transporting or
Ca2+ binding proteins (Berridge, 2005; Berridge et al.,
2000, 2003; Carafoli, 2005a; Saris and Carafoli, 2005;
Williams, 1994).
5.2. Ca2+ homeostasis in the RPE
RPE cells contain with 15 mmol/l higher amounts of
Ca2+ than other cells (Bellhorn and Lewis, 1976; Fishman
et al., 1977; Hess, 1975; Salceda and Riesgo-Escovar, 1990;
Ulshafer et al., 1990). Large amounts of this Ca2+ are
stored in melanosomes (Boulton, 1991; Boulton and
Dayhaw-Barker, 2001; Drager, 1985; Hess, 1975; Ulshafer
et al., 1990). To handle large amounts of Ca2+ the RPE
possesses several Ca2+ transporting proteins. A Na+/Ca2+
exchanger eliminates Ca2+ from the intracellular space in
exchange with 3 Na+ ions (Fijisawa et al., 1993; Loeffler
and Mangini, 1998; Mangini et al., 1997; Salceda, 1989). In
immunohistochemical studies, the transporter could be
identified as the cardiac subtype of Na+/Ca2+ exchanger
(Loeffler and Mangini, 1998), which is also found in
cardiac myocytes and eliminates Ca2+ from the intracellular space to initiate the rhythmic relaxation of the
myocardium. This Na+/Ca2+ exchanger represents a very
efficient Ca2+ transport system to extrude Ca2+ from the
intracellular space. Thus, the changes in intracellular free
Ca2+ in RPE cells can have the same efficient dynamics as
in cardiac myocytes. Due to its stoichiometry of 1 Ca2+
exchanged with 3 Na+, the Na+/Ca2+ exchanger is an
electrogenic transporter. Accordingly, elimination of Ca2+
from the intracellular space should lead to moderate cell
depolarization. On the other hand, strong depolarization
of the cell can change the transport direction in the way
that the Na+/Ca2+ exchanger now transports Ca2+from
the extracellular space into the cell. The extrusion of Ca2+
from the cytosol by the Na+/Ca2+ exchanger is supported
by the activity of the plasma membrane Ca2+-ATPase
which hydrolyzes ATP for an active transport of Ca2+ out
of the cell against its concentration gradient into the
extracellular space (Kennedy and Mangini, 1996). Furthermore, like many epithelia, RPE cells are connected to each
other via gap junctions, forming a functional syncytium
(Gomes et al., 2003; Himpens et al., 1999; Himpens and
Vereecke, 2000; Pearson et al., 2004; Stalmans and
Himpens, 1997). The cell-to-cell connection via gap
junction channels enables Ca2+ signals to spread between
the cells within the epithelial monolayer. Thus, increases in
intracellular free Ca2+ can influence the function of the
entire RPE.
In the past, a large number of receptors, which stimulate
an increase in intracellular free Ca2+ as a second messenger
have been identified in the RPE. Most of these receptors
act via stimulation of G proteins or tyrosine kinase activity
to initiate a formation and release of InsP3 into the cytosol.
InsP3 activates a release of Ca2+ from InsP3 sensitive
cytosolic Ca2+ stores (Ammar et al., 1998; Berridge, 2005;
Berridge et al., 2000, 2003; Crook and Polansky, 1992;
Feldman et al., 1991; Fragoso and Lopez-Colome, 1999;
Karihaloo et al., 1997; Kuriyama et al., 1992; Mergler and
Strauss, 2002; Nakashima et al., 1989; Strauss et al., 1996).
This in turn can stimulate an increase in the membrane
permeability for Ca2+ and an influx of extracellular
Ca2+ into the cell. The latter event results in a sustained
Ca2+ increase and a long-lasting change in cell function
(Berridge, 2005; Berridge et al., 2000, 2003). However,
some receptors, ionotropic glutamate receptors or purinergic P2X receptors, function as ligand-gated ion channels
and directly increase intracellular free Ca2+ (Ryan et al.,
1999). Other receptors such as purinergic P2Y receptors
increase intracellular free Ca2+ by release of Ca2+ only
from cytosolic Ca2+ stores without generating an influx of
Ca2+ into the cell (Collison et al., 2005; Mitchell, 2001;
Peterson et al., 1997; Poyer et al., 1996; Ryan et al., 1999;
Sullivan et al., 1997). In general, a sustained increase in
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
intracellular free Ca2+ requires an influx of extracellular Ca2+ into the cell and, thus, the presence of
Ca2+-conducting ion channels. Thus the understanding of
the Ca2+-dependent regulation of RPE cell function was
substantially improved with patch-clamp studies on RPE
cells uncovering the expression of different Ca2+ channels.
5.3. Ca2+ and RPE function
As stated above, the RPE has many functions which are
essential to maintain structural integrity and excitability of
photoreceptors (Bok, 1993; Steinberg, 1985; Strauss, 2005).
Many of these functions are regulated by changes in
intracellular free Ca2+. These are:
Dark adaptation of photoreceptor activity: This occurs by
Ca2+-dependent mechanisms inside the photoreceptor
outer segments (Korenbrot, 1995; Korenbrot and
Rebrik, 2002). The required Ca2+ enters the photoreceptor outer segments through cGMP-gated cation
channels, which are open in the dark and conduct Ca2+.
The subretinal Ca2+ concentration in the dark is larger
than in the light (Gallemore et al., 1994). A Ca2+ source
for this increase in subretinal Ca2+ might be melanosomes of the RPE (Drager, 1985; Hess, 1975; Lavallee
et al., 2003; Moriya et al., 1996; Salceda and SanchezChavez, 2000) which is suggested by the finding that
albino rats show slower dark adaptation than normal
pigmented rats (Behn et al., 2003).
Transepithelial transport of ions and water: Increases in
intracellular free Ca2+ achieved either using a Ca2+
ionophore (Joseph and Miller, 1992) or by the stimulation of a-adrenergic (Edelman and Miller, 1991; Joseph
and Miller, 1992; Nash and Osborne, 1995; Quinn and
Miller, 1992; Quinn et al., 2001; Rymer et al., 2001) or
purinergic receptors (Maminishkis et al., 2002; Mitchell,
2001; Peterson et al., 1997; Ryan et al., 1999; Sullivan
et al., 1997) were found to stimulate Cl and water
transport by RPE cells. The Ca2+-dependent stimulation of transepithelial Cl transport occurs by three
mechanisms. One is by modulation of the electrical
driving forces for ions to move across the cell membrane (Edelman et al., 1994b). This is achieved by
Ca2+-dependent opening of non-specific cation channels, K+ or Cl channels and subsequent changes in the
resting potential (Bialek et al., 1996; Fischmeister and
Hartzell, 2005; Hu et al., 1996; Joseph and Miller, 1992;
Miller and Edelman, 1990; Quinn and Miller, 1992;
Ryan et al., 1999; Ryan and Kelly, 1998; Sheu and Wu,
2003; Sheu et al., 2004; Strauss et al., 1996; Sun et al.,
2002; Tao and Kelly, 1996). The second mechanism is
opening of Ca2+-dependent Cl channels in the
basolateral membrane (Hartzell and Qu, 2003; Qu
et al., 2004; Quinn et al., 2001; Strauss et al., 1996,
1999; Ueda and Steinberg, 1994). The third mechanism
occurs via the activation of apically located K+
channels (see Section 3).
281
Phagocytosis: Studies on the regulation of phagocytosis
imply that the stimulation of increases in intracellular
free Ca2+ has a regulatory function (Greenberger and
Besharse, 1985; Hall et al., 1991; Heth and Marescalchi,
1994; Nakashima et al., 1989; Nguyen-Legros and
Hicks, 2000; Strauss et al., 1998b). However, the role
of the rise in intracellular free Ca2+ is not clear. Since a
participation of the InsP3 second messenger system was
found to play a role in the initiation of phagocytosis
(Heth and Marescalchi, 1994) an increase in intracellular
free Ca2+ should represent a starting signal for
phagocytosis (Berridge et al., 2003). However, in
another study an increase in intracellular free Ca2+
and subsequent activation of protein kinase C (PKC)
was described as a shut-off signal for phagocytosis (Hall
et al., 1991). These contradictory observations may be
explained by different patterns of Ca2+ increases
underlying these two different regulatory effects.
Secretion: Since in many tissues exocytosis and secretion
are known to be triggered by an increase in intracellularfree Ca2+, it is likely that these processes are triggered
by Ca2+ in the RPE (Barg, 2003; Berridge et al., 2000;
Mears, 2004; Satin, 2000; Shuttleworth, 1997). Stimulation by ATP or various growth factors is known to
induce secretion of growth factor by the RPE (Guillonneau et al., 1997; Mitchell, 2001; Reigada and Mitchell,
2005; Rosenthal et al., 2004, 2005; Slomiany and
Rosenzweig, 2004a, b). Since stimulation of the RPE
by growth factors (Rosenthal et al., 2004, 2005) as well
as by ATP (Mitchell, 2001; Peterson et al., 1997; Ryan
et al., 1999) have been described to result in an increase
in intracellular free Ca2+ it is very likely that these
increases in cytosolic Ca2+ not only represent a signal to
change gene expression (Rosenthal et al., 2005) but also
to trigger the release of other growth factors (Rosenthal
et al., 2004, 2005).
Differentiation: In general, growth factor-dependent
changes in gene expression were described to arise from
increases in intracellular free Ca2+. The underlying
changes in the gene expression are achieved by Ca2+activated phosphorylation of transcription factors (Fields
et al., 2005). Stimulation of the RPE by basic fibroblast
growth factor (bFGF) leads to an increase in intracellular
free Ca2+ as well as to changes in the expression of the
immediate early gene c-fos (Rosenthal et al., 2005).
In summary, a lot of RPE functions to maintain visual
function are controlled by increases in intracellular-free
Ca2+. Thus, Ca2+ channels, which provide major routes
for an influx of extracellular Ca2+ into the cell to increase
intracellular-free Ca2+ must play an important role in the
control of these functions.
5.4. Ca2+ channels of the RPE
So far three types of Ca2+-conducting ion channels have
been described in the RPE: voltage-dependent Ca2+
ARTICLE IN PRESS
282
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
channels, members of transient receptor potential (TRP)
channel family and ligand-gated Ca2+-conducting, nonselective cation channels.
5.4.1. Voltage-dependent Ca2+ channels
5.4.1.1. Voltage-dependent Ca2+ channels in general. Voltage-gated Ca2+ channels are highly specific Ca2+conducting channels which are mainly activated by changes
of the membrane potential toward more positive values
(i.e., by depolarization) (Catterall, 1998, 2000; Catterall et
al., 2005; Striessnig, 1999; Striessnig et al., 2004). Ten
voltage-dependent Ca2+ channels have been described
differing in their voltage-dependence, kinetic behaviour,
and pharmacology. The 10 different channels share a
common molecular architecture. They are composed of an
a1-subunit, a b-subunit, an a2d subunit, and in some cases a
g subunit. The a1-subunit forms the ion-conducting pore
and defines the pharmacological, electrophysiological, and
kinetic characteristics of the channel. The 10 different
channel types can be subdivided into the following
subfamilies: L-type channels (CaV1.1–1.4 or a1S, C, D
and F), N, P/Q and R-type channels (CaV2.1–2.3 or a1A, B
and E) and the T-type channels (CaV3.1–3.3 or a1G–I).
The known four b-subunits modify channel activity,
blocker sensitivity, and targeting of the a1-subunits to the
plasma membrane. In addition, nine different g subunits
and 4 different a2d subunits have been identified. Members
of both subunit types may be involved in intracellular
targeting of the a1 subunits and influence the electrophysiological properties of the Ca2+ channels.
2+
5.4.1.2. The voltage-dependent Ca
channels of the
RPE. So far only one subtype of voltage-dependent
Ca2+ channel has been characterized in detail in RPE cells
(Fig. 6). Ca2+ channels in cultured or in freshly isolated
RPE cells from various species including man were
characterized by patch-clamp recordings of whole-cell
Ba2+ or Ca2+ currents (Rosenthal and Strauss, 2002;
Sakai and Saito, 1997; Strauss et al., 1997, 2000; StrauX
and Wienrich, 1994; Ueda and Steinberg, 1993, 1995;
Wollmann et al., 2006). The usage of Ba2+ as a charge
carrier is a commonly used tool to study L-type Ca2+
channels because these channels display larger Ba2+ than
Ca2+ currents. RPE cells responded to depolarization to
potentials more positive than 30 mV with fast activating
and much more slowly inactivating inward currents (Fig. 6)
(Mergler and Strauss, 2002; Rosenthal et al., 2006; Sakai
and Saito, 1997; Ueda and Steinberg, 1993, 1995;
Wollmann et al., 2006). These currents are modulated by
dihydropyridine derivatives. Dihydropyridines specifically
modulate the activity of L-type channels. For example
BayK8644 stimulates L-type channel activity whereas
nifedipine or nimodipine inhibit L-type channels. In the
RPE, application of the Ca2+ channel activator BayK8644
resulted in larger currents whereas the Ca2+ channel
blocker nifedipine decreased the inward currents
(Rosenthal et al., 2001, 2006; Rosenthal and Strauss,
a
Nystatin
10 mM Ba2+
RPE
Whole-cell currents
b
+ 20 mV
-70 mV
c
0.5 nA
20 ms
d
-80
Pipette potential (mV)
-60 -40 -20
20
-800 control
BayK 8644 (5 µM)
-1600
Membrane current (pA)
Fig. 6. Example of voltage-dependent Ba2+ currents in cultured human
RPE cells indicating the presence L-type Ca2+ channels. (a) Schematic
diagram illustrating the recording technique: the patch-pipette which is
attached to the RPE cell enables a voltage-clamp of the cell membrane
potential as well as measurements of membrane currents at different
potentials. The pipette contains a solution which mimicks the ion
composition of the intracellular space. Furthermore, the pipette solution
contains an ionophore (nystatin), which perforates the membrane patch
inside the inner diameter of the pipette. With this configuration,
membrane currents could be measured without disturbance of the
intracellular milieu, which is essential to measure stable L-type channel
currents. Pipette and bath solution are K+-free to prevent the measurement of K+ currents superimposed to the Ca2+ channel currents. In
addition, the bath solution contains Ba2+ as a charge carrier for Ca2+
channel currents because with Ba2+ as a charge carrier Ca2+ channels
display larger currents. (b) Electrical stimulation to activate currents
through Ca2+ channels: from the holding potential of 70 mV the cells
were depolarized with nine voltage steps of 50 ms duration and +10 mV
increment. (c) Ba2+ currents activated by the stimulation protocol shown
in 6B in a cultured human RPE cell. (d) Current/voltage plot of the
currents shown in Fig. 6C: the maximal current amplitudes were plotted
against the potentials of the stimulation protocol. Furthermore, the effect
of the L-type channel opener BayK8644 is shown in this diagram.
2002; Sakai and Saito, 1997; Ueda and Steinberg, 1993;
Wollmann et al., 2006). These properties are characteristic
for L-type channels and were observed in RPE cells in
primary culture, RPE cell lines, and in freshly isolated RPE
cells. Despite the fact that between the different studies
L-type currents showed some differences in inactivation,
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
voltage-dependence, and dihydropyridine sensitivity, all
studies concluded that L-type channels represent the major
type of voltage-dependent Ca2+ channel in the RPE
(Rosenthal et al., 2006; Ueda and Steinberg, 1993;
Wollmann et al., 2006). Recently, it could be shown that
systemic application of the L-type channel blocker
nimodipine led to a reduction of the light-peak amplitude
in the rat direct-current electro-retinogram (DC-ERG)
(Rosenthal et al., 2006). Thus, these data indicate the
functional presence of L-type channels in the RPE of an
intact eye. Using Western blot techniques, the L-type a1
subunit was identified as the a1D or CaV1.3 subunit or the
neuroendocrine subtype of L-type channels (Rosenthal
et al., 2001, 2006; Strauss et al., 2000; Wollmann et al.,
2006). In the rat RPE cell line RPE-J, and in cultured
mouse RPE cells, this L-type channel appeared to be the
major subtype. The conclusion that CaV1.3 subunits
mainly form the L-type Ca2+ channel pore is supported
by the fact that rather high concentrations of dihydropyridine derivatives were required to decrease Ba2+ currents in
RPE cells (Rosenthal et al., 2006; Strauss et al., 1997). A
low sensitivity to dihydropyridines has been reported to be
characteristic for CaV1.3 when heterologousely expressed
(Koschak et al., 2001, 2003; Scholze et al., 2001). Finally,
RPE cells mainly express a splicing variant of CaV1.3
which has not been described thus far (Rosenthal et al.,
2006). It might be that properties of this splicing variant
could explain the differences between L-type currents in
RPE cells and those in other tissues or heterologeous
expression systems.
5.4.1.3. Role of voltage-dependent Ca2+ channels for RPE
function. The expression of CaV1.3 Ca2+ channel subunits, which are also termed as the neuroendocrine subtype
283
of L-type channels imply that these voltage-dependent
Ca2+ channels play a role in the regulation of secretion.
However, some questions have to be answered to conclude
that this might also be their function in RPE cells (Fig. 7).
The first question is whether high voltage-activated Ca2+
channels can play a functional role in retinal epithelial cells
with a resting membrane potential of 40 to 45 mV.
Investigation of CaV1.3-mediated currents in other cell
systems showed that they have the most negative activation
threshold and potential for half maximal activation among
all L-type channel subtypes (Koschak et al., 2001; Michna
et al., 2003; Scholze et al., 2001). Furthermore, in a study
using patch-clamp techniques in combination with measurements of intracellular free Ca2+ it could be shown that
L-type channels contribute to an increase in intracellular
free Ca2+at membrane potentials fixed to the resting
potential of RPE cells of about 40 mV (Mergler and
Strauss, 2002). This effect is achieved by activation of the
Ca2+/InsP3 second-messenger system. Intracellular application of InsP3 led to a phosphorylation-dependent shift in
the voltage-dependence of L-type channels to more
negative values, more close to the resting potential of
RPE cells (Mergler and Strauss, 2002). This increased the
number of open channels mediating a larger membrane
conductance for Ca2+ at the resting membrane potential.
The resulting influx of extracellular Ca2+ into the cell leads
to an increase in cytosolic free Ca2+. A comparable
mechanism was found for the bFGF-dependent stimulation of RPE cells (Rosenthal et al., 2001). Application of
bFGF leads to an increase in intracellular free Ca2+ by a
nifedipine-sensitive influx of Ca2+ into the cell through
L-type Ca2+ channels (Mergler et al., 1998). This effect
was not mediated by activation of the Ca2+/InsP3 secondmessenger system. Instead, the bFGF-dependent rise in
RPE
2+
Ca
st
s
ore
Melanosomes
Mit
och
on
dri
a
TRPC1
NMDA receptor
Glutamate
Ca2+
IP3
-Adrenaline
Ca2+
Ca2+
Gαq/11
-Growth factors
PLC
- ATP
TK
- AtCh
Ca2+
- Secretion
ATP
- Phagocytosis
Ca2+
- Transepithelial transport
P2X receptor
- Dark adaptation
CaV1.3
bFGF
FGFR2
Na+ Ca2+
Ca2+
PMCA
NCX1
Fig. 7. Schematic diagram summarizing different Ca2+-conducting ion channels and their impact on Ca2+ homoeostasis in RPE cells. The ion channels
are drawn in blue. The factors, which activate these ion channels are drawn in red. Connected second-messenger pathways are drawn in yellow (the box
summarises agonists stimulating these pathways). In light blue are membrane proteins, which are not ion channels but contribute to changes in the Ca2+
homoeostasis as well. Abbreviations are: ATP ¼ adenosine triphosphate; AtCh ¼ acetyl choline; bFGF ¼ basic fibroblast growth factor; CaV1.3 ¼ a1
Ca2+ channel subunit 1.3; Gaq/11 ¼ G protein a-subunit q/11; FGFR2 ¼ fibroblast growth factor receptor 2; IP3 ¼ inositol-1,4,5-trisphosphate;
NMDA ¼ N-methyl-D-aspartate; NCX-1 ¼ cardiac Na+/Ca2+ exchanger; PLC ¼ phospholipase; PMCA ¼ plasma membrane Ca2+-ATPase; P2X
receptor ¼ purinergic ionotropic receptor; TK ¼ tyrosine kinase; TRPC1 ¼ transient receptor potential channel C1.
ARTICLE IN PRESS
284
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
intracellular free Ca2+ results from the physical interaction
(Fig. 7) between the bFGF receptor FGFR2 and the
CaV1.3 subunits (Rosenthal et al., 2001). This interaction
leads to tyrosine phosphorylation of the Ca2+ channel and
a subsequent shift in the voltage-dependent activation
towards more negative potentials, again closer to the
resting potential of RPE cells. Also the resting activity of
L-type channels in the RPE is dependent on interaction
with a tyrosine kinase. L-type channels are constantly
activated by the cytosolic tyrosine kinase pp60csrc (Strauss
et al., 1997, 2000). This regulatory effect is based on a
physical interaction of the CaV1.3 subunits and pp60csrc
(Strauss et al., 2000). Among the serine/threonine kinases,
regulation of PKC seems to have the most important
impact on L-type channel activity in RPE cells. Based on
the application of various blockers it was concluded that it
is unlikely that protein kinase A or G have any effects on
L-type channel activity (Strauss et al., 1997). PKC has two
effects on L-type channel activity (Strauss et al., 1997). One
is the direct activation of this channel. With basic PKC
activity, pp60csrc and PKC have additive effects on the
L-type channel activity. However, a second function of
PKC emerges when PKC is further activated. With
stimulated PKC, not only the L-type channel activity
increases but also the effect of pp60csrc changes. Under
these conditions pp60csrc becomes an inhibitor of L-type
channel activity (Strauss et al., 1997). The underlying
mechanism is not clear. It is possible that different isoforms
of PKC are responsible for these two different effects of
L-type Ca2+ channel regulation in RPE cells.
In summary, L-type channels in the RPE contribute to
changes in intracellular free Ca2+ not by changes in the
membrane potential but by shifts of their voltagedependence which increases the number of open channels
at the resting potential of RPE cells. The next question is,
in which signalling pathways are L-type channels involved?
A major regulation by tyrosine kinase implies that L-type
channels in the RPE are involved in growth factordependent Ca2+ signalling because many growth factors
act via stimulation of tyrosine kinase-dependent pathways.
As mentioned above, this hypothesis has been proven for
bFGF (Rosenthal et al., 2001). Other growth factors such
as insuline-like growth factor-1 (IGF-1) are also very likely
candidates as an IGF-1-dependent regulation of L-type
channel activity has been shown in smooth muscle cells and
the RPE is known to express IGF-1 receptors (BenceHanulec et al., 2000; Rosenthal et al., 2004; Slomiany and
Rosenzweig, 2004a, b).
Another question is, what changes in cell function might
be achieved by increases in intracellular-free Ca2+mediated by activated L-type channels? One effect of
many growth factors is their influence on cell differentiation or cell function by changes in the gene expression
profile. These changes are regulated by increased expression of transcription factors (Fig. 7). In RPE cells the direct
opening of L-type channels by the dihydropyridine
compound BayK8644 resulted in an increased transcrip-
tion rate c-fos too (Rosenthal et al., 2005). Increased c-fos
expression is also induced by bFGF. However, the bFGFdependent expression of c-fos was independent from L-type
channel activity. Thus stimulation of L-type channels in the
RPE can change the expression of immediate early genes
which is a mechanism playing a role in the growth factordependent modulation of gene expression by other growth
factors than bFGF (Rosenthal et al., 2005).
As stated above, the subtype of L-type channels in the
RPE is the neuroendocrine subtype. This implies a role of
L-type channels in the regulation of secretion. The RPE is
known to secrete a variety of growth factors. Furthermore,
the secretion of growth factors by the RPE is regulated by
other growth factors (Campochiaro et al., 1994; Guillonneau et al., 1997; Hackett et al., 1997; Nagineni et al., 2003;
Ohno-Matsui et al., 2003; Rosenthal et al., 2004; Slomiany
and Rosenzweig, 2004a, b) which in some cases have been
shown to stimulate L-type channels via tyrosine kinasedependent phosphorylation. Thus the prominent role of
L-type channels in RPE cells is most likely the regulation of
growth factor secretion. Indeed, bFGF-induced secretion
of VEGF was reduced to the basic secretion rate observed
in the absence of bFGF after inhibition of L-type channels
(Rosenthal et al., 2005).
As stated above, L-type channels seem to participate in
the generation of the light-peak in the DC-ERG
(Rosenthal et al., 2006). The light-peak is a signal in the
human electro-oculogram (EOG, equivalent to the DC-ERG)
which is thought to derive from activation of Cl channels
in the basolateral membrane of the RPE (Gallemore et al.,
1988; Gallemore and Steinberg, 1989a, 1993). In a recent
study, it was shown that bestrophin-1, proposed to act as a
Ca2+-dependent Cl channel (Qu et al., 2004; Qu and
Hartzell, 2004; Sun et al., 2002), additionally regulates
L-type Ca2+ channels in the RPE (Rosenthal et al., 2006).
Furthermore, mutant bestrophins showed different effects
on L-type channel properties. The possible interaction of
the L-type channel with bestrophin-1, in other words
interaction of a Ca2+ channel and a Ca2+-dependent Cl
channel, would put the activation of the Ca2+-dependent
Cl channel into a close feedback mechanism thereby
influencing transepithelial Cl and fluid transport.
In summary, L-type channels can contribute to changes
in intracellular free Ca2+ at the resting potential of RPE
cells. The L-type channel-dependent changes in cytosolic
free Ca2+ participate in the regulation of growth factor
secretion and likely in the regulation of epithelial transport
of Cl and water. An influence on gene expression enables
an adaptation of transcriptional activity to the underlying
functional changes. To achieve this regulatory function
L-type channels mainly underlie the control by PKC
and tyrosine kinase activity.
5.4.2. TRP channels
5.4.2.1. TRP channels in general. TRP channels represent
a large family of Ca2+-conducting cation channels (Clapham et al., 2003; Inoue, 2005; Ramsey et al., 2005). The
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
name TRP derived from the Drosophila homologue TRP.
TRP channels are represented by six families with
individual members who can contribute to a large variety
of signalling pathways or sensory function. The family of
TRPC (classic) and of the TRPM (metastatin like)
channels are involved in intracellular signalling cascades
due to their activation by G protein subunits, metabolites
of second-messenger cascades, or by depletion of cytosolic
Ca2+ stores. TRPV (vanilloid receptor) and TRPA (with
ankryn repeats) channels play a role in sensory mechanisms
because they can be activated by a variety of physical
stimuli such as temperature or pH. One family represents
channels in intracellular organelles (TRPML) and the
function of the channels in other families is still unknown.
5.4.2.2. TRP channels in the RPE and their role in RPE
function. Early patch-clamp studies on rat RPE cells
indicated a possible presence of TRP channels (Poyer et al.,
1996). Poyer et al. (1996) described G protein-activated
cation channels, which were selective for many different
monovalent cations. Thus, it could be that the authors
described currents mediated by TRPC channels. However,
the cation currents described in the study by Poyer et al.
seemed not to be carried by Ca2+ and it seemed that Gaisubunits or at least pertussis toxin-sensitive G-proteins and
not Gaq/11 subunits were responsible for their activation.
These are not characteristic properties for TRPC channels.
Nevertheless, in a recent publication, the expression of
TRPC1 channels was detected in the human RPE cell line
ARPE-19 (Bollimuntha et al., 2005). As mentioned above,
TRPC channels are integrated into the InsP3/Ca2+ second
messenger pathway (Fig. 7) either by their function as
store-operated Ca2+ channels (SOCs) or by their activation
by Gaq/11 subunits (Clapham et al., 2003; Inoue, 2005;
Ramsey et al., 2005). In a study with combined measurements of membrane currents and changes in intracellular
free Ca2+ it could be shown that together with L-type
Ca2+ channels another type of Ca2+-conducting ion
channel contributes to InsP3-induced increases in intracellular free Ca2+ (Mergler and Strauss, 2002). This study
used a standard protocol for induction of capacitative
Ca2+ influx as a standard system to explore activation of
store-operated Ca2+ influx. Thereby, a component of
Ca2+ conductance insensitive to blockers for L-type
channels was detected. It might be that TRPC channels
underlie this additional InsP3-induced membrane conductance for Ca2+. In the RPE, TRPC channels could play a
role in a couple of different agonist-controlled Ca2+dependent signalling pathways. The RPE expresses P2Y
(Peterson et al., 1997; Reigada et al., 2005; Reigada and
Mitchell, 2005; Ryan et al., 1999; Sullivan et al., 1997),
muscarinic (Feldman et al., 1991; Friedman et al., 1988;
Gonzalez et al., 2004) and adrenergic receptors (Edelman
and Miller, 1991; Feldman et al., 1991; Joseph and Miller,
1992; Quinn and Miller, 1992; Quinn et al., 2001; Rymer
et al., 2001). These receptors are known to act via
stimulation of Gaq/11 proteins (Wettschureck and Offer-
285
manns, 2005) and to stimulate an increase in intracellular
free Ca2+ as second messenger.
Since TRPC channels can be activated by Gaq/11
proteins these ion channels might play a role in the
regulation of RPE function which are modulated by these
receptors: adrenergic or purinergic regulation of transepithelial ion transport and muscarinergic or purinergic
regulation of phagocytosis. Although the involvement of
TRPC channels in these regulatory pathways is not proven,
the presence of these channels might further help to
identify so far unknown membrane conductances for
Ca2+ which contribute to the underlying Ca2+ signalling.
Another feature of TRPC1 channels in RPE cells is
that they are interacting with beta-tubulin filaments of
the cytoskeleton (Bollimuntha et al., 2005). This interaction enables a regulation of temporal and spatial distribution of TRPC1 channels in the cell membrane. The
differentially regulated presence in the cell membrane
modulates the effectiveness of an agonist to influence
RPE cell function.
5.4.3. ATP receptors
5.4.3.1. ATP receptors in general. ATP can be released by
different cell types and can act as an autocrine or paracrine
extracellular messenger (Burnstock, 2004; Burnstock and
Knight, 2004). ATP can bind to two different receptor
types. These are the P2Y and P2X receptor families.
Whereas the P2Y receptors are coupled to activation of G
proteins (mainly Gaq/11 or Gai/O subunits) (Burnstock,
2004; Burnstock and Knight, 2004) and initiate an
intracellular second messenger cascade, the P2X receptors
are ligand-activated ion channels (North, 2002). ATP
binding to P2X receptors results in the activation of a
cation channel, which can conduct Na+ as well as Ca2+.
With this mechanism, ATP can directly contribute to
increases in intracellular free Ca2+ via stimulation of an
influx of extracellular Ca2+ into the cell. The P2X
receptors represent a gene family of 7 members which
can form homomeric or heteromeric ATP-activated ion
channels (North, 2002). Currents through P2X receptor
channels show fast inactivation and desensitization to
ATP. P2X receptors are expressed in a variety of tissues
and have been described to modulate synaptic currents,
contractility, and secretion.
5.4.3.2. P2X receptors in the RPE and their role for RPE
function. So far the expression of P2X receptors in the
RPE has not been shown by molecular biological or
protein biochemical methods. However, a study with rat
RPE cells on ATP-dependent signalling revealed changes in
the membrane conductance which indicate the functional
presence of P2X receptors (Ryan et al., 1999). Extracellular
application of ATP resulted in the depolarization of RPE
cells due to activation of cation channels (Fig. 7). This
cation conductance shared some properties known of P2X
receptors in heterologeous expression studies (North and
Surprenant, 2000).
ARTICLE IN PRESS
286
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
In the RPE, ATP-dependent signalling was found to play
a role in the regulation of transepithelial ion and water
transport, and phagocytosis (Collison et al., 2005; Peterson
et al., 1997; Quinn and Miller, 1992; Reigada et al., 2005;
Reigada and Mitchell, 2005; Ryan et al., 1999; Sullivan et
al., 1997). The main source for ATP seems to be the RPE
itself which can release ATP in response to a variety of
stimuli such as bFGF or hypotonic challenge (Mitchell,
2001; Reigada et al., 2005; Reigada and Mitchell, 2005).
ATP that functions as an autocrine messenger can do so by
binding to different purinergic receptors such as P2Y, P2X,
or adenosine receptors after degradation by ectonucleases
(Mitchell, 2001; Reigada et al., 2005; Reigada and
Mitchell, 2005). P2Y receptor stimulation leads to an
increased transepithelial transport of Cl and water
(Peterson et al., 1997). The responsible receptor-mediated
signalling promotes these effects in two ways. One is an
increase in intracellular free Ca2+ and the subsequent
activation of ion channels such as Ca2+-dependent Cl
channels which directly provide a transportation pathway
for ions (Peterson et al., 1997). So far, the ATP-induced
increases in intracellular-free Ca2+ were found to arise
mainly from release of Ca2+ from cytosolic Ca2+ stores
indicating that these are mediated by activation of P2Y
receptors (Peterson et al., 1997; Ryan et al., 1999). In
addition, it was found that ATP-dependent increases in
intracellular free Ca2+ activate Ca2+-dependent K+
channels leading to hyperpolarization of the RPE cell
(Ryan et al., 1999). This could be another mechanism
by which ATP modulates transepithelial ion transport:
modulation of the driving forces for ions to move across
membranes. In this case, the activation of K+ channels
would increase the driving force for Cl to leave the cell.
For the modulation of these driving forces the P2Y and
P2X receptors can interact. This was found in a study in
which the effects of ATP on the membrane conductance
were investigated (Ryan et al., 1999). Here, application of
ATP first resulted in a cell depolarization, which was
followed by cell hyperpolarization. The first event was
found to be dependent on the stimulation of P2X receptors
whereas the later event resulted from activation of Ca2+dependent BK Ca2+ channels (see Section 3).
5.4.4. Glutamate receptors
5.4.4.1. Glutamate receptors in general. Like ATP, the
neurotransmitter glutamate can bind to either ligand-gated
ion channels, the so called ionotropic glutamate receptors
(Kew and Kemp, 2005; Mayer, 2005), or to receptors which
activate G-protein-coupled intracellular second-messenger
cascades, the metabotropic glutamate receptors (Conn,
2003; Kew and Kemp, 2005).
5.4.4.2. Glutamate receptors in the RPE. In independent
studies, the functional expression of glutamate receptors
has been described in the RPE (Fig. 7). The RPE expresses
both metabotropic as well ionotropic glutamate receptors
(Feldman et al., 1991; Fragoso and Lopez-Colome, 1999;
Lopez-Colome et al., 1993, 1994; Uchida et al., 1998).
Stimulation of either metabotropic or ionotropic glutamate
receptors in the RPE result in an increase in the cytosolicfree Ca2+ (Feldman et al., 1991; Fragoso and LopezColome, 1999). The stimulation of metabotropic receptors
leads to an increase in intracellular free Ca2+ via
stimulation of a phospholipase C-dependent intracellular
second messenger cascade. Interestingly, this increase was
reduced after application of the L-type channel blocker
nifedipine (Fragoso and Lopez-Colome, 1999). This might
be enabled by the InsP3-dependent stimulation of L-type
Ca2+ channels, which has been described above. However,
glutamate-dependent increases in intracellular-free Ca2+
were also reduced in the presence of the ionotropic NMDA
receptor (N-methyl-D-aspartate receptor) antagonists. Thus
glutamate also activates NMDA receptors in RPE cells.
Because of the expression of two different receptor
subtypes of glutamate receptors, glutamate initiates a
complex pattern of intracellular Ca2+ signalling in RPE
cells. However, the effects of this Ca2+ signalling for RPE
function are not entirely clear. Recently, it could be shown
that stimulation of NMDA receptors in the RPE activates
the release of ATP from RPE cells (Reigada et al., 2006). In
the dark, photoreceptors release glutamate at their synaptic
terminals. It is likely that part of this glutamate reaches
RPE cells and leads to an adaptation of the RPE function
to the requirements of photoreceptor function in the dark.
One of these functions might be the regulation of the
phagocytic activity of RPE cells which is controlled by
changes in the illumination of the retina (Greenberger and
Besharse, 1985; Lopez-Colome et al., 1994).
6. Na+
In addition to its role as charge carrier in excitable cells
where a Na+ influx is responsible for the depolarization of
the plasma membrane, Na+ plays a central role in many
transport processes of diverse organic molecules like amino
acids, sugars, and neurotransmitters where the steep Na+
gradient is used for secondary active transport of these
molecules. Therefore, the Na+ homeostasis of a cell is of
great importance for its normal function.
Two main families of Na+ channels exist: the voltagegated Na+ channels (Nav1.1–Nav1.9) and the epithelial
Na+ channels (ENaCa, b, g and d). These two families
have very different functions. While voltage-gated channels
are found almost exclusively in excitable cells where they
are responsible for cell depolarization, ENaC are involved
in Na+ homeostasis in epithelial cells.
6.1. Na+ ions in RPE cell function
Over the apical membrane of the RPE cells a Na+
gradient is established by the Na+/K+-ATPase. Besides
the contribution to the negative membrane potential, this
gradient is used for the transport of various molecules from
the subretinal space into the cells. These molecules include
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
+
K+ and Cl, HCO
3 , H , lactate, GABA and taurine.
These transport mechanisms are involved in volume
regulation of the subretinal space, pH regulation, and the
removal of metabolic products and neurotransmitters. For
these tasks, Na+ is permanently secreted by the RPE into
the subretinal space. While it is well known that Na+
leaves the cells through the apical membrane by the Na+/
K+ ATPase activity, it is still a subject of speculation how
the Na+ enters the cells through the basolateral membrane.
The steep chemical gradient between the extracellular
space and the cytosol provides ideal conditions for
Na+-conducting ion channels to perform Na+ influx
through the basolateral membrane of the RPE.
6.2. Na+ channels of the RPE
In cultured RPE cells from rat, newt, and humans,
voltage-gated Na+ channels have been observe in patchclamp investigations (Botchkin and Matthews, 1994; Sakai
and Saito, 1997; Wen et al., 1994). Nevertheless, it has also
been shown that these Na+ channels appear only in RPE
cells when they are cultured for at least 24 h. Thus, there
should be other Na+-conducting proteins in the basolateral membrane. By immunohistochemistry and molecular
biological methods, different members of the other family
of Na+ channels (ENaC) have been detected in the RPE
(Dyka et al., 2005; Golestaneh et al., 2002; Mirshahi et al.,
1999). Though, the physiological data providing evidence
for their expression in RPE cells are still missing, their
contribution to the transepithelial transport seems very
likely since such a coordinated transport activity between
the Na+/K+ ATPase and ENaC localized in opposite
membranes in Na+ transport is well known from other
epithelial cell types e.g. in the collecting duct in the kidney.
Because of the missing electrophysiological data we will
not go further into detail regarding the Na+ channels in
the RPE.
7. Ion channels and RPE disease
Ion channel research improved the understanding of
many diseases in two ways.
On the one hand, analysis of ion channel function
resulted in a better understanding of the physiology of cell
function in general (Jurkat-Rott and Lehmann-Horn,
2004). This led also to new insights into the pathophysiology of diseases and, thus, into the development of
new treatments.
Together with genetic analyses of genes coding for ion
channels the term of channelopathies emerged (Celesia,
2001; Hubner and Jentsch, 2002; Jentsch et al., 2004;
Lehmann-Horn and Jurkat-Rott, 1999; Striessnig et al.,
2004). Channelopathies are disorders, which result from
malfunction of ion channels. These can be due to gain of
function, loss of function, or by dominant-negative effects
leading, for example, to a decreased number of channels in
the cell membrane.
287
7.1. Change or loss of ion channel function leading to RPE
diseases
Mainly changes in Cl channel function were found to
lead to degeneration of the retina.
Although not a channelopathy, the inactivation of the
ClC-2 channel gene in mouse results in a phenotype very
similar to the clinical picture of retinitis pigmentosa in man
(Bosl et al., 2001). A comparable cause for retinitis
pigmentosa was not found in the human disease, important
aspects of RPE function can be discussed from this mouse
model. Analysis of transepithelial transport properties of
the ClC-2 knock-out mouse revealed that the RPE in these
mice show no transepithelial potential (Bosl et al., 2001)
indicating the absence of transepithelial transport of Cl
and water. The first conclusion, which can be drawn from
this is that ClC-2 channels might provide the most
important efflux pathway for Cl across the basolateral
membrane according to the model of transepithelial
transport which has been described in the chapters above.
The second conclusion, which can be drawn from the
observations in the ClC-2 / mouse is the importance of
the transepithelial transport of water and Cl from the
subretinal space to the blood side for the maintenance of
photoreceptor function and survival. The absence of the
transport leads to the degeneration of photoreceptors.
More important for a human disease are the effects of
mutations in the gene coding for bestrophin-1, the VMD2
gene (Marquardt et al., 1998; Petrukhin et al., 1998).
Mutations in the VMD2 gene cause an inherited form of
macular degeneration with juvenile onset, Best’s vitelliform
macular dystrophy (Best, 1905; Cross and Bard, 1974;
Godel et al., 1986; Weingeist et al., 1982). It is discussed
that the mutations in the VMD2 gene lead to a changed
basolateral membrane conductance for Cl to explain an
important symptom for the diagnosis of Best’s disease.
Thus Best’s macular dystrophy might be understood as a
channelopathy. This diagnostic symptom is the reduction
of the light-rise or light-peak in the patient’s electrooculogram of the Best patients (Cross and Bard, 1974). An
analysis of the chick DC-ERG revealed that an increase in
the basolateral membrane conductance for Cl with
subsequent depolarization of the basolateral membrane
represents the underlying mechanism contributing to the
light-rise (Gallemore et al., 1988, 2004; Gallemore and
Steinberg, 1989b). It is believed that a light-peak substance
is released from the retina after illumination (Gallemore
et al., 1988, 2004; Gallemore and Steinberg, 1989b). This
substance diffuses to the RPE, binds to a receptor at
the apical membrane, and initiates an intracellular second
messenger cascade, which in turn most likely results in
the activation of a Ca2+-dependent Cl channel in the
basolateral membrane of the RPE. Thus, mutations in the
VMD2 gene lead to a decrease in the basolateral membrane
conductance for Cl in the RPE. Following this line
of reasoning it became a very attractive hypothesis
that the VMD2 gene product itself might function as
ARTICLE IN PRESS
288
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
a Ca2+-dependent Cl channel and that the mutations lead
to a loss in the Cl channel function. Indeed, the VMD2
gene product bestrophin-1 appears to function as
Ca2+-dependent Cl channel (see Section 4) (Sun et al.,
2002). Furthermore, 15 different mutant bestrophins were
investigated so far and it seems that all mutations lead to a
loss in Cl channel function with a dominant negative
effect on the wild-type allele. However, the hypothesis that
a loss of the Cl channel function of bestrophin causes the
leading symptom and the retinal degeneration in Best’s
disease is not supported by recent studies of the clinical
picture of patients with VMD2 mutations and by a study
investigating light-peak increases in bestrophin-1 /
mice. Patients carrying mutations in the VMD2 gene can
have a macular degeneration but normal electro-oculograms (Kramer et al., 2000; Pollack et al., 2005; Renner
et al., 2005; Wabbels et al., 2004, 2006). Or in other cases
the changes in the patient’s light-peak appeared secondary
to the onset of the macular degeneration (Wabbels et al.,
2004). Thus not all mutations, which lead to macular
degeneration seem to result in a loss of Cl channel
function. This impression is supported by an investigation
of a bestrophin-1 knockout mouse model. In these mice,
the absence of bestrophin-1 did not reduce the light-peak
amplitude in the DC-ERG (Marmorstein et al., 2006).
Furthermore, at low light-intensities, the bestrophin-1 /
mice showed increased light-peak amplitudes compared to
the wild-type mice. Thus, either the lack of the Cl channel
function in the RPE could be compensated for by another
Cl channel, which could also be the case in the human
RPE and would have comparable changes in the human
electro-oculogram. Or bestrophin-1 might have additional
functions to the Cl channel function involved in generation of the light peak and in the aetiology of the human
macular degeneration, if mutations have changed this
function. This additional function might be an influence of
bestrophin-1 on the Ca2+ homeostasis of the RPE as it has
been discussed in the chapter about Ca2+ channels of the
RPE (Rosenthal et al., 2006).
Another disease which might have an impact on RPE
function is cystic fibrosis (Blaug et al., 2003). The gene
product of the disease-causing gene, CFTR, is also
expressed in the RPE (Blaug et al., 2003; Reigada and
Mitchell, 2005; Weng et al., 2002; Wills et al., 2000). CFTR
is known to function as Cl channel (Jentsch et al., 2002).
Although cystic fibrosis patients show a decreased signal in
the electro-oculogram which is also discussed to be
associated with an activation of Cl channels in the RPE
(Blaug et al., 2003), the loss of CFTR Cl channel function
in patients with cystic fibrosis seems not to have any
degenerative effects on the macula (Blaug et al., 2003).
In summary, loss of ion channel function in the RPE can
have severe effects on the retina and lead to retinal
degeneration. Such a mechanism is discussed to be the
cause for an inherited form of macular degeneration, Best
vitelliform macular dystrophy. However, such a mechanism has not to been proven to be the case in Best disease.
7.2. Involvement of ion channels in signalling cascades
leading to degenerative processes in the retina
Normal ion channel function in the RPE can play a role
in the aetiology of degenerative diseases of the retina. If an
ion channel has an important contribution to a signal
pathway leading to the initiation of processes which mark
important steps in a chain of events leading to pathologic
changes in cell function then this ion channel may play a
key role in the aetiology of a disease. This may be the case
for L-type Ca2+ channels. As mentioned above, the RPE is
able to secrete a variety of growth factors in health and in
disease. Several lines of evidence indicate that growth
factors secreted by the RPE promote the development of
choroidal neovascularization in age-related macular degeneration (Amin et al., 1994; Frank, 1997; Holz et al., 2004;
Lambooij et al., 2003; Oh et al., 1999; Rosenthal et al.,
2004), the most common cause for blindness in industrialized countries. In this disease the complication of choroidal
neovascularization accounts for the major component of
vision loss (Ambati et al., 2003). In the chain of events
which lead to choroidal neovascularization the RPE seem
to play a central role by its secretion of the major
angiogenic factor, VEGF (Amin et al., 1994; Frank,
1997; Krzystolik et al., 2002; Lambooij et al., 2003; Lopez
et al., 1996; Rosenthal et al., 2004). The secretion of VEGF
by the RPE is under control of other growth factors which
might be secreted by photoreceptors or by the RPE itself
(Frank, 1997; Kondo et al., 2003; Mousa et al., 1999;
Nagineni et al., 2003; Rosenthal et al., 2004, 2005;
Slomiany and Rosenzweig, 2004a, b; Witmer et al., 2003).
Thus, the secretion of growth factors by the RPE has
several effects in the initiation of choroidal neovascularization. These are autocrine and paracrine stimulation. As
mentioned above, the L-type Ca2+ channels in the RPE
regulate secretion of VEGF. Due to regulation of L-type
channels by other growth factors or cytosolic tyrosine
kinase, which are stimulated by other growth factors,
L-type Ca2+ channels are also able to fulfil the autocrine
stimulation pathway of VEGF secretion. Thus, with this
important function, L-type channels might play a key role
in the initiation of choroidal neovascularization in agerelated macular degeneration and could be a target for the
development of strategies to prevent this severe complication (Strauss et al., 2003). This hypothesis is supported by
the observation that freshly isolated RPE cells from
patients with age-related macular degeneration and choroidal neovascularization still show Ba2+ currents with
properties of L-type Ca2+ channels (Strauss et al., 2003). A
sensitive reaction of L-type channels to degenerative
processes in the retina was detected in studies of L-type
channel function in RPE cells of an animal model for
human retinitis pigmentosa, the Royal College of Surgeons
(RCS) rat. Here, the L-type Ca2+ channels showed an
increased activity due to a changed regulation (Mergler
et al., 1998). Although the consequences of these effects
have not been further explored these observations show
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
that L-type channel dependent Ca2+-signalling is sensitively adapted to the changed patho-physiologic situation
in the degenerative process of the retina.
8. Summary
The patch-clamp technique provides a strong tool in the
identification and characterization of ion fluxes across
membranes. In the RPE it has been used to support the
data previously obtained by other techniques like molecular
biological, protein biochemical, and other electrophysiological studies with Ussing chambers. The patch-clamp
studies not only confirmed these data but also extended our
knowledge on ionic currents across the RPE membranes.
This in turn led us to the development of new ideas of how
ion channels may be involved in RPE cell functions.
So, it could be confirmed that a K+ conductance
through inwardly rectifying K+ channels contributes to
the K+ homoeostasis in the subretinal space. Considering
the additional expression of voltage-gated and Ca2+activated K+ channels, the picture of K+ transport by
the RPE could be extended.
Comparably, the proposed Cl conductance in the
basolateral membrane of RPE cells was confirmed by
patch-clamp studies. This technique proves to be important
for the identification of the function of a macular disease
associated protein, bestrophin. Based on patch-clamp data,
bestrophin is now thought to be a Cl channel and/or a
modulator of voltage-gated Ca2+ channels. These Ca2+
channels have been shown by the patch-clamp technique to
be involved in RPE cell functions like the secretion of
growth factors and in transcellular transport. However, the
investigation of ion channels in the RPE is not finished and
a closer investigation of bestrophin function, glutamate
receptors, P2X receptors, and TRPC channels will further
understanding and open new aspects to the role of the RPE
in visual function.
Acknowledgements
The authors thank Tyson Kinnick for the help, for
scientific input and stimulating fruitful discussions. The
work was supported by the Deutsche Forschnugsgemeinschaft grant DFG STR480/8-2 and DFG STR480/9-1.
References
Accardi, A., Miller, C., 2004. Secondary active transport mediated by a
prokaryotic homologue of ClC Cl channels. Nature 427 (6977),
803–807.
Acharya, S., Rodriguez, I.R., Moreira, E.F., Midura, R.J., Misono, K.,
Todres, E., Hollyfield, J.G., 1998. SPACR, a novel interphotoreceptor
matrix glycoprotein in human retina that interacts with hyaluronan.
J. Biol. Chem. 273 (47), 31599–31606.
Ackerman, M.J., Clapham, D.E., 1997. Ion channels—basic science and
clinical disease. N. Engl. J. Med. 336 (22), 1575–1586.
Adler, A.J., Southwick, R.E., 1992. Distribution of glucose and lactate in
the interphotoreceptor matrix. Ophthalmic Res. 24 (4), 243–252.
289
Adorante, J.S., 1995. Regulatory volume decrease in frog retinal pigment
epithelium. Am. J. Physiol. 268 (1 Pt 1), C89–C100.
Adorante, J.S., Edelman, J.L., 1997. The role of Cl channels in volume
regulation in bovine pigmented epithelial cells. Am. J. Physiol. 273
(4 Pt 1), C1435–C1437.
Adorante, J.S., Miller, S.S., 1990. Potassium-dependent volume regulation
in retinal pigment epithelium is mediated by Na,K,Cl cotransport.
J. Gen. Physiol. 96 (6), 1153–1176.
Aguilar-Bryan, L., Nichols, C.G., Wechsler, S.W., Clement, J.P.t., Boyd
III, A.E., Gonzalez, G., Herrera-Sosa, H., Nguy, K., Bryan, J.,
Nelson, D.A., 1995. Cloning of the beta cell high-affinity sulfonylurea
receptor: a regulator of insulin secretion. Science 268 (5209), 423–426.
Ambati, J., Ambati, B.K., Yoo, S.H., Ianchulev, S., Adamis, A.P., 2003.
Age-related macular degeneration: etiology, pathogenesis, and therapeutic strategies. Surv. Ophthalmol. 48 (3), 257–293.
Amin, R., Puklin, J.E., Frank, R.N., 1994. Growth factor localization in
choroidal neovascular membranes of age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 35 (8), 3178–3188.
Ammar, D.A., Hughes, B.A., Thompson, D.A., 1998. Neuropeptide Y
and the retinal pigment epithelium: receptor subtypes, signaling, and
bioelectrical responses. Invest. Ophthalmol. Vis. Sci. 39 (10),
1870–1878.
Anderson, M.P., Gregory, R.J., Thompson, S., Souza, D.W., Paul, S.,
Mulligan, R.C., Smith, A.E., Welsh, M.J., 1991. Demonstration that
CFTR is a chloride channel by alteration of its anion selectivity.
Science 253 (5016), 202–205.
Arshavsky, V., 2002. Like night and day: rods and cones have different
pigment regeneration pathways. Neuron 36 (1), 1–3.
Ashcroft, F.M., Gribble, F.M., 1999. ATP-sensitive K+ channels and
insulin secretion: their role in health and disease. Diabetologia 42 (8),
903–919.
Baehr, W., Wu, S.M., Bird, A.C., Palczewski, K., 2003. The retinoid cycle
and retina disease. Vis. Res. 43, 2957–2958.
Bakall, B., Marknell, T., Ingvast, S., Koisti, M.J., Sandgren, O., Li, W.,
Bergen, A.A., Andreasson, S., Rosenberg, T., Petrukhin, K., Wadelius,
C., 1999. The mutation spectrum of the bestrophin protein—
functional implications. Hum. Genet. 104 (5), 383–389.
Bakall, B., Marmorstein, L.Y., Hoppe, G., Peachey, N.S., Wadelius, C.,
Marmorstein, A.D., 2003. Expression and localization of bestrophin
during normal mouse development. Invest. Ophthalmol. Vis. Sci. 44
(8), 3622–3628.
Ban, Y., Rizzolo, L.J., 2000a. Differential regulation of tight junction
permeability during development of the retinal pigment epithelium.
Am. J. Physiol. Cell Physiol. 279 (3), C744–C750.
Ban, Y., Rizzolo, L.J., 2000b. Regulation of glucose transporters during
development of the retinal pigment epithelium. Dev. Brain Res. 121
(1), 89–95.
Barg, S., 2003. Mechanisms of exocytosis in insulin-secreting B-cells and
glucagon-secreting A-cells. Pharmacol. Toxicol. 92 (1), 3–13.
Barrett, T., Suzek, T.O., Troup, D.B., Wilhite, S.E., Ngau, W.C., Ledoux,
P., Rudnev, D., Lash, A.E., Fujibuchi, W., Edgar, R., 2005. NCBI
GEO: mining millions of expression profiles—database and tools.
Nucleic Acids Res. 33 (Database issue), D562–D566.
Bauer, C.K., Schafer, R., Schiemann, D., Reid, G., Hanganu, I., Schwarz,
J.R., 1999. A functional role of the erg-like inward-rectifying K+
current in prolactin secretion from rat lactotrophs. Mol. Cell
Endocrinol. 148 (1 and 2), 37–45.
Baylor, D., 1996. How photons start vision. Proc. Natl. Acad. Sci. USA 93
(2), 560–565.
Beatty, S., Koh, H., Phil, M., Henson, D., Boulton, M., 2000. The role of
oxidative stress in the pathogenesis of age-related macular degeneration. Surv. Ophthalmol. 45 (2), 115–134.
Becerra, S.P., Fariss, R.N., Wu, Y.Q., Montuenga, L.M., Wong, P.,
Pfeffer, B.A., 2004. Pigment epithelium-derived factor in the monkey
retinal pigment epithelium and interphotoreceptor matrix: apical
secretion and distribution. Exp. Eye Res. 78 (2), 223–234.
Begenisich, T., Melvin, J.E., 1998. Regulation of chloride channels in
secretory epithelia. J. Membr. Biol. 163 (2), 77–85.
ARTICLE IN PRESS
290
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Begenisich, T., Nakamoto, T., Ovitt, C.E., Nehrke, K., Brugnara, C.,
Alper, S.L., Melvin, J.E., 2004. Physiological roles of the intermediate
conductance, Ca2+-activated potassium channel Kcnn4. J. Biol.
Chem. 279 (46), 47681–47687.
Behn, D., Doke, A., Racine, J., Casanova, C., Chemtob, S., Lachapelle,
P., 2003. Dark adaptation is faster in pigmented than albino rats. Doc.
Ophthalmol. 106 (2), 153–159.
Bellhorn, M.B., Lewis, R.K., 1976. Localization of ions in retina by
secondary ion mass spectrometry. Exp. Eye Res. 22 (5), 505–518.
Bello, R.A., Magleby, K.L., 1998. Time-irreversible subconductance
gating associated with Ba2+ block of large conductance
Ca2+-activated K+ channels. J. Gen. Physiol. 111 (2), 343–362.
Bence-Hanulec, K.K., Marshall, J., Blair, L.A., 2000. Potentiation of
neuronal L calcium channels by IGF-1 requires phosphorylation of the
alpha1 subunit on a specific tyrosine residue. Neuron 27 (1), 121–131.
Berridge, M.J., 2005. Unlocking the secrets of cell signaling. Annu. Rev.
Physiol. 67, 1–21.
Berridge, M.J., Lipp, P., Bootman, M.D., 2000. The versatility and
universality of calcium signalling. Nat. Rev. Mol. Cell Biol. 1 (1),
11–21.
Berridge, M.J., Bootman, M.D., Roderick, H.L., 2003. Calcium signalling: dynamics, homeostasis and remodelling. Nat. Rev. Mol. Cell Biol.
4 (7), 517–529.
Besharse, J.C., Defoe, D., 1998. Role of the retinal pigment epithelium in
photoreceptor membrane turnover. In: Marmor, M.F., Wolfensberger,
T.J. (Eds.), The Reitnal Pigment Epithelium. Oxford University Press,
Oxford.
Best, F., 1905. Über eine hereditäre Maculaaffection: Beiträge zur
Vererbungslehre. Z Augenheilkd 13, 199–212.
Bialek, S., Miller, S.S., 1994. K+ and Cl transport mechanisms in bovine
pigment epithelium that could modulate subretinal space volume and
composition. J. Physiol. 475 (3), 401–417.
Bialek, S., Joseph, D.P., Miller, S.S., 1995. The delayed basolateral
membrane hyperpolarization of the bovine retinal pigment epithelium:
mechanism of generation. J. Physiol. 484 (Pt 1), 53–67.
Bialek, S., Quong, J.N., Yu, K., Miller, S.S., 1996. Nonsteroidal antiinflammatory drugs alter chloride and fluid transport in bovine retinal
pigment epithelium. Am. J. Physiol. 270 (4 Pt 1), C1175–C1189.
Bichet, D., Haass, F.A., Jan, L.Y., 2003. Merging functional studies with
structures of inward-rectifier K(+) channels. Nat. Rev. Neurosci.
4 (12), 957–967.
Blaauwgeers, H.G., Holtkamp, G.M., Rutten, H., Witmer, A.N.,
Koolwijk, P., Partanen, T.A., Alitalo, K., Kroon, M.E., Kijlstra, A.,
van Hinsbergh, V.W., Schlingemann, R.O., 1999. Polarized vascular
endothelial growth factor secretion by human retinal pigment
epithelium and localization of vascular endothelial growth factor
receptors on the inner choriocapillaris. Evidence for a trophic
paracrine relation. Am. J. Pathol. 155 (2), 421–428.
Blaug, S., Quinn, R., Quong, J., Jalickee, S., Miller, S.S., 2003. Retinal
pigment epithelial function: a role for CFTR? Doc. Ophthalmol. 106
(1), 43–50.
Bok, D., 1993. The retinal pigment epithelium: a versatile partner in
vision. J. Cell Sci. Suppl. 17, 189–195.
Bok, D., Hall, M.O., 1971. The role of the pigment epithelium in the
etiology of inherited retinal dystrophy in the rat. J. Cell Biol. 49 (3),
664–682.
Bollimuntha, S., Cornatzer, E., Singh, B.B., 2005. Plasma membrane
localization and function of TRPC1 is dependent on its interaction
with beta-tubulin in retinal epithelium cells. Vis. Neurosci. 22 (2),
163–170.
Bond, C.T., Pessia, M., Xia, X.M., Lagrutta, A., Kavanaugh, M.P.,
Adelman, J.P., 1994. Cloning and expression of a family of inward
rectifier potassium channels. Receptors Channels 2 (3), 183–191.
Bond, C.T., Maylie, J., Adelman, J.P., 1999. Small-conductance calciumactivated potassium channels. Ann. NY Acad. Sci. 868, 370–378.
Borgula, G.A., Karwoski, C.J., Steinberg, R.H., 1989. Lightevoked changes in extracellular pH in frog retina. Vis. Res. 29 (9),
1069–1077.
Bosl, M.R., Stein, V., Hubner, C., Zdebik, A.A., Jordt, S.E., Mukhopadhyay, A.K., Davidoff, M.S., Holstein, A.F., Jentsch, T.J., 2001.
Male germ cells and photoreceptors, both dependent on close cell-cell
interactions, degenerate upon ClC-2 Cl() channel disruption. EMBO
J. 20 (6), 1289–1299.
Botchkin, L.M., Matthews, G., 1993. Chloride current activated by
swelling in retinal pigment epithelium cells. Am. J. Physiol. 265
(4 Pt 1), C1037–C1045.
Botchkin, L.M., Matthews, G., 1994. Voltage-dependent sodium channels
develop in rat retinal pigment epithelium cells in culture. Proc. Natl.
Acad. Sci. USA 91 (10), 4564–4568.
Boulton, M., 1991. Ageing of the retinal pigment epithelium. In: Osborne,
N.N., Chader, G.J. (Eds.), Progress in Retinal Research. Pergamon
Press, Oxford, NY, pp. 125–151.
Boulton, M., 1998. The role of melanin in the RPE. In: Marmor, M.F.,
Wolfensberger, T.J. (Eds.), The Retinal Pigment Epithelium. Oxford
University Press, Oxford, pp. 65–68.
Boulton, M., Dayhaw-Barker, P., 2001. The role of the retinal pigment
epithelium: topographical variation and ageing changes. Eye 15,
384–389.
Braunstein, G.M., Roman, R.M., Clancy, J.P., Kudlow, B.A., Taylor,
A.L., Shylonsky, V.G., Jovov, B., Peter, K., Jilling, T., Ismailov II,
Benos, D.J., Schwiebert, L.M., Fitz, J.G., Schwiebert, E.M., 2001.
Cystic fibrosis transmembrane conductance regulator facilitates ATP
release by stimulating a separate ATP release channel for autocrine
control of cell volume regulation. J. Biol. Chem. 276 (9), 6621–6630.
Bringmann, A., Reichenbach, A., Wiedemann, P., 2004. Pathomechanisms of cystoid macular edema. Ophthalmic Res. 36 (5), 241–249.
Bringmann, A., Uckermann, O., Pannicke, T., Iandiev, I., Reichenbach,
A., Wiedemann, P., 2005. Neuronal versus glial cell swelling in the
ischaemic retina. Acta Ophthalmol. Scand. 83 (5), 528–538.
Bultmann, R., Starke, K., 1994. Blockade by 4,40 -diisothiocyanatostilbene-2,20 -disulphonate (DIDS) of P2X-purinoceptors in rat vas
deferens. Br. J. Pharmacol. 112 (2), 690–694.
Burnstock, G., 2004. Introduction: P2 receptors. Curr. Top. Med. Chem. 4
(8), 793–803.
Burnstock, G., Knight, G.E., 2004. Cellular distribution and functions of
P2 receptor subtypes in different systems. Int. Rev. Cytol. 240, 31–304.
Cahalan, M.D., Wulff, H., Chandy, K.G., 2001. Molecular properties and
physiological roles of ion channels in the immune system. J. Clin.
Immunol. 21 (4), 235–252.
Campochiaro, P.A., 1993. Cytokine production by retinal pigmented
epithelial cells. Int. Rev. Cytol. 146, 75–82.
Campochiaro, P.A., Hackett, S.F., Vinores, S.A., Freund, J., Csaky, C.,
LaRochelle, W., Henderer, J., Johnson, M., Rodriguez, I.R., Friedman, Z., et al., 1994. Platelet-derived growth factor is an autocrine
growth stimulator in retinal pigmented epithelial cells. J. Cell Sci. 107
(Pt 9), 2459–2469.
Carafoli, E., 2005a. Calcium—a universal carrier of biological signals.
Delivered on 3 July 2003 at the Special FEBS Meeting in Brussels.
FEBS J. 272 (5), 1073–1089.
Carafoli, E., 2005b. The symposia on calcium binding proteins and
calcium function in health and disease: an historical account, and an
appraisal of their role in spreading the calcium message. Cell Calcium
37 (4), 279–281.
Catalan, M., Niemeyer, M.I., Cid, L.P., Sepulveda, F.V., 2004.
Basolateral ClC-2 chloride channels in surface colon epithelium:
regulation by a direct effect of intracellular chloride. Gastroenterology
126 (4), 1104–1114.
Catterall, W.A., 1998. Structure and function of neuronal Ca2+ channels
and their role in neurotransmitter release. Cell Calcium 24 (5 and 6),
307–323.
Catterall, W.A., 2000. Structure and regulation of voltage-gated Ca2+
channels. Annu. Rev. Cell Dev. Biol. 16, 521–555.
Catterall, W.A., Perez-Reyes, E., Snutch, T.P., Striessnig, J., 2005.
International Union of Pharmacology. XLVIII. Nomenclature and
structure-function relationships of voltage-gated calcium channels.
Pharmacol. Rev. 57 (4), 411–425.
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Celesia, G.G., 2001. Disorders of membrane channels or channelopathies.
Clin. Neurophysiol. 112 (1), 2–18.
Chebib, M., Johnston, G.A., 1999. The ‘ABC’ of GABA receptors: a brief
review. Clin. Exp. Pharmacol. Physiol. 26 (11), 937–940.
Cherubini, E., Ben-Ari, Y., Ito, S., Krnjevic, K., 1991. Persistent pulsatile
release of glutamate induced by N-methyl-D-aspartate in neonatal rat
hippocampal neurones. J. Physiol. 436, 531–547.
Chipperfield, A.R., Harper, A.A., 2000. Chloride in smooth muscle. Prog.
Biophys. Mol. Biol. 74 (3–5), 175–221.
Civan, M.M., Marano, C.W., Matschinsky, F.W., Peterson-Yantorno, K.,
1994. Prolonged incubation with elevated glucose inhibits the
regulatory response to shrinkage of cultured human retinal pigment
epithelial cells. J. Membr. Biol. 139 (1), 1–13.
Clapham, D.E., 1998. The list of potential volume-sensitive chloride
currents continues to swell (and shrink). J. Gen. Physiol. 111 (5),
623–624.
Clapham, D.E., Montell, C., Schultz, G., Julius, D., 2003. International
Union of Pharmacology. XLIII. Compendium of voltage-gated ion
channels: transient receptor potential channels. Pharmacol. Rev. 55
(4), 591–596.
Coca-Prados, M., Sanchez-Torres, J., Peterson-Yantorno, K., Civan,
M.M., 1996. Association of ClC-3 channel with Cl transport by
human nonpigmented ciliary epithelial cells. J. Membr. Biol. 150 (2),
197–208.
Collison, D.J., Tovell, V.E., Coombes, L.J., Duncan, G., Sanderson, J.,
2005. Potentiation of ATP-induced Ca2+ mobilisation in human
retinal pigment epithelial cells. Exp. Eye Res. 80 (4), 465–475.
Colquhoun, D., 1991. Ion channels: this year’s Nobel prize in physiology
or medicine. BMJ 303 (6808), 938–939.
Conn, P.J., 2003. Physiological roles and therapeutic potential of
metabotropic glutamate receptors. Ann. NY Acad. Sci. 1003,
12–21.
Crook, R.B., Polansky, J.R., 1992. Neurotransmitters and neuropeptides
stimulate inositol phosphates and intracellular calcium in cultured
human nonpigmented ciliary epithelium. Invest. Ophthalmol. Vis. Sci.
33 (5), 1706–1716.
Cross, H.E., Bard, L., 1974. Electro-oculography in Best’s macular
dystrophy. Am. J. Ophthalmol. 77 (1), 46–50.
d’Anglemont de Tassigny, A., Souktani, R., Ghaleh, B., Henry, P.,
Berdeaux, A., 2003. Structure and pharmacology of swelling-sensitive
chloride channels, I(Cl,swell). Fundam. Clin. Pharmacol. 17 (5),
539–553.
Dawson, D.W., Volpert, O.V., Gillis, P., Crawford, S.E., Xu, H.,
Benedict, W., Bouck, N.P., 1999. Pigment epithelium-derived factor:
a potent inhibitor of angiogenesis. Science 285 (5425), 245–248.
Decher, N., Maier, M., Dittrich, W., Gassenhuber, J., Bruggemann, A.,
Busch, A.E., Steinmeyer, K., 2001. Characterization of TASK-4, a
novel member of the pH-sensitive, two-pore domain potassium
channel family. FEBS Lett. 492 (1 and 2), 84–89.
Deguchi, J., Yamamoto, A., Yoshimori, T., Sugasawa, K., Moriyama, Y.,
Futai, M., Suzuki, T., Kato, K., Uyama, M., Tashiro, Y., 1994.
Acidification of phagosomes and degradation of rod outer segments in
rat retinal pigment epithelium. Invest. Ophthalmol. Vis. Sci. 35 (2),
568–579.
Delmas, P., Brown, D.A., 2005. Pathways modulating neural KCNQ/M
(Kv7) potassium channels. Nat. Rev. Neurosci. 6 (11), 850–862.
Delori, F.C., Goger, D.G., Dorey, C.K., 2001. Age-related accumulation
and spatial distribution of lipofuscin in RPE of normal subjects.
Invest. Ophthalmol. Vis. Sci. 42 (8), 1855–1866.
DiMattio, J., Degnan, K.J., Zadunaisky, J.A., 1983. A model for
transepithelial ion transport across the isolated retinal pigment
epithelium of the frog. Exp. Eye Res. 37 (5), 409–420.
Dissmann, E., Wischmeyer, E., Spauschus, A., Pfeil, D.V., Karschin, C.,
Karschin, A., 1996. Functional expression and cellular mRNA
localization of a G protein-activated K+ inward rectifier isolated
from rat brain. Biochem. Biophys. Res. Commun. 223 (2), 474–479.
Dmitriev, A.V., Govardovskii, V.I., Schwahn, H.N., Steinberg, R.H.,
1999. Light-induced changes of extracellular ions and volume in the
291
isolated chick retina-pigment epithelium preparation. Vis. Neurosci. 16
(6), 1157–1167.
Do, C.W., Civan, M.M., 2004. Basis of chloride transport in ciliary
epithelium. J. Membr. Biol. 200 (1), 1–13.
Do, C.W., Lu, W., Mitchell, C.H., Civan, M.M., 2005. Inhibition of
swelling-activated Cl currents by functional anti-ClC-3 antibody in
native bovine non-pigmented ciliary epithelial cells. Invest. Ophthalmol. Vis. Sci. 46 (3), 948–955.
Doring, F., Derst, C., Wischmeyer, E., Karschin, C., Schneggenburger,
R., Daut, J., Karschin, A., 1998. The epithelial inward rectifier channel
Kir7.1 displays unusual K+ permeation properties. J. Neurosci. 18
(21), 8625–8636.
Dornonville de la Cour, M., 1993. Ion transport in the retinal pigment
epithelium. A study with double barrelled ion-selective microelectrodes. Acta Ophthalmol. Suppl. (209), 1–32.
Drager, U.C., 1985. Calcium binding in pigmented and albino eyes. Proc.
Natl. Acad. Sci. USA 82 (19), 6716–6720.
Duan, D., Cowley, S., Horowitz, B., Hume, J.R., 1999. A serine residue in
ClC-3 links phosphorylation-dephosphorylation to chloride channel
regulation by cell volume. J. Gen. Physiol. 113 (1), 57–70.
Dyka, F.M., May, C.A., Enz, R., 2005. Subunits of the epithelial sodium
channel family are differentially expressed in the retina of mice with
ocular hypertension. J. Neurochem. 94 (1), 120–128.
Edelman, J.L., Miller, S.S., 1991. Epinephrine stimulates fluid absorption
across bovine retinal pigment epithelium. Invest. Ophthalmol. Vis. Sci.
32 (12), 3033–3040.
Edelman, J.L., Lin, H., Miller, S.S., 1994a. Acidification stimulates
chloride and fluid absorption across frog retinal pigment epithelium.
Am. J. Physiol. 266 (4 Pt 1), C946–C956.
Edelman, J.L., Lin, H., Miller, S.S., 1994b. Potassium-induced chloride
secretion across the frog retinal pigment epithelium. Am. J. Physiol.
266 (4 Pt 1), C957–C966.
Edwards, R.B., Brandt, J.T., Hardenbergh, G.S., 1987. A 31,000-dalton
protein released by cultured human retinal pigment epithelium. Invest.
Ophthalmol. Vis. Sci. 28 (7), 1213–1218.
Eggermont, J., 2004. Calcium-activated chloride channels: (un)known,
(un)loved? Proc. Am. Thorac. Soc. 1 (1), 22–27.
Endo, E.G., Yao, X.Y., Marmor, M.F., 1988. Pigment adherence as a
measure of retinal adhesion: dependence on temperature. Invest.
Ophthalmol. Vis. Sci. 29 (9), 1390–1396.
Ettaiche, M., Heurteaux, C., Blondeau, N., Borsotto, M., Tinel, N.,
Lazdunski, M., 2001. ATP-sensitive potassium channels (K(ATP)) in
retina: a key role for delayed ischemic tolerance. Brain Res. 890 (1),
118–129.
Evans, M.G., Marty, A., 1986. Calcium-dependent chloride currents in
isolated cells from rat lacrimal glands. J. Physiol. 378, 437–460.
Faundez, V., Hartzell, H.C., 2004. Intracellular chloride channels:
determinants of function in the endosomal pathway. Sci. STKE 2004
(233) re8.
Feldman, E.L., Randolph, A.E., Johnston, G.C., DelMonte, M.A.,
Greene, D.A., 1991. Receptor-coupled phosphoinositide hydrolysis in human retinal pigment epithelium. J. Neurochem. 56 (6),
2094–2100.
Feranchak, A.P., Doctor, R.B., Troetsch, M., Brookman, K., Johnson,
S.M., Fitz, J.G., 2004. Calcium-dependent regulation of secretion in
biliary epithelial cells: the role of apamin-sensitive SK channels.
Gastroenterology 127 (3), 903–913.
Fields, R.D., Lee, P.R., Cohen, J.E., 2005. Temporal integration of
intracellular Ca2+ signaling networks in regulating gene expression by
action potentials. Cell Calcium 37 (5), 433–442.
Fijisawa, K., Ye, J., Zadunaisky, J.A., 1993. A Na+/Ca2+ exchange
mechanism in apical membrane vesicles of the retinal pigment
epithelium. Curr. Eye Res. 12 (3), 261–270.
Finnemann, S.C., 2003. Role of alphavbeta5 integrin in regulating
phagocytosis by the retinal pigment epithelium. Adv. Exp. Med. Biol.
533, 337–342.
Fischmeister, R., Hartzell, H.C., 2005. Volume sensitivity of the
bestrophin family of chloride channels. J. Physiol. 562 (Pt 2), 477–491.
ARTICLE IN PRESS
292
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Fishman, M.L., Oberc, M.A., Hess, H.H., Engel, W.K., 1977. Ultrastructural demonstration of calcium in retina, retinal pigment
epithelium and choroid. Exp. Eye Res. 24 (4), 341–353.
Fordyce, C.B., Jagasia, R., Zhu, X., Schlichter, L.C., 2005. Microglia
Kv1.3 channels contribute to their ability to kill neurons. J. Neurosci.
25 (31), 7139–7149.
Foskett, J.K., 1990. [Ca2+]i modulation of Cl content controls cell
volume in single salivary acinar cells during fluid secretion. Am. J.
Physiol. 259 (6 Pt 1), C998–C1004.
Fox, J.A., Steinberg, R.H., 1992. Voltage-dependent currents in isolated
cells of the turtle retinal pigment epithelium. Pflugers Arch. 420 (5 and
6), 451–460.
Fox, J.A., Pfeffer, B.A., Fain, G.L., 1988. Single-channel recordings from
cultured human retinal pigment epithelial cells. J. Gen. Physiol. 91 (2),
193–222.
Fragoso, G., Lopez-Colome, A.M., 1999. Excitatory amino acid-induced
inositol phosphate formation in cultured retinal pigment epithelium.
Vis. Neurosci. 16 (2), 263–269.
Frambach, D.A., Misfeldt, D.S., 1983. Furosemide-sensitive Cl transport
in embryonic chicken retinal pigment epithelium. Am. J. Physiol. 244
(6), F679–F685.
Frank, R.N., 1997. Growth factors in age-related macular degeneration:
pathogenic and therapeutic implications. Ophthalmic Res. 29 (5),
341–353.
Friedman, Z., Hackett, S.F., Campochiaro, P.A., 1988. Human retinal
pigment epithelial cells possess muscarinic receptors coupled to
calcium mobilization. Brain Res. 446 (1), 11–16.
Fujii, S., Gallemore, R.P., Hughes, B.A., Steinberg, R.H., 1992. Direct
evidence for a basolateral membrane Cl conductance in toad retinal
pigment epithelium. Am. J. Physiol. 262 (2 Pt 1), C374–C383.
Fuller, C.M., Benos, D.J., 1992. Cftr!. Am. J. Physiol. 263 (2 Pt 1),
C267–C286.
Fuller, C.M., Ji, H.L., Tousson, A., Elble, R.C., Pauli, B.U., Benos, D.J.,
2001. Ca(2+)-activated Cl() channels: a newly emerging anion
transport family. Pflugers Arch. 443 (Suppl. 1), S107–S110.
Gadsby, D.C., Vergani, P., Csanady, L., 2006. The ABC protein turned
chloride channel whose failure causes cystic fibrosis. Nature 440
(7083), 477–483.
Gallemore, R.P., Steinberg, R.H., 1989a. Effects of DIDS on the chick
retinal pigment epithelium. I. Membrane potentials, apparent resistances, and mechanisms. J. Neurosci. 9 (6), 1968–1976.
Gallemore, R.P., Steinberg, R.H., 1989b. Effects of DIDS on the chick
retinal pigment epithelium. II. Mechanism of the light peak and other
responses originating at the basal membrane. Issue Series Title:
J. Neurosci. 9 (6), 1977–1984.
Gallemore, R.P., Steinberg, R.H., 1993. Light-evoked modulation of
basolateral membrane Cl conductance in chick retinal pigment
epithelium: the light peak and fast oscillation. J. Neurophysiol. 70 (4),
1669–1680.
Gallemore, R.P., Griff, E.R., Steinberg, R.H., 1988. Evidence in support
of a photoreceptoral origin for the ‘‘light-peak substance’’. Invest.
Ophthalmol. Vis. Sci. 29 (4), 566–571.
Gallemore, R.P., Hernandez, E., Tayyanipour, R., Fujii, S., Steinberg,
R.H., 1993. Basolateral membrane Cl and K+ conductances of the
dark-adapted chick retinal pigment epithelium. J. Neurophysiol. 70
(4), 1656–1668.
Gallemore, R.P., Li, J.D., Govardovskii, V.I., Steinberg, R.H., 1994.
Calcium gradients and light-evoked calcium changes outside rods in
the intact cat retina. Vis. Neurosci. 11 (4), 753–761.
Gallemore, R.P., Hughes, B.A., Miller, S.S., 1997. Retinal pigment
epithelial transport mechanisms and their contributions to the
electroretinogram. Prog. Ret. Eye Res. 16 (4), 509–566.
Gallemore, R.P., Bok, D., Hu, J., Maruiwa, F., Yocom, J., Strauss, O.,
Peachey, N.S., Marmorstein, A.D., 2004. Role for bestrophin in
generating the light peak of the DC Electroretinogram. Invest.
Ophthalmol. Vis. Sci. 45 e-abstract 1761.
Gandhi, R., Elble, R.C., Gruber, A.D., Schreur, K.D., Ji, H.L., Fuller,
C.M., Pauli, B.U., 1998. Molecular and functional characterization of
a calcium-sensitive chloride channel from mouse lung. J. Biol. Chem.
273 (48), 32096–32101.
Garcia, M.L., Garcia-Calvo, M., Hidalgo, P., Lee, A., MacKinnon, R.,
1994. Purification and characterization of three inhibitors of voltagedependent K+ channels from Leiurus quinquestriatus var. hebraeus
venom. Biochemistry 33 (22), 6834–6839.
Garron, L.K., 1963. The ultrastructure of the RPE with observations on
the choriocapillaris and Bruch’s membrane. Trans. Am. Ophthalmol.
Soc. 61, 545.
George Chandy, K., Wulff, H., Beeton, C., Pennington, M., Gutman,
G.A., Cahalan, M.D., 2004. K+ channels as targets for specific
immunomodulation. Issue Series Title: Trends Pharmacol. Sci. 25 (5),
280–289.
Ghatta, S., Nimmagadda, D., Xu, X., O’Rourke, S.T., 2006. Largeconductance, calcium-activated potassium channels: structural and
functional implications. Pharmacol. Ther. 110 (1), 103–116.
Godel, V., Chaine, G., Regenbogen, L., Coscas, G., 1986. Best’s
vitelliform macular dystrophy. Acta Opthalmol. 175 (Suppl.), 1–31.
Gola, M., Crest, M., 1993. Colocalization of active KCa channels and
Ca2+ channels within Ca2+ domains in helix neurons. Neuron 10 (4),
689–699.
Goldstein, S.A., Bockenhauer, D., O’Kelly, I., Zilberberg, N., 2001.
Potassium leak channels and the KCNK family of two-P-domain
subunits. Nat. Rev. Neurosci. 2 (3), 175–184.
Goldstein, S.A., Bayliss, D.A., Kim, D., Lesage, F., Plant, L.D., Rajan, S.,
2005. International Union of Pharmacology. LV. Nomenclature and
molecular relationships of two-P potassium channels. Pharmacol. Rev.
57 (4), 527–540.
Golestaneh, N., Picaud, S., Mirshahi, M., 2002. The mineralocorticoid
receptor in rodent retina: ontogeny and molecular identity. Mol. Vis. 8,
221–225.
Gomes, P., Malfait, M., Himpens, B., Vereecke, J., 2003. Intercellular
Ca(2+)-transient propagation in normal and high glucose solutions in
rat retinal epithelial (RPE-J) cells during mechanical stimulation. Cell
Calcium 34 (2), 185–192.
Gonzales-Fernandez, F., 2003. Interphotoreceptor retinoid-binding
protein—an old gene for new eyes. Issue Series Title: Vis. Res. 43,
3021–3036.
Gonzalez III, A., Crittenden, E.L., Garcia, D.M., 2004. Activation of
muscarinic acetylcholine receptors elicits pigment granule dispersion in
retinal pigment epithelium isolated from bluegill. BMC Neurosci. 5, 23.
Greenberger, L.M., Besharse, J.C., 1985. Stimulation of photoreceptor
disc shedding and pigment epithelial phagocytosis by glutamate,
aspartate, and other amino acids. J. Comp. Neurol. 239 (4), 361–372.
Greger, R., 2000. Role of CFTR in the colon. Issue Series Title: Annu.
Rev. Physiol. 62, 467–491.
Griff, E.R., Steinberg, R.H., 1984. Changes in apical [K+] produce
delayed basal membrane responses of the retinal pigment epithelium in
the gecko. J. Gen. Physiol. 83 (2), 193–211.
Grissmer, S., Nguyen, A.N., Cahalan, M.D., 1993. Calcium-activated
potassium channels in resting and activated human T lymphocytes.
Expression levels, calcium dependence, ion selectivity, and pharmacology. J. Gen. Physiol. 102 (4), 601–630.
Grunnet, M., Kaufmann, W.A., 2004. Coassembly of big conductance
Ca2+-activated K+ channels and L-type voltage-gated Ca2+ channels
in rat brain. J. Biol. Chem. 279 (35), 36445–36453.
Guillonneau, X., Regnier-Ricard, F., Dupuis, C., Courtois, Y., Mascarelli,
F., 1997. FGF2-stimulated release of endogenous FGF1 is associated
with reduced apoptosis in retinal pigmented epithelial cells. Exp. Cell
Res. 233 (1), 198–206.
Gutman, G.A., Chandy, K.G., Grissmer, S., Lazdunski, M., McKinnon,
D., Pardo, L.A., Robertson, G.A., Rudy, B., Sanguinetti, M.C.,
Stuhmer, W., Wang, X., 2005. International Union of Pharmacology.
LIII. Nomenclature and molecular relationships of voltage-gated
potassium channels. Pharmacol. Rev. 57 (4), 473–508.
Guymer, R., Luthert, P., Bird, A., 1999. Changes in Bruch’s membrane
and related structures with age. Issue Series Title: Prog. Retin. Eye
Res. 18 (1), 59–90.
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Hackett, S.F., Schoenfeld, C.L., Freund, J., Gottsch, J.D., Bhargave, S.,
Campochiaro, P.A., 1997. Neurotrophic factors, cytokines and stress
increase expression of basic fibroblast growth factor in retinal
pigmented epithelial cells. Exp. Eye Res. 64 (6), 865–873.
Hageman, G.S., Johnson, L.V., 1991. Structure, composition and function
of the retinal interphotoreceptor matrix. Prog. Retin. Eye. Res.,
207–249.
Hagen, A.R., Barabote, R.D., Saier, M.H., 2005. The bestrophin family of
anion channels: identification of prokaryotic homologues. Mol.
Membr. Biol. 22 (4), 291–302.
Hall, M.O., Abrams, T.A., Mittag, T.W., 1991. ROS ingestion by RPE
cells is turned off by increased protein kinase C activity and by
increased calcium. Exp. Eye Res. 52 (5), 591–598.
Hamann, S., 2002. Molecular mechanisms of water transport in the eye.
Int. Rev. Cytol. 215, 395–431.
Hamill, O.P., Marty, A., Neher, E., Sakmann, B., Sigworth, F.J., 1981.
Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 391 (2),
85–100.
Hara-Chikuma, M., Yang, B., Sonawane, N.D., Sasaki, S., Uchida, S.,
Verkman, A.S., 2005. ClC-3 chloride channels facilitate endosomal
acidification and chloride accumulation. J. Biol. Chem. 280 (2),
1241–1247.
Hartzell, C., Putzier, I., Arreola, J., 2005a. Calcium-activated chloride
channels. Annu. Rev. Physiol. 67, 719–758.
Hartzell, C., Qu, Z., Putzier, I., Artinian, L., Chien, L.T., Cui, Y., 2005b.
Looking chloride channels straight in the eye: bestrophins, lipofuscinosis, and retinal degeneration. Physiology (Bethesda) 20, 292–302.
Hartzell, H.C., Qu, Z., 2003. Chloride currents in acutely isolated
Xenopus retinal pigment epithelial cells. J. Physiol. 549 (Pt 2),
453–469.
Hernandez, E.V., Hu, J.G., Frambach, D.A., Gallemore, R.P., 1995.
Potassium conductances in cultured bovine and human retinal pigment
epithelium. Invest. Ophthalmol. Vis. Sci. 36 (1), 113–122.
Hess, H.H., 1975. The high calcium content of retinal pigmented
epithelium. Issue Series Title: Exp. Eye Res. 21 (5), 471–479.
Heth, C.A., Marescalchi, P.A., 1994. Inositol triphosphate generation in
cultured rat retinal pigment epithelium. Invest. Ophthalmol. Vis. Sci.
35 (2), 409–416.
Hevers, W., Luddens, H., 1998. The diversity of GABAA receptors.
Pharmacological and electrophysiological properties of GABAA
channel subtypes. Mol. Neurobiol. 18 (1), 35–86.
Hille, B., 2001. Ion Channels of Excitable Membranes. Sinauer Associates,
Inc., Sunderland, USA.
Himpens, B., Vereecke, J., 2000. [Intra- and intercellular Ca(2+)-signal
transduction]. Verh. K Acad. Geneeskd. Belg. 62 (6), 501–563.
Himpens, B., Stalmans, P., Gomez, P., Malfait, M., Vereecke, J., 1999.
Intra- and intercellular Ca2+ signaling in retinal pigment epithelial
cells during mechanical stimulation. FASEB J. 13 (Suppl.), S63–S68.
Ho, K., Nichols, C.G., Lederer, W.J., Lytton, J., Vassilev, P.M.,
Kanazirska, M.V., Hebert, S.C., 1993. Cloning and expression of an
inwardly rectifying ATP-regulated potassium channel. Nature 362
(6415), 31–38.
Ho, W.K., Kim, I., Lee, C.O., Youm, J.B., Lee, S.H., Earm, Y.E., 1999.
Blockade of HERG channels expressed in Xenopus laevis oocytes by
external divalent cations. Biophys. J. 76 (4), 1959–1971.
Hoffman, S., Gopalakrishna, R., Gundimeda, U., Murata, T., Spee, C.,
Ryan, S.J., Hinton, D.R., 1998. Verapamil inhibits proliferation,
migration and protein kinase C activity in human retinal pigment
epithelial cells. Exp. Eye Res. 67 (1), 45–52.
Hoffmann, E.K., 1992. Cell swelling and volume regulation. Can. J.
Physiol. Pharmacol. 70 (Suppl.), S310–S313.
Hollyfield, J.G., 1999. Hyaluronan and the functional organization of the
interphotoreceptor matrix. Invest. Ophthalmol. Vis. Sci. 40 (12),
2767–2769.
Holz, F.G., Pauleikhoff, D., Klein, R., Bird, A.C., 2004. Pathogenesis of
lesions in late age-related macular disease. Am. J. Ophthalmol. 137 (3),
504–510.
293
Hsu, S.C., Molday, R.S., 1994. Glucose metabolism in photoreceptor
outer segments. Its role in phototransduction and in NADPHrequiring reactions. J. Biol. Chem. 269 (27), 17954–17959.
Hu, J.G., Gallemore, R.P., Bok, D., Frambach, D.A., 1996. Chloride
transport in cultured fetal human retinal pigment epithelium. Exp. Eye
Res. 62 (4), 443–448.
Huang, B., Karwoski, C.J., 1992. Light-evoked expansion of subretinal
space volume in the retina of the frog. J. Neurosci. 12 (11), 4243–4252.
Huang, M.H., Wu, S.N., Chen, C.P., Shen, A.Y., 2002. Inhibition of
Ca2+-activated and voltage-dependent K+ currents by 2-mercaptophenyl-1,4-naphthoquinone in pituitary GH3 cells: contribution to its
antiproliferative effect. Life Sci. 70 (10), 1185–1203.
Hubner, C.A., Jentsch, T.J., 2002. Ion channel diseases. Hum. Mol.
Genet. 11 (20), 2435–2445.
Hughes, B.A., Segawa, Y., 1993. cAMP-activated chloride currents in
amphibian retinal pigment epithelial cells. J. Physiol. 466, 749–766.
Hughes, B.A., Steinberg, R.H., 1990. Voltage-dependent currents in
isolated cells of the frog retinal pigment epithelium. J. Physiol. 428,
273–297.
Hughes, B.A., Takahira, M., 1996. Inwardly rectifying K+ currents in
isolated human retinal pigment epithelial cells. Invest. Ophthalmol.
Vis. Sci. 37 (6), 1125–1139.
Hughes, B.A., Takahira, M., 1998. ATP-dependent regulation of inwardly
rectifying K+ current in bovine retinal pigment epithelial cells. Am. J.
Physiol. 275 (5 Pt 1), C1372–C1383.
Hughes, B.A., Miller, S.S., Machen, T.E., 1984. Effects of cyclic AMP on
fluid absorption and ion transport across frog retinal pigment
epithelium. Measurements in the open-circuit state. J. Gen. Physiol.
83 (6), 875–899.
Hughes, B.A., Miller, S.S., Farber, D.B., 1987. Adenylate cyclase
stimulation alters transport in frog retinal pigment epithelium. Am.
J. Physiol. 252 (4 Pt 1), C385–C395.
Hughes, B.A., Miller, S.S., Joseph, D.P., Edelman, J.L., 1988. cAMP
stimulates the Na+–K+ pump in frog retinal pigment epithelium. Am.
J. Physiol. 254 (1 Pt 1), C84–C98.
Hughes, B.A., Adorante, J.S., Miller, S.S., Lin, H., 1989. Apical
electrogenic NaHCO3 cotransport. A mechanism for HCO3 absorption across the retinal pigment epithelium. J. Gen. Physiol. 94 (1),
125–150.
Hughes, B.A., Shaikh, A., Ahmad, A., 1995a. Effects of Ba2+ and Cs+ on
apical membrane K+ conductance in toad retinal pigment epithelium.
Am. J. Physiol. 268 (5 Pt 1), C1164–C1172.
Hughes, B.A., Takahira, M., Segawa, Y., 1995b. An outwardly rectifying
K+ current active near resting potential in human retinal pigment
epithelial cells. Am. J. Physiol. 269 (1 Pt 1), C179–C187.
Hughes, B.A., Gallemore, R.P., Miller, S.S., 1998. Transport mechanisms
in the retinal pigment epithelium. In: Marmor, M.F., Wolfensberger,
T.J. (Eds.), The Retinal Pigment Epithelium. Oxford University Press,
New York, Oxford, pp. 103–134.
Hughes, B.A., Yang, D., Swaminathan, A., Pattnaik, B., 2006. Expression
of KCNQ Potassium Channels in Primate Retinal Pigment Epithelium
(ARVO Abstract). Invest. Ophthalmol. Vis. Sci.
Hurst, R.S., Toro, L., Stefani, E., 1996. Molecular determinants of
external barium block in Shaker potassium channels. FEBS Lett. 388
(1), 59–65.
Immel, J., Steinberg, R.H., 1986. Spatial buffering of K+ by the retinal
pigment epithelium in frog. J. Neurosci. 6 (11), 3197–3204.
Inagaki, N., Gonoi, T., Clement, J.P.t., Namba, N., Inazawa, J.,
Gonzalez, G., Aguilar-Bryan, L., Seino, S., Bryan, J., 1995a.
Reconstitution of IKATP: an inward rectifier subunit plus the
sulfonylurea receptor. Science 270 (5239), 1166–1170.
Inagaki, N., Tsuura, Y., Namba, N., Masuda, K., Gonoi, T., Horie, M.,
Seino, Y., Mizuta, M., Seino, S., 1995b. Cloning and functional
characterization of a novel ATP-sensitive potassium channel ubiquitously expressed in rat tissues, including pancreatic islets, pituitary,
skeletal muscle, and heart. J. Biol. Chem. 270 (11), 5691–5694.
Inagaki, N., Gonoi, T., Clement, J.P., Wang, C.Z., Aguilar-Bryan, L.,
Bryan, J., Seino, S., 1996. A family of sulfonylurea receptors
ARTICLE IN PRESS
294
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
determines the pharmacological properties of ATP-sensitive K+
channels. Neuron 16 (5), 1011–1017.
Inoue, R., 2005. TRP channels as a newly emerging non-voltage-gated
CA2+ entry channel superfamily. Curr. Pharm. Des. 11 (15),
1899–1914.
Irschick, E.U., Haas, G., Geiger, M., Singer, W., Ritsch-Marte, M.,
Konwalinka, G., Frick, M., Gottinger, W., Huemer, H.P., 2006.
Phagocytosis of human retinal pigment epithelial cells: evidence of a
diurnal rhythm, involvement of the cytoskeleton and interference of
antiviral drugs. Ophthalmic. Res. 38 (3), 164–174.
Ishida, K., Panjwani, N., Cao, Z., Streilein, J.W., 2003. Participation of
pigment epithelium in ocular immune privilege. 3. Epithelia cultured
from iris, ciliary body, and retina suppress T-cell activation by
partially non-overlapping mechanisms. Ocul. Immunol. Inflamm. 11
(2), 91–105.
Ishii, M., Horio, Y., Tada, Y., Hibino, H., Inanobe, A., Ito, M., Yamada,
M., Gotow, T., Uchiyama, Y., Kurachi, Y., 1997. Expression and
clustered distribution of an inwardly rectifying potassium channel,
KAB-2/Kir4.1, on mammalian retinal Muller cell membrane: their
regulation by insulin and laminin signals. J. Neurosci. 17 (20),
7725–7735.
Issa, N.P., Hudspeth, A.J., 1994. Clustering of Ca2+ channels and
Ca(2+)-activated K+ channels at fluorescently labeled presynaptic
active zones of hair cells. Proc. Natl. Acad. Sci. USA 91 (16),
7578–7582.
Jackson, P.S., Morrison, R., Strange, K., 1994. The volume-sensitive
organic osmolyte-anion channel VSOAC is regulated by nonhydrolytic
ATP binding. Am. J. Physiol. 267 (5 Pt 1), C1203–C1209.
Jan, L.Y., Jan, Y.N., 1997. Voltage-gated and inwardly rectifying
potassium channels. J. Physiol. 505 (Pt 2), 267–282.
Jentsch, T.J., Friedrich, T., Schriever, A., Yamada, H., 1999. The CLC
chloride channel family. Pflugers Arch. 437 (6), 783–795.
Jentsch, T.J., Stein, V., Weinreich, F., Zdebik, A.A., 2002. Molecular
structure and physiological function of chloride channels. Physiol.
Rev. 82 (2), 503–568.
Jentsch, T.J., Hubner, C.A., Fuhrmann, J.C., 2004. Ion channels: function
unravelled by dysfunction. Nat. Cell Biol. 6 (11), 1039–1047.
Jentsch, T.J., Maritzen, T., Zdebik, A.A., 2005a. Chloride channel
diseases resulting from impaired transepithelial transport or vesicular
function. J. Clin. Invest. 115 (8), 2039–2046.
Jentsch, T.J., Neagoe, I., Scheel, O., 2005b. CLC chloride channels and
transporters. Curr. Opin. Neurobiol. 15 (3), 319–325.
Jentsch, T.J., Poet, M., Fuhrmann, J.C., Zdebik, A.A., 2005c. Physiological functions of CLC Cl channels gleaned from human genetic
disease and mouse models. Annu. Rev. Physiol. 67, 779–807.
Joiner, W.J., Basavappa, S., Vidyasagar, S., Nehrke, K., Krishnan, S.,
Binder, H.J., Boulpaep, E.L., Rajendran, V.M., 2003. Active K+
secretion through multiple KCa-type channels and regulation by IKCa
channels in rat proximal colon. Am. J. Physiol. Gastrointest. Liver
Physiol. 285 (1), G185–G196.
Jordt, S.E., Jentsch, T.J., 1997. Molecular dissection of gating in the ClC-2
chloride channel. EMBO J. 16 (7), 1582–1592.
Joseph, D.P., Miller, S.S., 1991. Apical and basal membrane ion transport
mechanisms in bovine retinal pigment epithelium. J. Physiol. 435,
439–463.
Joseph, D.P., Miller, S.S., 1992. Alpha-1-adrenergic modulation of K and
Cl transport in bovine retinal pigment epithelium. J. Gen. Physiol. 99
(2), 263–290.
Jurkat-Rott, K., Lehmann-Horn, F., 2004. The patch clamp
technique in ion channel research. Curr. Pharm. Biotechnol. 5 (4),
387–395.
Karihaloo, A., Kato, K., Greene, D.A., Thomas, T.P., 1997. Protein
kinase and Ca2+ modulation of myo-inositol transport in cultured
retinal pigment epithelial cells. Am. J. Physiol. 273 (2 Pt 1),
C671–C678.
Kasper, D., Planells-Cases, R., Fuhrmann, J.C., Scheel, O., Zeitz, O.,
Ruether, K., Schmitt, A., Poet, M., Steinfeld, R., Schweizer, M.,
Kornak, U., Jentsch, T.J., 2005. Loss of the chloride channel ClC-7
leads to lysosomal storage disease and neurodegeneration. EMBO J.
24 (5), 1079–1091.
Keller, S.K., Jentsch, T.J., Koch, M., Wiederholt, M., 1986. Interactions
of pH and K+ conductance in cultured bovine retinal pigment
epithelial cells. Am. J. Physiol. 250 (1 Pt 1), C124–C137.
Keller, S.K., Jentsch, T.J., Janicke, I., Wiederholt, M., 1988. Regulation
of intracellular pH in cultured bovine retinal pigment epithelial cells.
Pflugers Arch. 411 (1), 47–52.
Kennedy, B.G., 1990. Na+–K+–Cl cotransport in cultured cells derived
from human retinal pigment epithelium. Am. J. Physiol. 259 (1 Pt 1),
C29–C34.
Kennedy, B.G., 1994. Volume regulation in cultured cells derived from
human retinal pigment epithelium. Am. J. Physiol. 266 (3 Pt 1),
C676–C683.
Kennedy, B.G., Mangini, N.J., 1996. Plasma membrane calcium-ATPase
in cultured human retinal pigment epithelium. Exp. Eye Res. 63 (5),
547–556.
Kenyon, E., Maminishkis, A., Joseph, D.P., Miller, S.S., 1997. Apical and
basolateral membrane mechanisms that regulate pHi in bovine retinal
pigment epithelium. Am. J. Physiol. 273 (2 Pt 1), C456–C472.
Kew, J.N., Kemp, J.A., 2005. Ionotropic and metabotropic glutamate
receptor structure and pharmacology. Psychopharmacology (Berl.) 179
(1), 4–29.
Kidd, J.F., Thorn, P., 2000. Intracellular Ca2+ and Cl channel activation
in secretory cells. Annu. Rev. Physiol. 62, 493–513.
King, G.L., Suzuma, K., 2000. Pigment-epithelium-derived factor—a key
coordinator of retinal neuronal and vascular functions. N. Engl. J.
Med. 342 (5), 349–351.
Kniesel, U., Wolburg, H., 1993. Tight junction complexity in the retinal
pigment epithelium of the chicken during development. Neurosci. Lett.
149 (1), 71–74.
Kofuji, P., Davidson, N., Lester, H.A., 1995. Evidence that neuronal Gprotein-gated inwardly rectifying K+ channels are activated by G beta
gamma subunits and function as heteromultimers. Proc. Natl. Acad.
Sci. USA 92 (14), 6542–6546.
Kojima, S., Rahner, C., Peng, S., Rizzolo, L.J., 2002. Claudin 5 is
transiently expressed during the development of the retinal pigment
epithelium. J. Membr. Biol. 186 (2), 81–88.
Konari, K., Sawada, N., Zhong, Y., Isomura, H., Nakagawa, T., Mori,
M., 1995. Development of the blood-retinal barrier in vitro: formation
of tight junctions as revealed by occludin and ZO-1 correlates with the
barrier function of chick retinal pigment epithelial cells. Exp. Eye Res.
61 (1), 99–108.
Kondo, T., Vicent, D., Suzuma, K., Yanagisawa, M., King, G.L.,
Holzenberger, M., Kahn, C.R., 2003. Knockout of insulin and IGF-1
receptors on vascular endothelial cells protects against retinal
neovascularization. J. Clin. Invest. 111 (12), 1835–1842.
Korenbrot, J.I., 1995. Ca2+ flux in retinal rod and cone outer segments:
differences in Ca2+ selectivity of the cGMP-gated ion channels and
Ca2+ clearance rates. Cell Calcium 18 (4), 285–300.
Korenbrot, J.I., Rebrik, T.I., 2002. Tuning outer segment Ca2+ homeostasis to phototransduction in rods and cones. Adv. Exp. Med. Biol.
514, 179–203.
Kornak, U., Kasper, D., Bosl, M.R., Kaiser, E., Schweizer, M., Schulz,
A., Friedrich, W., Delling, G., Jentsch, T.J., 2001. Loss of the ClC-7
chloride channel leads to osteopetrosis in mice and man. Cell 104 (2),
205–215.
Koschak, A., Reimer, D., Huber, I., Grabner, M., Glossmann, H., Engel,
J., Striessnig, J., 2001. alpha 1D (CaV1.3) subunits can form L-type
Ca2+ channels activating at negative voltages. J. Biol. Chem. 276 (25),
22100–22106.
Koschak, A., Reimer, D., Walter, D., Hoda, J.C., Heinzle, T., Grabner,
M., Striessnig, J., 2003. Cav1.4alpha1 subunits can form slowly
inactivating dihydropyridine-sensitive L-type Ca2+ channels lacking
Ca2+-dependent inactivation. J. Neurosci. 23 (14), 6041–6049.
Kramer, F., White, K., Pauleikhoff, D., Gehrig, A., Passmore, L., Rivera,
A., Rudolph, G., Kellner, U., Andrassi, M., Lorenz, B., Rohrschneider, K., Blankenagel, A., Jurklies, B., Schilling, H., Schutt, F., Holz,
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
F.G., Weber, B.H., 2000. Mutations in the VMD2 gene are associated
with juvenile-onset vitelliform macular dystrophy (Best disease) and
adult vitelliform macular dystrophy but not age-related macular
degeneration. Eur. J. Hum. Genet. 8 (4), 286–292.
Kramer, F., Stohr, H., Weber, B.H., 2004. Cloning and characterization
of the murine Vmd2 RFP-TM gene family. Cytogenet. Genome Res.
105 (1), 107–114.
Krapivinsky, G., Medina, I., Eng, L., Krapivinsky, L., Yang, Y.,
Clapham, D.E., 1998. A novel inward rectifier K+ channel with
unique pore properties. Neuron 20 (5), 995–1005.
Krzystolik, M.G., Afshari, M.A., Adamis, A.P., Gaudreault, J., Gragoudas, E.S., Michaud, N.A., Li, W., Connolly, E., O’Neill, C.A., Miller,
J.W., 2002. Prevention of experimental choroidal neovascularization
with intravitreal anti-vascular endothelial growth factor antibody
fragment. Arch. Ophthalmol. 120 (3), 338–346.
Kubo, Y., Baldwin, T.J., Jan, Y.N., Jan, L.Y., 1993a. Primary structure
and functional expression of a mouse inward rectifier potassium
channel. Nature 362 (6416), 127–133.
Kubo, Y., Reuveny, E., Slesinger, P.A., Jan, Y.N., Jan, L.Y., 1993b.
Primary structure and functional expression of a rat G-proteincoupled muscarinic potassium channel. Nature 364 (6440), 802–806.
Kubo, Y., Adelman, J.P., Clapham, D.E., Jan, L.Y., Karschin, A.,
Kurachi, Y., Lazdunski, M., Nichols, C.G., Seino, S., Vandenberg,
C.A., 2005. International Union of Pharmacology. LIV. Nomenclature
and molecular relationships of inwardly rectifying potassium channels.
Pharmacol. Rev. 57 (4), 509–526.
Kunzelmann, K., Bleich, M., Warth, R., Levy-Holzman, R., Garty, H.,
Schreiber, R., 2001a. Expression and function of colonic epithelial
KvLQT1 K+ channels. Clin. Exp. Pharmacol. Physiol. 28 (1 and 2),
79–83.
Kunzelmann, K., Hubner, M., Schreiber, R., Levy-Holzman, R., Garty,
H., Bleich, M., Warth, R., Slavik, M., von Hahn, T., Greger, R.,
2001b. Cloning and function of the rat colonic epithelial K+ channel
KVLQT1. J. Membr. Biol. 179 (2), 155–164.
Kuriyama, S., Yoshimura, N., Ohuchi, T., Tanihara, H., Ito, S., Honda,
Y., 1992. Neuropeptide-induced cytosolic Ca2+ transients and
phosphatidylinositol turnover in cultured human retinal pigment
epithelial cells. Brain Res. 579 (2), 227–233.
Kusaka, S., Horio, Y., Fujita, A., Matsushita, K., Inanobe, A., Gotow,
T., Uchiyama, Y., Tano, Y., Kurachi, Y., 1999. Expression and
polarized distribution of an inwardly rectifying K+ channel, Kir4.1, in
rat retinal pigment epithelium. J. Physiol. 520 (Pt 2), 373–381.
Kusaka, S., Inanobe, A., Fujita, A., Makino, Y., Tanemoto, M.,
Matsushita, K., Tano, Y., Kurachi, Y., 2001. Functional Kir7.1
channels localized at the root of apical processes in rat retinal pigment
epithelium. J. Physiol. 531 (Pt 1), 27–36.
la Cour, M., 1985. The retinal pigment epithelium controls the potassium
activity in the subretinal space. Acta Ophthalmol. Suppl. 173, 9–10.
la Cour, M., 1991a. Kinetic properties and Na+ dependence of rheogenic
Na(+)-HCO3- co-transport in frog retinal pigment epithelium.
J. Physiol. 439, 59–72.
la Cour, M., 1991b. pH homeostasis in the frog retina: the role of
Na+:HCO
3 co-transport in the retinal pigment epithelium. Acta
Ophthalmol. (Copenh) 69 (4), 496–504.
La Cour, M., 1992. Cl transport in frog retinal pigment epithelium. Exp.
Eye Res. 54 (6), 921–931.
la Cour, M., Lund-Andersen, H., Zeuthen, T., 1986. Potassium transport
of the frog retinal pigment epithelium: autoregulation of potassium
activity in the subretinal space. J. Physiol. 375, 461–479.
Lambooij, A.C., van Wely, K.H., Lindenbergh-Kortleve, D.J., Kuijpers,
R.W., Kliffen, M., Mooy, C.M., 2003. Insulin-like growth factor-I and
its receptor in neovascular age-related macular degeneration. Invest.
Ophthalmol. Vis. Sci. 44 (5), 2192–2198.
Lang, F., Ritter, M., Volkl, H., Haussinger, D., 1993. The biological
significance of cell volume. Ren. Physiol. Biochem. 16 (1 and 2), 48–65.
Lang, F., Busch, G.L., Ritter, M., Volkl, H., Waldegger, S., Gulbins, E.,
Haussinger, D., 1998a. Functional significance of cell volume
regulatory mechanisms. Physiol. Rev. 78 (1), 247–306.
295
Lang, F., Busch, G.L., Volkl, H., 1998b. The diversity of volume
regulatory mechanisms. Cell. Physiol. Biochem. 8 (1 and 2), 1–45.
Lang, F., Foller, M., Lang, K.S., Lang, P.A., Ritter, M., Gulbins, E.,
Vereninov, A., Huber, S.M., 2005. Ion channels in cell proliferation
and apoptotic cell death. J. Membr. Biol. 205 (3), 147–157.
LaVail, M.M., 1976. Rod outer segment disk shedding in rat retina:
relationship to cyclic lighting. Science 194 (4269), 1071–1074.
Lavallee, C.R., Chalifoux, J.R., Moosally, A.J., Balkema, G.W., 2003.
Elevated free calcium levels in the subretinal space elevate the absolute
dark-adapted threshold in hypopigmented mice. J. Neurophysiol. 90
(6), 3654–3662.
Lehmann-Horn, F., Jurkat-Rott, K., 1999. Voltage-gated ion channels
and hereditary disease. Physiol. Rev. 79 (4), 1317–1372.
Leipziger, J., 2003. Control of epithelial transport via luminal P2
receptors. Am. J. Physiol. Renal Physiol. 284 (3), F419–F432.
Lerche, W., 1963. Electron microscopic observations of Bruch’s membrane in the human eye. Ber. Dtsch. Ophthalmol. Ges. 65, 384.
Leroy, C., Dagenais, A., Berthiaume, Y., Brochiero, E., 2004. Molecular
identity and function in transepithelial transport of K(ATP) channels
in alveolar epithelial cells. Am. J. Physiol. Lung Cell Mol. Physiol. 286
(5), L1027–L1037.
Lesage, F., Lazdunski, M., 2000. Molecular and functional properties of
two-pore-domain potassium channels. Am. J. Physiol. Renal. Physiol.
279 (5), F793–F801.
Li, J.D., Gallemore, R.P., Dmitriev, A., Steinberg, R.H., 1994a. Lightdependent hydration of the space surrounding photoreceptors in chick
retina. Invest. Ophthalmol. Vis. Sci. 35 (6), 2700–2711.
Li, J.D., Govardovskii, V.I., Steinberg, R.H., 1994b. Light-dependent
hydration of the space surrounding photoreceptors in the cat retina.
Vis. Neurosci. 11 (4), 743–752.
Li, M., McCann, J.D., Liedtke, C.M., Nairn, A.C., Greengard, P., Welsh,
M.J., 1988. Cyclic AMP-dependent protein kinase opens chloride
channels in normal but not cystic fibrosis airway epithelium. Nature
331 (6154), 358–360.
Liebman, P.A., Parker, K.R., Dratz, E.A., 1987. The molecular
mechanism of visual excitation and its relation to the structure and
composition of the rod outer segment. Annu. Rev. Physiol. 49,
765–791.
Lin, H., Miller, S.S., 1991. pHi regulation in frog retinal pigment
epithelium: two apical membrane mechanisms. Am. J. Physiol. 261
(1 Pt 1), C132–C142.
Liu, X., Chang, Y., Reinhart, P.H., Sontheimer, H., Chang, Y., 2002.
Cloning and characterization of glioma BK, a novel BK channel
isoform highly expressed in human glioma cells. J. Neurosci. 22 (5),
1840–1849.
Loeffler, K.U., Mangini, N.J., 1998. Immunohistochemical localization of
Na+/Ca2+ exchanger in human retina and retinal pigment epithelium.
Graefes Arch. Clin. Exp. Ophthalmol. 236 (12), 929–933.
Loewen, M.E., Bekar, L.K., Gabriel, S.E., Walz, W., Forsyth, G.W.,
2002a. pCLCA1 becomes a cAMP-dependent chloride conductance
mediator in Caco-2 cells. Biochem. Biophys Res. Commun. 298 (4),
531–536.
Loewen, M.E., Gabriel, S.E., Forsyth, G.W., 2002b. The calciumdependent chloride conductance mediator pCLCA1. Am. J. Physiol.
Cell Physiol. 283 (2), C412–C421.
Loewen, M.E., Smith, N.K., Hamilton, D.L., Grahn, B.H., Forsyth,
G.W., 2003. CLCA protein and chloride transport in canine retinal
pigment epithelium. Am. J. Physiol. Cell Physiol. 285 (5),
C1314–C1321.
Lopez-Colome, A.M., Salceda, R., Fragoso, G., 1993. Specific interaction
of glutamate with membranes from cultured retinal pigment epithelium. J. Neurosci. Res. 34 (4), 454–461.
Lopez-Colome, A.M., Fragoso, G., Wright, C.E., Sturman, J.A., 1994.
Excitatory amino acid receptors in membranes from cultured human
retinal pigment epithelium. Curr. Eye Res. 13 (8), 553–560.
Lopez, P.F., Sippy, B.D., Lambert, H.M., Thach, A.B., Hinton, D.R.,
1996. Transdifferentiated retinal pigment epithelial cells are immunoreactive for vascular endothelial growth factor in surgically excised
ARTICLE IN PRESS
296
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
age-related macular degeneration-related choroidal neovascular membranes. Invest. Ophthalmol. Vis. Sci. 37 (5), 855–868.
Lu, Z., 2004. Mechanism of rectification in inward-rectifier K+ channels.
Annu. Rev. Physiol. 66, 103–129.
Ma, T., Thiagarajah, J.R., Yang, H., Sonawane, N.D., Folli, C., Galietta,
L.J., Verkman, A.S., 2002. Thiazolidinone CFTR inhibitor identified
by high-throughput screening blocks cholera toxin-induced intestinal
fluid secretion. J. Clin. Invest. 110 (11), 1651–1658.
MacDonald, P.E., Wheeler, M.B., 2003. Voltage-dependent K(+)
channels in pancreatic beta cells: role, regulation and potential as
therapeutic targets. Diabetologia 46 (8), 1046–1062.
Mahon, G.J., Anderson, H.R., Gardiner, T.A., McFarlane, S., Archer,
D.B., Stitt, A.W., 2004. Chloroquine causes lysosomal dysfunction in
neural retina and RPE: implications for retinopathy. Curr. Eye Res. 28
(4), 277–284.
Mainster, M.A., 1987. Light and macular degeneration: a biophysical and
clinical perspective. Eye 1 (Part 2), 304–310.
Maminishkis, A., Jalickee, S., Blaug, S.A., Rymer, J., Yerxa, B.R.,
Peterson, W.M., Miller, S.S., 2002. The P2Y(2) receptor agonist
INS37217 stimulates RPE fluid transport in vitro and retinal
reattachment in rat. Invest. Ophthalmol. Vis. Sci. 43 (11),
3555–3566.
Mangini, N.J., Haugh-Scheidt, L., Valle, J.E., Cragoe Jr., E.J., Ripps, H.,
Kennedy, B.G., 1997. Sodium–calcium exchanger in cultured human
retinal pigment epithelium. Exp. Eye Res. 65 (6), 821–834.
Mannerstrom, M., Maenpaa, H., Toimela, T., Salminen, L., Tahti, H.,
2001. The phagocytosis of rod outer segments is inhibited by selected
drugs in retinal pigment epithelial cell cultures. Pharmacol. Toxicol. 88
(1), 27–33.
Marcus, D.C., Sunose, H., Liu, J., Shen, Z., Scofield, M.A., 1997. P2U
purinergic receptor inhibits apical IsK/KvLQT1 channel via protein
kinase C in vestibular dark cells. Am. J. Physiol. 273 (6 Part 1),
C2022–C2029.
Marcus, D.C., Sunose, H., Liu, J., Bennett, T., Shen, Z., Scofield, M.A.,
Ryan, A.F., 1998. Protein kinase C mediates P2U purinergic receptor
inhibition of K+ channel in apical membrane of strial marginal cells.
Hear Res. 115 (1 and 2), 82–92.
Marmor, M.F., 1983. From sea lemons to c-waves. Cell Mol. Neurobiol.
3 (4), 285–295.
Marmor, M.F., 1990. Control of subretinal fluid: experimental and clinical
studies. Eye 4 (Pt 2), 340–344.
Marmor, M.F., 1993. Mechanisms of retinal adhesion. Prog. Retin. Eye
Res. 12, 179–203.
Marmor, M.F., 1999. Mechanisms of fluid accumulation in retinal edema.
Doc. Ophthalmol. 97 (3 and 4), 239–249.
Marmor, M.F., 2005. Mechanisms of normal retinal adhesion marmor. In:
Ryan, S.J. (Ed.), The Retina, vol. 3, p. 112.
Marmor, M.F., Yao, X.Y., 1989. The enhancement of retinal adhesiveness
by ouabain appears to involve cellular edema. Invest. Ophthalmol. Vis.
Sci. 30 (7), 1511–1514.
Marmorstein, A.D., 2001. The polarity of the retinal pigment epithelium.
Traffic 2 (12), 867–872.
Marmorstein, A.D., Finnemann, S.C., Bonilha, V.L., Rodriguez-Boulan,
E., 1998. Morphogenesis of the retinal pigment epithelium: toward
understanding retinal degenerative diseases. Ann. NY Acad. Sci. 857,
1–12.
Marmorstein, A.D., Marmorstein, L.Y., Rayborn, M., Wang, X.,
Hollyfield, J.G., Petrukhin, K., 2000. Bestrophin, the product of the
Best vitelliform macular dystrophy gene (VMD2), localizes to the
basolateral plasma membrane of the retinal pigment epithelium. Proc.
Natl. Acad. Sci. USA 97 (23), 12758–12763.
Marmorstein, A.D., Stanton, J.B., Yocom, J., Bakall, B., Schiavone,
M.T., Wadelius, C., Marmorstein, L.Y., Peachey, N.S., 2004. A model
of Best vitelliform macular dystrophy in rats. Invest. Ophthalmol. Vis.
Sci. 45, 3733–3739.
Marmorstein, L.Y., Wu, J., McLaughlin, P., Yocom, J., Karl, M.O.,
Neussert, R., Wimmers, S., Stanton, J.B., Gregg, R.G., Strauss, O.,
Peachey, N.S., Marmorstein, A.D., 2006. The Light peak of the
electroretinogram is dependent on voltage-gated calcium channels
and antagonized by Bestrophin (Best-1). J. Gen. Physiol. 127 (5),
577–589.
Marquardt, A., Stohr, H., Passmore, L.A., Kramer, F., Rivera, A.,
Weber, B.H., 1998. Mutations in a novel gene, VMD2, encoding a
protein of unknown properties cause juvenile-onset vitelliform macular
dystrophy (Best’s disease). Hum. Mol. Genet. 7 (9), 1517–1525.
Marquardt, A., Stohr, H., White, K., Weber, B.H., 2000. cDNA cloning,
genomic structure, and chromosomal localization of three members of
the human fatty acid desaturase family. Genomics 66 (2), 175–183.
Marrion, N.V., Tavalin, S.J., 1998. Selective activation of Ca2+-activated
K+ channels by co-localized Ca2+ channels in hippocampal neurons.
Nature 395 (6705), 900–905.
Marshall, J., Hussain, A.A., Starita, C., Moore, D.J., Patmore, A.L.,
1998. Aging and Bruch’s membrane. In: Marmor, M.F., Wolfensberger, T.J. (Eds.), The Retinal Pigment Epithelium. Oxford University
Press, New York, Oxford, pp. 669–692.
Masi, A., Becchetti, A., Restano-Cassulini, R., Polvani, S., Hofmann, G.,
Buccoliero, A.M., Paglierani, M., Pollo, B., Taddei, G.L., Gallina, P.,
Di Lorenzo, N., Franceschetti, S., Wanke, E., Arcangeli, A., 2005.
hERG1 channels are overexpressed in glioblastoma multiforme and
modulate VEGF secretion in glioblastoma cell lines. Br. J. Cancer 93
(7), 781–792.
Mata, N.L., Radu, R.A., Clemmons, R.C., Travis, G.H., 2002.
Isomerization and oxidation of vitamin A in cone-dominant retinas:
a novel pathway for visual-pigment regeneration in daylight. Neuron
36 (1), 69–80.
Matchkov, V.V., Aalkjaer, C., Nilsson, H., 2004. A cyclic GMPdependent calcium-activated chloride current in smooth-muscle cells
from rat mesenteric resistance arteries. J. Gen. Physiol. 123 (2),
121–134.
Mayer, M.L., 2005. Glutamate receptor ion channels. Curr. Opin.
Neurobiol. 15 (3), 282–288.
McCarty, N.A., 2000. Permeation through the CFTR chloride channel.
J. Exp. Biol. 203 (Part 13), 1947–1962.
Meadows, H.J., Benham, C.D., Cairns, W., Gloger, I., Jennings, C.,
Medhurst, A.D., Murdock, P., Chapman, C.G., 2000. Cloning,
localisation and functional expression of the human orthologue of
the TREK-1 potassium channel. Pflugers Arch. 439 (6), 714–722.
Mears, D., 2004. Regulation of insulin secretion in islets of Langerhans by
Ca(2+)channels. J. Membr. Biol. 200 (2), 57–66.
Melvin, J.E., 1999. Chloride channels and salivary gland function. Crit.
Rev. Oral Biol. Med. 10 (2), 199–209.
Mergler, S., Strauss, O., 2002. Stimulation of L-type Ca(2+) channels by
increase of intracellular InsP3 in rat retinal pigment epithelial cells.
Exp. Eye Res. 74 (1), 29–40.
Mergler, S., Steinhausen, K., Wiederholt, M., Strauss, O., 1998. Altered
regulation of L-type channels by protein kinase C and protein tyrosine
kinases as a pathophysiologic effect in retinal degeneration. FASEB J.
12 (12), 1125–1134.
Meyer, C.H., Hotta, K., Peterson, W.M., Toth, C.A., Jaffe, G.J., 2002.
Effect of INS37217, a P2Y(2) receptor agonist, on experimental retinal
detachment and electroretinogram in adult rabbits. Invest. Ophthalmol. Vis. Sci. 43 (11), 3567–3574.
Michna, M., Knirsch, M., Hoda, J.C., Muenkner, S., Langer, P., Platzer,
J., Striessnig, J., Engel, J., 2003. CaV1.3 (alpha1D) Ca2+ currents in
neonatal outer hair cells of mice. J. Physiol. 553 (Part 3), 747–758.
Middleton, R.E., Pheasant, D.J., Miller, C., 1996. Homodimeric
architecture of a ClC-type chloride ion channel. Nature 383 (6598),
337–340.
Miller, S., Farber, D., 1984. Cyclic AMP modulation of ion transport
across frog retinal pigment epithelium. Measurements in the shortcircuit state. J. Gen. Physiol. 83 (6), 853–874.
Miller, S.S., Edelman, J.L., 1990. Active ion transport pathways in the
bovine retinal pigment epithelium. J. Physiol. 424, 283–300.
Miller, S.S., Hughes, B.A., Machen, T.E., 1982. Fluid transport across
retinal pigment epithelium is inhibited by cyclic AMP. Proc. Natl.
Acad. Sci. USA 79 (6), 2111–2115.
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Miller, S.S., Steinberg, R.H., 1977a. Active transport of ions across frog
retinal pigment epithelium. Exp. Eye Res. 25 (3), 235–248.
Miller, S.S., Steinberg, R.H., 1977b. Passive ionic properties of frog retinal
pigment epithelium. J. Membr. Biol. 36 (4), 337–372.
Miller, S.S., Steinberg, R.H., 1982. Potassium transport across the frog
retinal pigment epithelium. J. Membr. Biol. 67 (3), 199–209.
Mirshahi, M., Nicolas, C., Mirshahi, S., Golestaneh, N., d’Hermies, F.,
Agarwal, M.K., 1999. Immunochemical analysis of the sodium
channel in rodent and human eye. Exp. Eye Res. 69 (1), 21–32.
Mitchell, C.H., 2001. Release of ATP by a human retinal pigment
epithelial cell line: potential for autocrine stimulation through
subretinal space. J. Physiol. 534 (Part 1), 193–202.
Morishige, K., Takahashi, N., Jahangir, A., Yamada, M., Koyama, H.,
Zanelli, J.S., Kurachi, Y., 1994. Molecular cloning and functional
expression of a novel brain-specific inward rectifier potassium channel.
FEBS Lett. 346 (2 and 3), 251–256.
Moriya, T., Miyashita, Y., Arai, J., Kusunoki, S., Abe, M., Asami, K.,
1996. Light-sensitive response in melanophores of Xenopus laevis: I.
Spectral characteristics of melanophore response in isolated tail fin of
Xenopus tadpole. J. Exp. Zool. 276 (1), 11–18.
Moroi-Fetters, S.E., Earley, O., Hirakata, A., Caron, M.G., Jaffe, G.J.,
1995. Binding, coupling, and mRNA subtype heterogeneity of alpha
1-adrenergic receptors in cultured human RPE. Exp. Eye Res. 60 (5),
527–532.
Morris, A.P., 1999. The regulation of epithelial cell cAMP- and calciumdependent chloride channels. Adv. Pharmacol. 46, 209–251.
Moseley, H., Foulds, W.S., Allan, D., Kyle, P.M., 1984. Routes of
clearance of radioactive water from the rabbit vitreous. Br. J.
Ophthalmol. 68 (3), 145–151.
Mousa, S.A., Lorelli, W., Campochiaro, P.A., 1999. Role of hypoxia and
extracellular matrix-integrin binding in the modulation of angiogenic
growth factors secretion by retinal pigmented epithelial cells. J. Cell
Biochem. 74 (1), 135–143.
Nagineni, C.N., Samuel, W., Nagineni, S., Pardhasaradhi, K., Wiggert,
B., Detrick, B., Hooks, J.J., 2003. Transforming growth factor-beta
induces expression of vascular endothelial growth factor in human
retinal pigment epithelial cells: Involvement of mitogen-activated
protein kinases. J. Cell Physiol. 197 (3), 453–462.
Nakashima, S., Tsunematsu, Y., Nozawa, Y., 1989. Phosphoinositide
turnover and Ca2+ mobilization during phagocytosis in cultured chick
retinal pigment epithelial cells. Nippon Ganka Gakkai Zasshi 93 (1),
149–154.
Nash, M., Flanigan, T., Leslie, R., Osborne, N., 1999. Serotonin-2A
receptor mRNA expression in rat retinal pigment epithelial cells.
Ophthalmic Res. 31 (1), 1–4.
Nash, M.S., Osborne, N.N., 1995. Agonist-induced effects on cyclic AMP
metabolism are affected in pigment epithelial cells of the Royal College
of Surgeons rat. Neurochem. Int. 27 (3), 253–262.
Neher, E., Sakmann, B., 1976. Single-channel currents recorded from
membrane of denervated frog muscle fibres. Nature 260 (5554),
799–802.
Neher, E., Sakmann, B., 1992. The patch clamp technique. Sci. Am. 266
(3), 44–51.
Newman, E., Reichenbach, A., 1996. The Muller cell: a functional element
of the retina. Trends Neurosci. 19 (8), 307–312.
Nguyen-Legros, J., Hicks, D., 2000. Renewal of photoreceptor outer
segments and their phagocytosis by the retinal pigment epithelium. Int.
Rev. Cytol. 196, 245–313.
Nichols, C.G., Lopatin, A.N., 1997. Inward rectifier potassium channels.
Annu. Rev. Physiol. 59, 171–191.
Niemeyer, M.I., Cid, L.P., Zuniga, L., Catalan, M., Sepulveda, F.V.,
2003. A conserved pore-lining glutamate as a voltage- and chloridedependent gate in the ClC-2 chloride channel. J. Physiol. 553 (Part 3),
873–879.
Nilius, B., Droogmans, G., 2003. Amazing chloride channels: an overview.
Acta Physiol. Scand. 177 (2), 119–147.
Nilius, B., Eggermont, J., Voets, T., Droogmans, G., 1996. Volumeactivated Cl channels. Gen. Pharmacol. 27 (7), 1131–1140.
297
Nilius, B., Eggermont, J., Voets, T., Buyse, G., Manolopoulos, V.,
Droogmans, G., 1997. Properties of volume-regulated anion channels
in mammalian cells. Prog. Biophys. Mol. Biol. 68 (1), 69–119.
Nishiyama, K., Sakaguchi, H., Hu, J.G., Bok, D., Hollyfield, J.G., 2002.
Claudin localization in cilia of the retinal pigment epithelium. Anat.
Rec. 267 (3), 196–203.
North, R.A., 2002. Molecular physiology of P2X receptors. Physiol. Rev.
82 (4), 1013–1067.
North, R.A., Surprenant, A., 2000. Pharmacology of cloned P2X
receptors. Annu. Rev. Pharmacol. Toxicol. 40, 563–580.
Oakley II, B., Steinberg, R.H., 1982. Effects of maintained illumination
upon [K+]0 in the subretinal space of the frog retina. Vis. Res. 22 (7),
767–773.
O’Grady, S.M., Lee, S.Y., 2005. Molecular diversity and function of
voltage-gated (Kv) potassium channels in epithelial cells. Int.
J. Biochem. Cell Biol. 37 (8), 1578–1594.
Oh, H., Takagi, H., Takagi, C., Suzuma, K., Otani, A., Ishida, K.,
Matsumura, M., Ogura, Y., Honda, Y., 1999. The potential angiogenic
role of macrophages in the formation of choroidal neovascular
membranes. Invest. Ophthalmol. Vis. Sci. 40 (9), 1891–1898.
Ohno-Matsui, K., Yoshida, T., Uetama, T., Mochizuki, M., Morita, I.,
2003. Vascular endothelial growth factor upregulates pigment
epithelium-derived factor expression via VEGFR-1 in human retinal
pigment epithelial cells. Biochem. Biophys. Res. Commun. 303 (3),
962–967.
Okada, Y., 2004. Ion channels and transporters involved in cell volume
regulation and sensor mechanisms. Cell Biochem. Biophys. 41 (2),
233–258.
Okada, Y., Maeno, E., 2001. Apoptosis, cell volume regulation and
volume-regulatory chloride channels. Comp. Biochem. Physiol. A
Mol. Integr. Physiol. 130 (3), 377–383.
Okada, Y., Maeno, E., Shimizu, T., Dezaki, K., Wang, J., Morishima, S.,
2001. Receptor-mediated control of regulatory volume decrease
(RVD) and apoptotic volume decrease (AVD). J. Physiol. 532 (Part
1), 3–16.
Osborne, N.N., Fitzgibbon, F., Nash, M., Liu, N.P., Leslie, R.,
Cholewinski, A., 1993. Serotonergic, 5-HT2, receptor-mediated
phosphoinositide turnover and mobilization of calcium in cultured
rat retinal pigment epithelium cells. Vis. Res. 33 (16), 2171–2179.
Park, J.B., Kim, H.J., Ryu, P.D., Moczydlowski, E., 2003. Effect of
phosphatidylserine on unitary conductance and Ba2+ block of the BK
Ca2+-activated K+ channel: re-examination of the surface charge
hypothesis. J. Gen. Physiol. 121 (5), 375–397.
Patel, A.J., Maingret, F., Magnone, V., Fosset, M., Lazdunski, M.,
Honore, E., 2000. TWIK-2, an inactivating 2P domain K+ channel.
J. Biol. Chem. 275 (37), 28722–28730.
Pauli, B.U., Abdel-Ghany, M., Cheng, H.C., Gruber, A.D., Archibald,
H.A., Elble, R.C., 2000. Molecular characteristics and functional
diversity of CLCA family members. Clin. Exp. Pharmacol. Physiol. 27
(11), 901–905.
Pearson, R.A., Catsicas, M., Becker, D.L., Bayley, P., Luneborg, N.L.,
Mobbs, P., 2004. Ca(2+) signalling and gap junction coupling within
and between pigment epithelium and neural retina in the developing
chick. Eur. J. Neurosci. 19 (9), 2435–2445.
Pearson, W.L., Dourado, M., Schreiber, M., Salkoff, L., Nichols, C.G.,
1999. Expression of a functional Kir4 family inward rectifier K+
channel from a gene cloned from mouse liver. J. Physiol. 514 (Part 3),
639–653.
Pepperberg, D.R., Okajima, T.L., Wiggert, B., Ripps, H., Crouch, R.K.,
Chader, G.J., 1993. Interphotoreceptor retinoid-binding protein
(IRBP). Molecular biology and physiological role in the visual cycle
of rhodopsin. Mol. Neurobiol. 7 (1), 61–85.
Pessia, M., Tucker, S.J., Lee, K., Bond, C.T., Adelman, J.P., 1996.
Subunit positional effects revealed by novel heteromeric inwardly
rectifying K+ channels. EMBO J. 15 (12), 2980–2987.
Peters, S., Reinthal, E., Blitgen-Heinecke, P., Bartz-Schmidt, K.U.,
Schraermeyer, U., 2006. Inhibition of lysosomal degradation in retinal
pigment epithelium cells induces exocytosis of phagocytic residual
ARTICLE IN PRESS
298
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
material at the basolateral plasma membrane. Ophthalmic Res. 38 (2),
83–88.
Peterson, W.M., Meggyesy, C., Yu, K., Miller, S.S., 1997. Extracellular
ATP activates calcium signaling, ion, and fluid transport in retinal
pigment epithelium. J. Neurosci. 17 (7), 2324–2337.
Petrukhin, K., Koisti, M.J., Bakall, B., Li, W., Xie, G., Marknell, T.,
Sandgren, O., Forsman, K., Holmgren, G., Andreasson, S., Vujic, M.,
Bergen, A.A., McGarty-Dugan, V., Figueroa, D., Austin, C.P.,
Metzker, M.L., Caskey, C.T., Wadelius, C., 1998. Identification of
the gene responsible for Best macular dystrophy. Nature Genetics 19
(3), 241–247.
Picollo, A., Pusch, M., 2005. Chloride/proton antiporter activity of
mammalian CLC proteins ClC-4 and ClC-5. Nature 436 (7049),
420–423.
Pinto, L.H., Klumpp, D.J., 1998. Localization of potassium channels in
the retina. Prog. Retin. Eye Res. 17 (2), 207–230.
Piwon, N., Gunther, W., Schwake, M., Bosl, M.R., Jentsch, T.J., 2000.
ClC-5 Cl-channel disruption impairs endocytosis in a mouse model
for Dent’s disease. Nature 408 (6810), 369–373.
Plant, L.D., Rajan, S., Goldstein, S.A., 2005. K2P channels and their
protein partners. Curr. Opin. Neurobiol. 15 (3), 326–333.
Pollack, K., Kreuz, F.R., Pillunat, L.E., 2005. Best’s disease with normal
EOG Fallvorstellung einer familiaren Makuladystrophie. Ophthalmologe.
Poyer, J.F., Ryan, J.S., Kelly, M.E., 1996. G protein-mediated activation
of a nonspecific cation current in cultured rat retinal pigment epithelial
cells. J. Membr. Biol. 153 (1), 13–26.
Prat, A.G., Reisin, I.L., Ausiello, D.A., Cantiello, H.F., 1996. Cellular
ATP release by the cystic fibrosis transmembrane conductance
regulator. Am. J. Physiol. 270 (2 Part 1), C538–C545.
Pusch, M., 2004. Ca(2+)-activated chloride channels go molecular. J. Gen.
Physiol. 123 (4), 323–325.
Pusch, M., Jentsch, T.J., 2005. Unique structure and function of chloride
transporting CLC proteins. IEEE Trans. Nanobiosci. 4 (1), 49–57.
Pusch, M., Jordt, S.E., Stein, V., Jentsch, T.J., 1999. Chloride dependence
of hyperpolarization-activated chloride channel gates. J. Physiol. 515
(Part 2), 341–353.
Pusch, M., Zifarelli, G., Murgia, A.R., Picollo, A., Babini, E., 2006.
Channel or transporter? The CLC saga continues. Exp. Physiol. 91 (1),
149–152.
Qu, Z., Hartzell, C., 2004. Determinants of anion permeation in the
second transmembrane domain of the mouse bestrophin-2 chloride
channel. J. Gen. Physiol. 124 (4), 371–382.
Qu, Z., Wei, R.W., Mann, W., Hartzell, H.C., 2003. Two bestrophins
cloned from Xenopus laevis oocytes express Ca(2+)-activated Cl()
currents. J. Biol. Chem. 278 (49), 49563–49572.
Qu, Z., Fischmeister, R., Hartzell, C., 2004. Mouse bestrophin-2 is a bona
fide Cl() channel: identification of a residue important in anion
binding and conduction. J. Gen. Physiol. 123 (4), 327–340.
Qu, Z., Chien, L.T., Cui, Y., Hartzell, H.C., 2006a. The anion-selective
pore of the bestrophins, a family of chloride channels associated with
retinal degeneration. J. Neurosci. 26 (20), 5411–5419.
Qu, Z., Cui, Y., Hartzell, C., 2006b. A short motif in the C-terminus of
mouse bestrophin 4 inhibits its activation as a Cl channel. FEBS Lett.
580 (8), 2141–2146.
Quinn, R.H., Miller, S.S., 1992. Ion transport mechanisms in native
human retinal pigment epithelium. Invest. Ophthalmol. Vis. Sci. 33
(13), 3513–3527.
Quinn, R.H., Quong, J.N., Miller, S.S., 2001. Adrenergic receptor
activated ion transport in human fetal retinal pigment epithelium.
Invest. Ophthalmol. Vis. Sci. 42 (1), 255–264.
Ramsey, I.S., Delling, M., Clapham, D.E., 2005. An Introduction to TRP
Channels. Annu. Rev. Physiol.
Reigada, D., Mitchell, C.H., 2005. Release of ATP from retinal pigment
epithelial cells involves both CFTR and vesicular transport. Am. J.
Physiol. Cell Physiol. 288 (1), C132–C140.
Reigada, D., Lu, W., Zhang, X., Friedman, C., Pendrak, K., McGlinn, A.,
Stone, R.A., Laties, A.M., Mitchell, C.H., 2005. Degradation of
extracellular ATP by the retinal pigment epithelium. Am. J. Physiol.
Cell Physiol. 289 (3), C617–C624.
Reigada, D., Lu, W., Mitchell, C.H., 2006. Glutamate acts at NMDA
receptors on fresh bovine and on cultured human retinal pigment
epithelial cells to trigger release of ATP. J. Physiol. 575 (Pt 3), 707–720.
Renner, A.B., Tillack, H., Kraus, H., Kramer, F., Mohr, N., Weber, B.H.,
Foerster, M.H., Kellner, U., 2005. Late onset is common in best
macular dystrophy associated with VMD2 gene mutations. Ophthalmology 112 (4), 586–592.
Riordan, J.R., Rommens, J.M., Kerem, B., Alon, N., Rozmahel, R.,
Grzelczak, Z., Zielenski, J., Lok, S., Plavsic, N., Chou, J.L., et al.,
1989. Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 245 (4922), 1066–1073.
Robbins, J., Ikeda, H., 1989. Benzodiazepines and the mammalian retina.
I. Autoradiographic localisation of receptor sites and the lack of effect
on the electroretinogram. Brain Res. 479 (2), 313–322.
Roberts, W.M., Jacobs, R.A., Hudspeth, A.J., 1990. Colocalization of ion
channels involved in frequency selectivity and synaptic transmission at
presynaptic active zones of hair cells. J. Neurosci. 10 (11), 3664–3684.
Rosenthal, R., Strauss, O., 2002. Ca2+-channels in the RPE. Adv. Exp.
Med. Biol. 514, 225–235.
Rosenthal, R., Thieme, H., Strauss, O., 2001. Fibroblast growth factor
receptor 2 (FGFR2) in brain neurons and retinal pigment epithelial
cells act via stimulation of neuroendocrine L-type channels (Ca(v)1.3).
FASEB J. 15 (6), 970–977.
Rosenthal, R., Wohlleben, H., Malek, G., Schlichting, L., Thieme, H.,
Bowes Rickman, C., Strauss, O., 2004. Insulin-like growth factor-1
contributes to neovascularization in age-related macular degeneration.
Biochem. Biophys. Res. Commun. 323 (4), 1203–1208.
Rosenthal, R., Malek, G., Salomon, N., Peill-Meininghaus, M., Coeppicus, L., Wohlleben, H., Wimmers, S., Bowes Rickman, C., Strauss, O.,
2005. The fibroblast growth factor receptors, FGFR-1 and FGFR-2,
mediate two independent signalling pathways in human retinal
pigment epithelial cells. Biochem. Biophys. Res. Commun. 337 (1),
241–247.
Rosenthal, R., Bakall, B., Kinnick, T., Peachey, N., Wimmers, S.,
Wadelius, C., Marmorstein, A., Strauss, O., 2006. Expression of
bestrophin-1, the product of the VMD2 gene, modulates voltagedependent Ca2+ channels in retinal pigment epithelial cells. FASEB J.
20 (1), 178–180.
Ryan, J.S., Baldridge, W.H., Kelly, M.E., 1999. Purinergic regulation of
cation conductances and intracellular Ca2+ in cultured rat retinal
pigment epithelial cells. J. Physiol. 520 (Part 3), 745–759.
Ryan, J.S., Kelly, M.E., 1998. Activation of a nonspecific cation current in
rat cultured retinal pigment epithelial cells: involvement of a G(alpha i)
subunit protein and the mitogen-activated protein kinase signalling
pathway. Br. J. Pharmacol. 124 (6), 1115–1122.
Rymer, J., Miller, S.S., Edelman, J.L., 2001. Epinephrine-induced
increases in [Ca2+](in) and KCl-coupled fluid absorption in bovine
RPE. Invest. Ophthalmol. Vis. Sci. 42 (8), 1921–1929.
Sakai, H., Saito, T., 1997. Na+ and Ca2+ channel expression in cultured
newt retinal pigment epithelial cells: comparison with neuronal types
of ion channels. J. Neurobiol. 32 (4), 377–390.
Sakmann, B., Neher, E., 1984. Patch clamp techniques for studying ionic
channels in excitable membranes. Annu. Rev. Physiol. 46, 455–472.
Sakura, H., Ammala, C., Smith, P.A., Gribble, F.M., Ashcroft, F.M.,
1995. Cloning and functional expression of the cDNA encoding a
novel ATP-sensitive potassium channel subunit expressed in pancreatic
beta-cells, brain, heart and skeletal muscle. FEBS Lett. 377 (3),
338–344.
Salceda, R., 1989. 45Ca uptake by retinal pigment epithelial cells. Invest.
Ophthalmol. Vis. Sci. 30 (10), 2114–2117.
Salceda, R., Riesgo-Escovar, J.R., 1990. Characterization of calcium
uptake in chick retinal pigment epithelium. Pigment Cell Res. 3 (3),
141–145.
Salceda, R., Sanchez-Chavez, G., 2000. Calcium uptake, release and
ryanodine binding in melanosomes from retinal pigment epithelium.
Cell Calcium 27 (4), 223–229.
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Santarelli, L.C., Wassef, R., Heinemann, S.H., Hoshi, T., 2006. Three
methionine residues located within the regulator of conductance for
K+ (RCK) domains confer oxidative sensitivity to large-conductance
Ca2+-activated K+ channels. J. Physiol. 571 (Part 2), 329–348.
Sardini, A., Amey, J.S., Weylandt, K.H., Nobles, M., Valverde, M.A.,
Higgins, C.F., 2003. Cell volume regulation and swelling-activated
chloride channels. Biochim. Biophys. Acta 1618 (2), 153–162.
Saris, N.E., Carafoli, E., 2005. A historical review of cellular calcium
handling, with emphasis on mitochondria. Biochemistry (Mosc) 70 (2),
187–194.
Sarkadi, B., Parker, J.C., 1991. Activation of ion transport pathways by
changes in cell volume. Biochim. Biophys. Acta 1071 (4), 407–427.
Satin, L.S., 2000. Localized calcium influx in pancreatic beta-cells: its
significance for Ca2+-dependent insulin secretion from the islets of
Langerhans. Endocrine 13 (3), 251–262.
Scheel, O., Zdebik, A.A., Lourdel, S., Jentsch, T.J., 2005. Voltagedependent electrogenic chloride/proton exchange by endosomal CLC
proteins. Nature 436 (7049), 424–427.
Scholze, A., Plant, T.D., Dolphin, A.C., Nurnberg, B., 2001. Functional
expression and characterization of a voltage-gated CaV1.3 (alpha1D)
calcium channel subunit from an insulin-secreting cell line. Mol.
Endocrinol. 15 (7), 1211–1221.
Scott, R.H., Sutton, K.G., Griffin, A., Stapleton, S.R., Currie, K.P., 1995.
Aspects of calcium-activated chloride currents: a neuronal perspective.
Pharmacol. Ther. 66 (3), 535–565.
Segawa, Y., Hughes, B.A., 1994. Properties of the inwardly rectifying K+
conductance in the toad retinal pigment epithelium. J. Physiol. 476 (1),
41–53.
Shen, Z., Marcus, D.C., 1998. Divalent cations inhibit IsK/KvLQT1
channels in excised membrane patches of strial marginal cells. Hear
Res. 123 (1 and 2), 157–167.
Sheu, S.J., Wu, S.N., 2003. Mechanism of inhibitory actions of oxidizing
agents on calcium-activated potassium current in cultured pigment
epithelial cells of the human retina. Invest. Ophthalmol. Vis. Sci. 44
(3), 1237–1244.
Sheu, S.J., Wu, S.N., Hu, D.N., Chen, J.F., 2004. The influence of
hypotonicity on large-conductance calcium-activated potassium channels in human retinal pigment epithelial cells. J. Ocul. Pharmacol.
Ther. 20 (6), 563–575.
Sheu, S.J., Wu, S.N., Hu, D.N., 2005. Stretch-stimulated activity of large
conductance calcium-activated potassium channels in human retinal
pigment epithelial cells. J. Ocul. Pharmacol. Ther. 21 (6), 429–435.
Shimura, M., Yuan, Y., Chang, J.T., Zhang, S., Campochiaro, P.A.,
Zack, D.J., Hughes, B.A., 2001. Expression and permeation properties
of the K(+) channel Kir7.1 in the retinal pigment epithelium.
J. Physiol. 531 (Part 2), 329–346.
Shirao, Y., Steinberg, R.H., Griff, E.R., 1987. K+-modulation of Na+/
K+ ATPase of the frog retinal pigment epithelium. Neurosci. Res.
(Suppl. 6), S1–S13.
Shuttleworth, T.J., 1997. Intracellular Ca2+ signalling in secretory cells.
J. Exp. Biol. 200 (Part 2), 303–314.
Sigworth, F.J., 1986. The patch clamp is more useful than anyone had
expected. Fed. Proc. 45 (12), 2673–2677.
Slomiany, M.G., Rosenzweig, S.A., 2004a. Autocrine effects of IGF-1
induced VEGF and IGFBP-3 secretion in retinal pigment epithelial cell
line ARPE-19. Am. J. Physiol. Cell Physiol. 287 (3), C746–C753.
Slomiany, M.G., Rosenzweig, S.A., 2004b. IGF-1-induced VEGF and
IGFBP-3 secretion correlates with increased HIF-1 alpha expression
and activity in retinal pigment epithelial cell line D407. Invest.
Ophthalmol. Vis. Sci. 45 (8), 2838–2847.
Soltoff, S.P., McMillian, M.K., Talamo, B.R., Cantley, L.C., 1993.
Blockade of ATP binding site of P2 purinoceptors in rat parotid acinar
cells by isothiocyanate compounds. Biochem. Pharmacol. 45 (9),
1936–1940.
Spauschus, A., Lentes, K.U., Wischmeyer, E., Dissmann, E., Karschin,
C., Karschin, A., 1996. A G-protein-activated inwardly rectifying K+
channel (GIRK4) from human hippocampus associates with other
GIRK channels. J. Neurosci. 16 (3), 930–938.
299
Spring, K.R., 1998. Routes and mechanism of fluid transport by epithelia.
Annu. Rev. Physiol. 60, 105–119.
Spring, K.R., Ericson, A.C., 1982. Epithelial cell volume modulation and
regulation. J. Membr. Biol. 69 (3), 167–176.
Stalmans, P., Himpens, B., 1997. Confocal imaging of Ca2+ signaling in
cultured rat retinal pigment epithelial cells during mechanical and
pharmacologic stimulation. Invest. Ophthalmol. Vis. Sci. 38 (1),
176–187.
Stamer, W.D., Bok, D., Hu, J., Jaffe, G.J., McKay, B.S., 2003.
Aquaporin-1 channels in human retinal pigment epithelium: role in
transepithelial water movement. Invest. Ophthalmol. Vis. Sci. 44 (6),
2803–2808.
Stanton, J.B., Goldberg, A.F., Hoppe, G., Marmorstein, L.Y., Marmorstein, A.D., 2006. Hydrodynamic properties of porcine bestrophin-1 in
Triton X-100. Biochim. Biophys. Acta 1758 (2), 241–247.
Steele, F.R., Chader, G.J., Johnson, L.V., Tombran-Tink, J., 1992.
Pigment epithelium-derived factor: neurotrophic activity and identification as a member of the serine protease inhibitor gene family. Proc.
Natl. Acad. Sci. USA 90, 1526–1530.
Steinberg, R.H., 1985. Interactions between the retinal pigment epithelium
and the neural retina. Doc. Ophthalmol. 60 (4), 327–346.
Steinberg, R.H., Miller, S., 1973. Aspects of electrolyte transport in frog
pigment epithelium. Exp. Eye Res. 16 (5), 365–372.
Steinberg, R.H., Schmidt, R., Brown, K.T., 1970. Intracellular responses
to light from cat pigment epithelium: origin of the electroretinogram
c-wave. Nature 227 (5259), 728–730.
Steinberg, R.H., Miller, S.S., Stern, W.H., 1978. Initial observations on
the isolated retinal pigment epithelium-choroid of the cat. Invest.
Ophthalmol. Vis. Sci. 17 (7), 675–678.
Steinberg, R.H., Oakley II, B., Niemeyer, G., 1980. Light-evoked changes
in [K+]0 in retina of intact cat eye. J. Neurophysiol. 44 (5), 897–921.
Steinberg, R.H., Linsenmeier, R.A., Griff, E.R., 1983. Three light-evoked
responses of the retinal pigment epithelium. Vision Res. 23 (11),
1315–1323.
Stobrawa, S.M., Breiderhoff, T., Takamori, S., Engel, D., Schweizer, M.,
Zdebik, A.A., Bosl, M.R., Ruether, K., Jahn, H., Draguhn, A., Jahn,
R., Jentsch, T.J., 2001. Disruption of ClC-3, a chloride channel
expressed on synaptic vesicles, leads to a loss of the hippocampus.
Neuron 29 (1), 185–196.
Stohr, H., Marquardt, A., Nanda, I., Schmid, M., Weber, B.H., 2002.
Three novel human VMD2-like genes are members of the evolutionary
highly conserved RFP-TM family. Eur. J. Hum. Genet. 10 (4),
281–284.
Stohr, H., Milenkowic, V., Weber, B.H., 2005. VMD2 and its role in
Best’s disease and other retinopathies. Ophthalmologe 102 (2),
116–121.
Strange, K., 2004. Cellular volume homeostasis. Adv. Physiol. Educ. 28
(1–4), 155–159.
Strange, K., Emma, F., Jackson, P.S., 1996. Cellular and molecular
physiology of volume-sensitive anion channels. Am. J. Physiol. 270
(3 Pt 1), C711–C730.
Strauss, O., 2005. The retinal pigment epithelium in visual function.
Physiol. Rev. 85 (3), 845–881.
Strauss, O., Richard, G., Wienrich, M., 1993. Voltage-dependent
potassium currents in cultured human retinal pigment epithelial cells.
Biochem. Biophys. Res. Commun. 191 (3), 775–781.
Strauss, O., Weiser, T., Wienrich, M., 1994. Potassium currents in cultured
cells of the rat retinal pigment epithelium. Comp. Biochem. Physiol. A
Physiol. 109 (4), 975–983.
Strauss, O., Wiederholt, M., Wienrich, M., 1996. Activation of Cl
currents in cultured rat retinal pigment epithelial cells by intracellular
applications of inositol-1,4,5-triphosphate: differences between rats
with retinal dystrophy (RCS) and normal rats. J. Membr. Biol. 151 (2),
189–200.
Strauss, O., Mergler, S., Wiederholt, M., 1997. Regulation of L-type
calcium channels by protein tyrosine kinase and protein kinase C in
cultured rat and human retinal pigment epithelial cells. FASEB J 11
(11), 859–867.
ARTICLE IN PRESS
300
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Strauss, O., Steinhausen, K., Wienrich, M., Wiederholt, M., 1998a.
Activation of a Cl-conductance by protein kinase-dependent phosphorylation in cultured rat retinal pigment epithelial cells. Exp. Eye
Res. 66 (1), 35–42.
Strauss, O., Stumpff, F., Mergler, S., Wienrich, M., Wiederholt, M.,
1998b. The Royal College of Surgeons rat: an animal model for
inherited retinal degeneration with a still unknown genetic defect. Acta
Anat. (Basel) 162 (2–3), 101–111.
Strauss, O., Steinhausen, K., Mergler, S., Stumpff, F., Wiederholt, M.,
1999. Involvement of protein tyrosine kinase in the InsP3-induced
activation of Ca2+-dependent Cl currents in cultured cells of the rat
retinal pigment epithelium. J. Membr. Biol. 169 (3), 141–153.
Strauss, O., Buss, F., Rosenthal, R., Fischer, D., Mergler, S., Stumpff, F.,
Thieme, H., 2000. Activation of neuroendocrine L-type channels
(alpha1D subunits) in retinal pigment epithelial cells and brain neurons
by pp60 (c-src). Biochem. Biophys. Res. Commun. 270 (3), 806–810.
Strauss, O., Rosenthal, R., Dey, D., Beninde, J., Wollmann, G., Thieme,
H., Wiederholt, M., 2002. Effects of protein kinase C on delayed
rectifier K+ channel regulation by tyrosine kinase in rat retinal
pigment epithelial cells. Invest. Ophthalmol. Vis. Sci. 43 (5),
1645–1654.
Strauss, O., Heimann, H., Foerster, M.H., Agostini, H., Hansen, L.L.,
Rosenthal, R., 2003. Activation of L-type Ca2+ Channels is Necessary
for Growth Factor-dependent Stimulation of VEGF Secretion by RPE
Cells. Invest. Ophthalmol. Vis. Sci. (44) e-abstract 3926.
StrauX, O., Wienrich, M., 1994. Ca2+-conductances in cultured rat retinal
pigment epithelial cells. J. Cell Physiol. 160, 89–96.
Streilein, J.W., 2003. Ocular immune privilege: therapeutic opportunities
from an experiment of nature. Nat. Rev. Immunol. 3 (11), 879–889.
Streilein, J.W., Ma, N., Wenkel, H., Ng, T.F., Zamiri, P., 2002.
Immunobiology and privilege of neuronal retina and pigment
epithelium transplants. Vis. Res. 42 (4), 487–495.
Striessnig, J., 1999. Pharmacology, structure and function of cardiac
L-type Ca2+ channels. Cell Physiol. Biochem. 9 (4–5), 242–269.
Striessnig, J., Hoda, J.C., Koschak, A., Zaghetto, F., Mullner, C.,
Sinnegger-Brauns, M.J., Wild, C., Watschinger, K., Trockenbacher,
A., Pelster, G., 2004. L-type Ca2+ channels in Ca2+ channelopathies.
Biochem. Biophys. Res. Commun. 322 (4), 1341–1346.
Sugasawa, K., Deguchi, J., Okami, T., Yamamoto, A., Omori, K.,
Uyama, M., Tashiro, Y., 1994. Immunocytochemical analyses of
distributions of Na, K-ATPase and GLUT1, insulin and transferrin
receptors in the developing retinal pigment epithelial cells. Cell Struct.
Funct. 19 (1), 21–28.
Sullivan, D.M., Erb, L., Anglade, E., Weisman, G.A., Turner, J.T., Csaky,
K.G., 1997. Identification and characterization of P2Y2 nucleotide
receptors in human retinal pigment epithelial cells. J. Neurosci. Res. 49
(1), 43–52.
Sumita, R., 1961. The fine structure of Bruch’s membrane in the choroid.
Acta Soc. Ophthalmol. Jpn. 28, 1188.
Sun, H., Tsunenari, T., Yau, K.W., Nathans, J., 2002. The vitelliform
macular dystrophy protein defines a new family of chloride channels.
Proc. Natl. Acad. Sci. USA 99 (6), 4008–4013.
Sundelin, S., Wihlmark, U., Nilsson, S.E., Brunk, U.T., 1998. Lipofuscin
accumulation in cultured retinal pigment epithelial cells reduces their
phagocytic capacity. Curr. Eye Res. 17 (8), 851–857.
Sundelin, S.P., Terman, A., 2002. Different effects of chloroquine and
hydroxychloroquine on lysosomal function in cultured retinal pigment
epithelial cells. Apmis 110 (6), 481–489.
Sunose, H., Liu, J., Marcus, D.C., 1997. cAMP increases K+ secretion via
activation of apical IsK/KvLQT1 channels in strial marginal cells.
Hear Res. 114 (1 and 2), 107–116.
Suzuki, M., Morita, T., Iwamoto, T., 2006. Diversity of Cl() channels.
Cell Mol. Life Sci. 63 (1), 12–24.
Takahashi, N., Morishige, K., Jahangir, A., Yamada, M., Findlay, I.,
Koyama, H., Kurachi, Y., 1994. Molecular cloning and functional
expression of cDNA encoding a second class of inward rectifier
potassium channels in the mouse brain. J. Biol. Chem. 269 (37),
23274–23279.
Takahira, M., Hughes, B.A., 1997. Isolated bovine retinal pigment
epithelial cells express delayed rectifier type and M-type K+ currents.
Am. J. Physiol. 273 (3 Part 1), C790–C803.
Tanito, M., Nishiyama, A., Tanaka, T., Masutani, H., Nakamura, H.,
Yodoi, J., Ohira, A., 2002. Change of redox status and modulation by
thiol replenishment in retinal photooxidative damage. Invest. Ophthalmol. Vis. Sci. 43 (7), 2392–2400.
Tao, Q., Kelly, M.E., 1996. Calcium-activated potassium current in
cultured rabbit retinal pigment epithelial cells. Curr. Eye Res. 15 (3),
237–246.
Tao, Q., Rafuse, P.E., Kelly, M.E., 1994. Potassium currents in cultured
rabbit retinal pigment epithelial cells. J. Membr. Biol. 141 (2),
123–138.
Thiagarajah, J.R., Verkman, A.S., 2003. CFTR pharmacology and its role
in intestinal fluid secretion. Curr. Opin. Pharmacol. 3 (6), 594–599.
Thompson, D.A., Gal, A., 2003. Vitamin A metabolism in the retinal
pigment epithelium: genes, mutations, and diseases. Prog. Retin. Eye
Res. 22, 683–703.
Toimela, T., Salminen, L., Tahti, H., 1998. Effects of tamoxifen,
toremifene and chloroquine on the lysosomal enzymes in cultured
retinal pigment epithelial cells. Pharmacol. Toxicol. 83 (6), 246–251.
Topert, C., Doring, F., Wischmeyer, E., Karschin, C., Brockhaus, J.,
Ballanyi, K., Derst, C., Karschin, A., 1998. Kir2.4: a novel K+ inward
rectifier channel associated with motoneurons of cranial nerve nuclei.
J. Neurosci. 18 (11), 4096–4105.
Tsuboi, S., Pederson, J.E., 1988a. Effect of plasma osmolality and
intraocular pressure on fluid movement across the blood-retinal
barrier. Invest. Ophthalmol. Vis. Sci. 29 (11), 1747–1749.
Tsuboi, S., Pederson, J.E., 1988b. Volume flow across the isolated retinal
pigment epithelium of cynomolgus monkey eyes. Invest. Ophthalmol.
Vis. Sci. 29 (11), 1652–1655.
Tsuboi, S., Manabe, R., Iizuka, S., 1986. Aspects of electrolyte transport
across isolated dog retinal pigment epithelium. Am. J. Physiol. 250
(5 Pt 2), F781–F784.
Tsunenari, T., Sun, H., Williams, J., Cahill, H., Smallwood, P., Yau,
K.W., Nathans, J., 2003. Structure-function analysis of the bestrophin
family of anion channels. J. Biol. Chem. 278 (42), 41114–41125.
Tsunenari, T., Nathans, J., Yau, K.W., 2006. Ca2+-activated Cl current
from human bestrophin-4 in excised membrane patches. J. Gen.
Physiol. 127 (6), 749–754.
Turnheim, K., Plass, H., Wyskovsky, W., 2002. Basolateral potassium
channels of rabbit colon epithelium: role in sodium absorption and
chloride secretion. Biochim. Biophys. Acta 1560 (1 and 2), 51–66.
Uchida, N., Kiuchi, Y., Miyamoto, K., Uchida, J., Tobe, T., Tomita, M.,
Shioda, S., Nakai, Y., Koide, R., Oguchi, K., 1998. Glutamatestimulated proliferation of rat retinal pigment epithelial cells. Eur. J.
Pharmacol. 343 (2 and 3), 265–273.
Ueda, Y., Steinberg, R.H., 1993. Voltage-operated calcium channels in
fresh and cultured rat retinal pigment epithelial cells. Invest.
Ophthalmol. Vis. Sci. 34 (12), 3408–3418.
Ueda, Y., Steinberg, R.H., 1994. Chloride currents in freshly isolated rat
retinal pigment epithelial cells. Exp. Eye Res. 58 (3), 331–342.
Ueda, Y., Steinberg, R.H., 1995. Dihydropyridine-sensitive calcium
currents in freshly isolated human and monkey retinal pigment
epithelial cells. Invest. Ophthalmol. Vis. Sci. 36 (2), 373–380.
Uehara, F., Matthes, M.T., Yasumura, D., LaVail, M.M., 1990. Lightevoked changes in the interphotoreceptor matrix. Science 248 (4963),
1633–1636.
Ulshafer, R.J., Allen, C.B., Rubin, M.L., 1990. Distributions of elements
in the human retinal pigment epithelium. Arch. Ophthalmol. 108 (1),
113–117.
van Best, J.A., Putting, B.J., Oosterhuis, J.A., Zweypfenning, R.C.,
Vrensen, G.F., 1997. Function and morphology of the retinal pigment
epithelium after light-induced damage. Microsc. Res. Technol. 36 (2),
77–88.
Vergani, P., Basso, C., Mense, M., Nairn, A.C., Gadsby, D.C., 2005.
Control of the CFTR channel’s gates. Biochem. Soc. Trans. 33 (Pt 5),
1003–1007.
ARTICLE IN PRESS
S. Wimmers et al. / Progress in Retinal and Eye Research 26 (2007) 263–301
Vergara, C., Latorre, R., Marrion, N.V., Adelman, J.P., 1998. Calciumactivated potassium channels. Curr. Opin. Neurobiol. 8 (3), 321–329.
Verkman, A.S., 2003. Role of aquaporin water channels in eye function.
Exp. Eye Res. 76 (2), 137–143.
Wabbels, B., Preising, M.N., Kretschmann, U., Demmler, A., Lorenz, B.,
2006. Genotype-phenotype correlation and longitudinal course in ten
families with Best vitelliform macular dystrophy. Graefes Arch. Clin.
Exp. Ophthalmol. 244, 1453–1466.
Wabbels, B.K., Demmler, A., Preising, M., Lorenz, B., 2004. Fundus
autofluorescence in patients with genetically determined Best vitelliform macular dystrophy: Evaluation of genotype-phenotype correlation and longitudinal course. Invest. Ophthalmol. Vis. Sci. 45
e-abstract 1762.
Warth, R., 2003. Potassium channels in epithelial transport. Pflugers
Arch. 446 (5), 505–513.
Warth, R., Barhanin, J., 2003. Function of K+ channels in the intestinal
epithelium. J. Membr. Biol. 193 (2), 67–78.
Wei, A.D., Gutman, G.A., Aldrich, R., Chandy, K.G., Grissmer, S.,
Wulff, H., 2005. International Union of Pharmacology. LII. Nomenclature and molecular relationships of calcium-activated potassium
channels. Pharmacol. Rev. 57 (4), 463–472.
Weiger, T.M., Hermann, A., Levitan, I.B., 2002. Modulation of calciumactivated potassium channels. J. Comp. Physiol. A Neuroethol. Sens.
Neural Behav. Physiol. 188 (2), 79–87.
Weingeist, T.A., Kobrin, J.L., Watzke, R.C., 1982. Histopathology of
Best’s macular dystrophy. Arch. Ophthalmol. 100 (7), 1108–1114.
Wen, R., Lui, G.M., Steinberg, R.H., 1993. Whole-cell K+ currents in
fresh and cultured cells of the human and monkey retinal pigment
epithelium. J. Physiol. 465, 121–147.
Wen, R., Lui, G.M., Steinberg, R.H., 1994. Expression of a tetrodotoxinsensitive Na+ current in cultured human retinal pigment epithelial
cells. J. Physiol. 476 (2), 187–196.
Weng, T.X., Godley, B.F., Jin, G.F., Mangini, N.J., Kennedy, B.G., Yu,
A.S., Wills, N.K., 2002. Oxidant and antioxidant modulation of
chloride channels expressed in human retinal pigment epithelium. Am.
J. Physiol. Cell Physiol. 283 (3), C839–C849.
Wenkel, H., Streilein, J.W., 2000. Evidence that retinal pigment epithelium
functions as an immune-privileged tissue. Invest. Ophthalmol. Vis. Sci.
41 (11), 3467–3473.
Wettschureck, N., Offermanns, S., 2005. Mammalian G proteins and their
cell type specific functions. Physiol. Rev. 85 (4), 1159–1204.
Wettstein, M., Peters-Regehr, T., Kubitz, R., Fischer, R., Holneicher, C.,
Monnighoff, I., Haussinger, D., 2000. Release of osmolytes induced by
phagocytosis and hormones in rat liver. Am. J. Physiol. Gastrointest.
Liver Physiol. 278 (2), G227–G233.
White, K., Marquardt, A., Weber, B.H., 2000. VMD2 mutations in
vitelliform macular dystrophy (Best disease) and other maculopathies.
Hum. Mutat. 15 (4), 301–308.
Williams, C.D., Rizzolo, L.J., 1997. Remodeling of junctional complexes
during the development of the outer blood-retinal barrier. Anat. Rec.
249 (3), 380–388.
Williams, R.J., 1974. Calcium ions: their ligands and their functions.
Biochem. Soc. Symp. (39), 133–138.
301
Williams, R.J., 1994. Calcium-binding proteins in normal and transformed
cells. Cell Calcium 16 (5), 339–346.
Wills, N.K., Weng, T., Mo, L., Hellmich, H.L., Yu, A., Wang, T.,
Buchheit, S., Godley, B.F., 2000. Chloride channel expression in
cultured human fetal RPE cells: response to oxidative stress. Invest.
Ophthalmol. Vis. Sci. 41 (13), 4247–4255.
Winkler, B.S., 1995. A quantitative assessment of glucose metabolism in
the isolated rat retina. In: Christen, Y., Doly, M., M.T, D.-L. (Eds.),
Les Seminares Ophthalmologiques d’IPSEN. Vision et Adaptation,
vol. 6. Elsevier, Paris, pp. 79–96.
Witmer, A.N., Vrensen, G.F., Van Noorden, C.J., Schlingemann, R.O.,
2003. Vascular endothelial growth factors and angiogenesis in eye
disease. Prog. Retin. Eye Res. 22 (1), 1–29.
Wollmann, G., Lenzner, S., Berger, W., Rosenthal, R., Karl, M.O.,
Strauss, O., 2006. Voltage-dependent ion channels in the mouse
RPE: comparison with Norrie disease mice. Vis. Res. 46 (5),
688–698.
Wood, J.P., Osborne, N.N., 1996. Immunological localization of
GABAA-receptor subunits in cultured human and rat retinal pigment
epithelium. Exp. Eye Res. 63 (1), 113–116.
Wu, J., Marmorstein, A.D., Peachey, N.S., 2006. Functional abnormalities in the retinal pigment epithelium of CFTR mutant mice. Exp. Eye
Res. 83, 424–428.
Yang, D., Pan, A., Swaminathan, A., Kumar, G., Hughes, B.A., 2003.
Expression and localization of the inwardly rectifying potassium
channel Kir7.1 in native bovine retinal pigment epithelium. Invest.
Ophthalmol. Vis. Sci. 44 (7), 3178–3185.
Yang, Z., Xu, H., Cui, N., Qu, Z., Chanchevalap, S., Shen, W., Jiang, C.,
2000. Biophysical and molecular mechanisms underlying the modulation of heteromeric Kir4.1–Kir5.1 channels by CO2 and pH. J. Gen.
Physiol. 116 (1), 33–45.
Yellen, G., 2002. The voltage-gated potassium channels and their relatives.
Nature 419 (6902), 35–42.
Yu, F.H., Yarov-Yarovoy, V., Gutman, G.A., Catterall, W.A., 2005.
Overview of molecular relationships in the voltage-gated ion channel
superfamily. Pharmacol. Rev. 57 (4), 387–395.
Yuan, Y., Shimura, M., Hughes, B.A., 2003. Regulation of inwardly
rectifying K+ channels in retinal pigment epithelial cells by
intracellular pH. J. Physiol. 549 (Part 2), 429–438.
Zarbin, M.A., Anholt, R.R., 1991. Benzodiazepine receptors in the eye.
Invest. Ophthalmol. Vis. Sci. 32 (9), 2579–2587.
Zdebik, A.A., Cuffe, J.E., Bertog, M., Korbmacher, C., Jentsch, T.J.,
2004. Additional disruption of the ClC-2 Cl() channel does not
exacerbate the cystic fibrosis phenotype of cystic fibrosis transmembrane conductance regulator mouse models. J. Biol. Chem. 279 (21),
22276–22283.
Zhao, S., Rizzolo, L.J., Barnstable, C.J., 1997. Differentiation and
transdifferentiation of the retinal pigment epithelium. Int. Rev. Cytol.
171, 225–266.
Ziganshin, A.U., Ziganshina, L.E., King, B.F., Pintor, J., Burnstock, G.,
1996. Effects of P2-purinoceptor antagonists on degradation of
adenine nucleotides by ecto-nucleotidases in folliculated oocytes of
Xenopus laevis. Biochem. Pharmacol. 51 (7), 897–901.