Silica in rubber – Possibilities and Challenges
advertisement

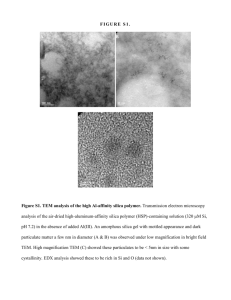
Silica in rubber – Possibilities and Challenges Joint seminar Kumi-instituutti & MOL 1 Silica in rubber – Possibilities and Challenges 2 Tampere Wilma K. Dierkes Tampere University of Technology Department of Material Science Laboratory of Plastics and Elastomer Technology 33720 Tampere Finland University of Twente Department of Elastomer Technology and Engineering 7500 AE Enschede The Netherlands Enschede 3 Visiting Professorship Education 1991 1993 2005 Fondation Universitaire University of Hanover (Germany): Luxembourgeoise, Arlon (Belgium), postacademical education: ‘Diplom Chemie’ European Environmental Sciences University Twente, Department of Rubber Technology, Enschede (Netherlands), PhD: ‘Economic mixing of silicarubber compounds’ Profession 1991 1993 1999 2001 Degussa University Twente, Rubber Resources, Robert Bosch Belgium: Enschede: the Netherlands: R&D, Produktie Researcher Technical Service Ass. Professor (Belgium): R&D, Production 2009 Tampere University of Technology, Finland: Visiting Professor 4 Contents Silica in rubber Silica/silanes in rubber Challenges Possibilities Coupling agents Surface Dual phase modification silica Summary In-situ Silica Particle size distribution Silica in rubber 5 History 1950 Silica and silicates as replacement of carbon black 1970 Precipitated silica (lower costs than pyrogenic silica) First applications: shoe soles (colored, transparent) 1990 Silica in heavyservice tires (improvement of cutting and chipping resistance, better adhesion to textile and metal), 10-25 phr silica 2010 Silica-silane technology for passenger car tire treads ? Rolling resistance Abrasion resistance Wet grip Current silica consumption in rubber: app. 0,5 mill. tons/year Silica/silanes in rubber 6 History 1970 Organofunctional silanes as adhesion promoters for glass fiber – polymer systems Introduction of bifunctional silanes as coupling agents 1990 F. Thurn, S. Wolff (KGK 28, 733ff, 1975): best silanes for highest reinforcing effect and minimal influence on scorch Bis-(triethoxysilyl propyl)tetra- and disulfides as coupling agents Combinations: sulfursilanemoieties -SCN -S-CS-OR -S-CS-NHR -S-C(NH)(NH2) -SH -S-S-S-S-S-S-S-S-S- -Si(OCH3) 3 -Si(OC2H5) 3 -Si(i-OC3H7) 3 -Si(n-OC3H7) 3 -Si(CO4H9) 3 -Si(OC8H17) 3 Bis(triethoxysilylpropyl)disulfane 7 Silica in rubber Silica/silanes in rubber Challenges Possibilities Coupling agents Surface Dual phase modification silica Summary In-situ Silica Particle size distribution 8 Challenges Dispersion Particle size distribution & structure Compound stability Torque Silica filled rubber blend Flocculation Good dispersion Polymer 1 Time Silica Polymer 2 Compatibility & polarity match Polymers Fillers EPDM SBR NR NBR 16.4 16.6 17.5 19.7 Carbon black 70.0 Surface tension [mJ/m2] Silica Challenges 9 Dispersion & particle size distribution Filler dispersion depends on: Polymer: Type, molecular weight Filler: Particle size distribution, structure, surface activity Blend: A. Schröder, M. Klüppel, R. H. Schuster and J. Heidberg, Kautsch. Gummi Kunstst., 53, 257 (2000). Interphase transfer of fillers T.A. Vilgis, Polymer, 46(12), 4223 (2005) Challenges 10 Dispersion EPDM with untreated silica S-SBR with untreated silica NBR with untreated silica Silane treated silica reinforced NBR C2H2-plasma treated silica reinforced NBR (H5C2-O)3Si – C3H6 – Sn – C3H6 – Si (OC2H5)3 Image size: 2,5 µ Challenges 11 Polarity & compatibility match Polymers Curing agents EPDM SBR NR NBR CBS DCBS MBT 16.4 16.6 17.5 19.7 20.4 21.8 22.5 Fillers Carbon black Silica 70.0 Surface tension [mJ/m2] Incompatibility of the components (polarity, unsaturation) Dispersion and wetting difficulties of the filler Re-agglomeration of the filler Low adhesion of polymer on the filler surface Challenges 12 Torque Compound stability Flocculation Good dispersion Time Re-agglomeration of the filler (Viscosity) changes during storage 13 Silica in rubber Silica/silanes in rubber Challenges Possibilities Coupling agents Surface Dual phase modification silica Summary In-situ Silica Particle size distribution Possibilities: Coupling agent 14 Polymer-filler incompatibility OH Si EtO O O Si O Silica (CH ) S S 23 Si O O Si (CH ) S S 23 particle O O S Si O Si (CH ) S O 23 Si OEt OEt O O Si Si OH (CH ) S EtO 23 S Silica: hydrophilic Si Rubber: hydrophobic Possibilities: Coupling agent Different types of coupling agents Structure of different coupling agents Blocked silane S-[3-(triethoxysilyl)propyl]ester of octanethionic acid (EtO)3Si - (CH2)3 - S - (CO) - (CH2)6 - CH3 NXT Silane, GE Silicones, Wilton, CT, USA [ ]n Oligomeric silane (+ activator) Bis(triethoxysilyl)polybutadiene, n = 27 (EtO) 3 Si - (CH 2 -CH=CH -CH 2 )n - Si(EtO)3 Rhenofit 1715, Rheinchemie, Mannheim, Germany Monomeric silane Bis(triethoxysilylpropyl)disulfane (TESPD) (EtO)3Si - (CH2)3 - S-S - (CH2)3 - Si(EtO)3 Si 266 Evonik (Degussa), Frankfurt, Germany 15 Possibilities: Coupling agent 16 Different types of coupling agents TESPD, 145ºC blocked silane, 155ºC 3.0 2.5 2.0 1.5 Log shear modulus G'(0.56%)-G'(100%) [MPa] filler-filler interaction 3.5 Filler-filler Filler-filler interaction interaction Log shear deformation 1.0 0.5 Payne effect: 0.0 0 2 4 6 8 10 Silanzation time [minutes] 12 14 Destruction of the filler network (filler-filler interaction) at strains > 1% results in modulus decrease Dierkes, W., University Twente, thesis: Economic mixing of silica-rubber compounds, ISBN 90-365-2185-8, 2005 Possibilities: Coupling agent 17 Different types of coupling agents 90 60 135°C 30 0 1.0 2.5 5.0 Silanization time [minutes] 155°C 150 ML(1+4), 100ºC [MU] 145°C 120 165°C 90 60 Blocked silane 30 G'(0,56%) - G'(100%) [MPa] TESPD 120 2.5 2.0 1.5 1.0 0.5 0.0 1.0 2.5 5.0 Silanization time [minutes] G'(0,56%) - G'(100%) [MPa] ML(1+4), 100ºC [MU] 150 2.5 2.0 1.5 1.0 0.5 0.0 0 1.0 2.5 5.0 Silanization time [minutes] ML(1+4), 100ºC [MU] 150 B 120 90 60 Oligomeric silane 30 0 1.0 2.5 5.0 Silanization time [minutes] G'(0,56%) - G'(100%) [MPa] 1.0 2.5 5.0 Silanization time [minutes] 2.5 2.0 1.5 1.0 0.5 0.0 1.0 2.5 5.0 Silanization time [minutes] Dierkes, W., University Twente, thesis: Economic mixing of silica-rubber compounds, ISBN 90-365-2185-8, 2005 Payne effect Viscosity Possibilities: Surface modification 18 Plasma polymerization: film characteristics Monomer Plasma-Polymer - highly crosslinked - two-dimensional network - disordered structure - thermally, chemically stable - very adherent - bulk properties preserved B Tailored interphases between filler and polymer for better - compatibility - wettability - dispersion - stability Possibilities: Surface modification 19 Plasma polymerization: filler morphology Untreated silica M. Tiwari, University Twente, thesis; to be published in 2010 Polyacetylene-treated silica Possibilities: Surface modification 20 Plasma polymerization: Reduction in polarity M. Tiwari, University Twente, thesis; to be published in 2010 Weight of water penetration (g) 2,0 Untreated silica 1,8 Polyacetylene coated silica Polypyrrole coated silica 1,6 Polythiofene coated silica 1,4 1,2 1,0 0,8 0,6 0,4 0,2 0,0 0 2 4 6 8 10 Time (minutes) 12 14 16 Possibilities: Surface modification 21 Plasma polymerization: Polymer blend recipe PA: polyacetylene S: S-SBR E: EPDM PPy: polypyrrole Components T: silane (TESPD) PTh: polythiophene SEU SEPA SEPTh SEPPy SET phr phr phr phr phr S-SBR 50 50 50 50 50 EPDM 50 50 50 50 50 Silica 50 50 50 50 50 ZnO 2.5 2.5 2.5 2.5 2.5 Stearic acid 2.5 2.5 2.5 2.5 2.5 -- -- -- -- 4 Sulfur 1.5 1.5 1.5 1.5 1.04 CBS 1.5 1.5 1.5 1.5 1.5 DPG S: 1.5 SE:0.75 S: 1.5 SE:0.75 S: 1.5 SE:0.75 S: 1.5 SE:0.75 S: 1.5 SE:0.75 TMTD 0.4 0.4 0.4 0.4 0.4 ZBEC 0.75 0.75 0.75 0.75 0.75 Silane (TESPT) Possibilities: Surface modification 22 Plasma polymerization: properties 60 0,4 0,3 0,2 0,1 0,0 10 Reinforcement parameter 0,6 Bound rubber content (%) 50 40 30 20 SPA SPPy SPTh ST SU SPA SPPy SPTh ST 0 SU SPA 0,5 0,4 0,3 0,2 0,1 ST 8 50 6 40 30 4 20 2 10 0 0 SEU 0,0 SEPTh SPTh Reinforcement parameter Bound rubber content (%) 0,6 SPPy 10 60 0,7 SEPPy 2 Sample code 70 SEPA 4 Sample code 0,8 G'(0.56%) - G'(100.04%) [MPa] SBR-EPDM blend Sample code SEU 6 10 0 SU 8 SET Sample code Bound rubber: Polymer chains are adsorbed / bond to the filler surface SEPA SEPPy SEPTh Sample code SET SEU SEPA SEPPy SEPTh SET Sample code Smax − Smin m − 1 = αF F 0 0 S max − S min mP The lower αF, Smax Torque G'(0.56%) - G'(100.04%) [MPa] Straight SBR 70 0,5 Dispersion Filler-polymer interaction M. Tiwari, University Twente, thesis; to be published in 2010 Filler-filler interaction 0,7 the better the dispersion Smin Time Possibilities: Surface modification 23 Plasma polymerization Straight SBR 28 SU SPA SPPy SPTh ST 20 SEU SEPA SEPTH SEPPy SET 14 Stress (MPa) 24 Stress (MPa) SBR - EPDM blend 16 16 12 12 10 8 6 8 4 4 2 0 0 0 100 200 300 400 Strain (%) 500 600 700 800 0 50 100 150 200 250 300 Strain (%) M. Tiwari, University Twente, thesis; to be published in 2010 Possibilities: Silica-carbon black combinations 24 Dual phase silica Silica domains distributed throughout the aggregates of the filler (CRX2124, Cabot) Carbon black with silica shell on the particle surface (CRX4210, Cabot) http://www.cabot-corp.com/cws/businesses.nsf/8969ddd26dc8427385256c2c004dad01/ 91da3d4fd303e09c85256c7a00502230/$FILE/CRX4000-012-FTF'01-Florida%20meeting%2001-01.pdf Possibilities: Silica-carbon black combinations 25 Dual phase silica: filler-filler interaction (Payne effect) Carbon black with silica on the particle surface (CRX4210, Cabot) Silica domains distributed throughout the aggregates of the filler (CRX2124, Cabot) OESSBR: oil extended S-SBR M.-J. Wang, M. Morris: Recent Developments in Fillers for Tire Applications; in: Current Topics in Elastomers Research, A.K. Bhomick (ed.), CRC Press, 2008 Possibilities: Silica-carbon black combinations 26 Dual phase silica: rolling resistance (tan δ) Rolling resistance Heat build-up tan δ Abrasion Low temperature properties Wet traction Temperature, ºC CSDPF: Carbon silicon dual phase filler distributed within carbon black (Cabot) Meng-Jiao Wang, Ping Zhang, Khaled Mahmud, meeting of the Rubber Division, American Chemical Society, Dallas, TX, April 4 - 6, 2000 Possibilities: In-situ silica 27 Process Sol-gel process with TEOS and n-butylamine as catalyst - Swelling of thin rubber sheets in TEOS - Immersion in an aqueous solution of n-butylamine TEOS Tetraethyl orthosilicate Particle size: appr. 10 nm to 40 nm Max. in-situ silica concentration so far: 43 phr (practical loading for e.g. tire compounds: appr. 80 phr) Y. IKEDA, Y. KAMEDA, J. Sol-Gel Sci. Technol. 31, 137–142, 2004 Possibilities: In-situ silica 28 In Natural Rubber Silica concentration: 33 phr Surface treatment: 0,5 phr γ-MPS γ-Mercaptopropyltrimethoxysilane NR-V 1 2 4 3 2 3 4 1 NR-V: vulcanized 1. NR-mix - V: mill-blended, vulcanized 2. NR-mix - γ -V: mill-blended, with γ-MPS, vulcanized 3. NR-in situ -V: in situ silica, vulcanized 4. NR-in situ - γ -V: in situ silica, with γ-MPS, vulcanized K. Murakami et al., J. Mater. Sci. 38, p. 1447 (2003) Possibilities: Particle size distribution 29 Blends with nano fillers Improving filler-filler network formation with preservation of the strong filler-polymer interaction by blends of conventional fillers + nano particles SBR compound Example: Carbon black + CNT’s LILIANE BOKOBZA J. Polym. Sci. Part B: Polymer Physics, 46, 1939–1951, 2008 30 Possibilities of silica technology Summary ¾ Silica/silane allowed to shift the magic triangle of tire performance to a higher level: a promising start ¾ Main functional difference with CB: strong (covalent) filler-polymer bond ¾ Critical aspect: morphology (good distribution and dispersion, strong polymer-filler interaction) Ö Challenge: Improved and stable polymer-filler network Ö Possibilities: + Surface modification of silica + Improved particle size distribution and structure 31 Acknowledgements M. Tiwari UT-ETE & Teijin Twaron Silica in rubber – Possibilities and Challenges Thank you for your attention! 32